Impact Factor
Theranostics 2019; 9(26):8127-8137. doi:10.7150/thno.36718 This issue Cite
Research Paper
Strategy to enhance transgene expression in proximity of amyloid plaques in a mouse model of Alzheimer's disease
1. Biological Sciences, Hurvitz Brain Sciences Research Program, Sunnybrook Research Institute, Toronto, ON, Canada
2. Department of Laboratory Medicine and Pathobiology, University of Toronto, Toronto, ON Canada
3. Department of Neurology, Georg-August-Universität Göttingen, Göttingen, Germany
4. Physical Sciences, Sunnybrook Research Institute, Toronto, ON, Canada
5. Department of Medical Biophysics, University of Toronto, Toronto, ON, Canada
Abstract
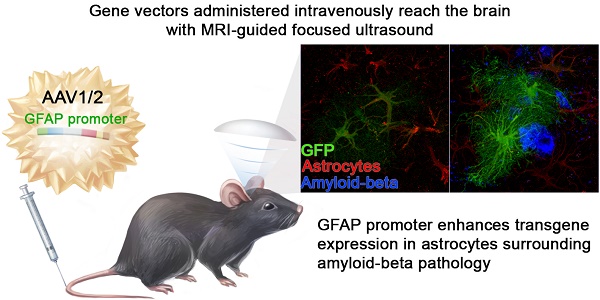
Gene therapy can be designed to efficiently counter pathological features characteristic of neurodegenerative disorders. Here, we took advantage of the glial fibrillary acidic protein (GFAP) promoter to preferentially enhance transgene expression near plaques composed of amyloid-beta peptides (Aβ), a hallmark of Alzheimer's disease (AD), in the TgCRND8 mouse model of amyloidosis.
Methods: The delivery of intravenously injected recombinant adeno-associated virus mosaic serotype 1/2 (rAAV1/2) to the cortex and hippocampus of TgCRND8 mice was facilitated using transcranial MRI-guided focused ultrasound in combination with microbubbles (MRIgFUS), which transiently and locally increases the permeability of the blood-brain barrier (BBB). rAAV1/2 expression of the reporter green fluorescent protein (GFP) under a GFAP promoter was compared to GFP expression driven by the constitutive human beta actin (HBA) promoter.
Results: MRIgFUS targeting the cortex and hippocampus facilitated the entry of rAAV1/2 and GFP expression under the GFAP promoter was localized to GFAP-positive astrocytes. Adjacent to Aβ plaques where GFAP is upregulated, the volume, surface area, and fluorescence intensity of the transgene GFP were greater in rAAV1/2-GFAP-GFP compared to rAAV1/2-HBA-GFP treated animals. In peripheral organs, GFP expression was particularly strong in the liver, irrespective of the promoter.
Conclusion: The GFAP promoter enhanced transgene expression in proximity of Aβ plaques in the brain of TgCRND8 mice, and it also resulted in significant expression in the liver. Future gene therapies for neurological disorders could benefit from using a GFAP promoter to regulate transgene expression in response to disease-induced astrocytic reactivity.
Keywords: focused ultrasound, gene expression, TgCRND8 mice, astrocytes, amyloid-beta peptides
 Global reach, higher impact
Global reach, higher impact