Impact Factor
Theranostics 2020; 10(3):1197-1212. doi:10.7150/thno.39320 This issue Cite
Research Paper
Smooth muscle SIRT1 reprograms endothelial cells to suppress angiogenesis after ischemia
1. Department of Biochemistry and Molecular Biology, College of Basic Medicine, Key Laboratory of Medical Biotechnology of Hebei Province, Hebei Medical University, Shijiazhuang, China.
2. Department of Vascular Surgery of Second Hospital, Hebei Medical University, Shijiazhuang, China.
3. Department of Gastroenterology of Second Hospital, Hebei Medical University, Shijiazhuang, China.
4. Key Laboratory of Integrative Medicine on Liver-kidney patterns of Hebei Province, College of Integrated Chinese and Western Medicine, Hebei University of Chinese Medicine, Shijiazhuang, China.
5. School of Biotechnology and Biomolecular Sciences, University of New South Wales, Sydney, Australia.
Abstract
Objective: Vascular smooth muscle cells (VSMCs) undergo the phenotypic changes from contractile to synthetic state during vascular remodeling after ischemia. SIRT1 protects against stress-induced vascular remodeling via maintaining VSMC differentiated phenotype. However, the effect of smooth muscle SIRT1 on the functions of endothelial cells (ECs) has not been well clarified. Here, we explored the role of smooth muscle SIRT1 in endothelial angiogenesis after ischemia and the underlying mechanisms.
Methods: We performed a femoral artery ligation model using VSMC specific human SIRT1 transgenic (SIRT1-Tg) and knockout (KO) mice. Angiogenesis was assessed in in vivo by quantification of the total number of capillaries, wound healing and matrigel plug assays, and in vitro ECs by tube formation, proliferation and migration assays. The interaction of HIF1α with circRNA was examined by using RNA immunoprecipitation, RNA pull-down and in situ hybridization assays.
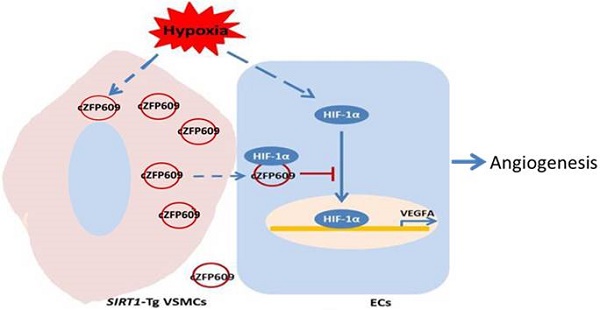
Results: The blood flow recovery was significantly attenuated in SIRT1-Tg mice, and markedly improved in SIRT1-Tg mice treated with SIRT1 inhibitor EX527 and in SIRT1-KO mice. The density of capillaries significantly decreased in the ischemic gastrocnemius of SIRT1-Tg mice compared with SIRT1-KO and WT mice, with reduced expression of VEGFA, which resulted in decreased number of arterioles. We identified that the phenotypic switching of SIRT1-Tg VSMCs was attenuated in response to hypoxia, with high levels of contractile proteins and reduced expression of the synthetic markers and NG2, compared with SIRT1-KO and WT VSMCs. Mechanistically, SIRT1-Tg VSMCs inhibited endothelial angiogenic activity induced by hypoxia via the exosome cZFP609. The cZFP609 was delivered into ECs, and detained HIF1α in the cytoplasm via its interaction with HIF1α, thereby inhibiting VEGFA expression and endothelial angiogenic functions. Meantime, the high cZFP609 expression was observed in the plasma of the patients with atherosclerotic or diabetic lower extremity peripheral artery disease, associated with reduced ankle-brachial index. Knockdown of cZFP609 improved blood flow recovery after hindlimb ischemia in SIRT1-Tg mice.
Conclusions: Our findings demonstrate that SIRT1 may impair the plasticity of VSMCs. cZFP609 mediates VSMCs to reprogram endothelial functions, and serves as a valuable indicator to assess the prognosis and clinical outcomes of ischemic diseases.
Keywords: cZFP609, angiogenesis, vascular smooth muscle cells, ischemia, exosomes.
 Global reach, higher impact
Global reach, higher impact