13.3
Impact Factor
Theranostics 2020; 10(4):1733-1745. doi:10.7150/thno.36022 This issue Cite
Research Paper
Imaging the Landmarks of Vascular Recovery
1. Department of Bioengineering, University of Illinois at Urbana-Champaign, Urbana, IL
2. Department of Electrical and Computer Engineering, University of Illinois at Urbana-Champaign, Urbana, IL
3. Department of Chemistry, University of Illinois at Urbana-Champaign, Urbana, IL
4. SimBioSys, Inc., Champaign, IL
5. Beckman Institute for Advanced Science and Technology, Urbana, IL
6. Department of Internal Medicine, Yale University School of Medicine, New Haven, CT
7. Department of Medical Laboratory Diagnostics, Medical University of Gdansk, Poland
8. Biobanking and Biomolecular Resources Research Infrastructure Poland (BBMRI.PL), Gdansk, Poland
*These authors contributed equally to this work
Abstract
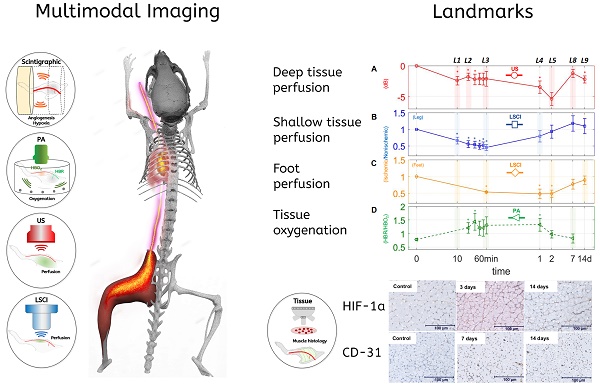
Background: Peripheral arterial disease (PAD) is a major worldwide health concern. Since the late 1990s therapeutic angiogenesis has been investigated as an alternative to traditional PAD treatments. Although positive preclinical results abound in the literature, the outcomes of human clinical trials have been discouraging. Among the challenges the field has faced has been a lack of standardization of the timings and measures used to validate new treatment approaches.
Methods: In order to study the spatiotemporal dynamics of both perfusion and neovascularization in mice subjected to surgically-induced hindlimb ischemia (n= 30), we employed three label-free imaging modalities (a novel high-sensitivity ultrasonic Power Doppler methodology, laser speckle contrast, and photoacoustic imaging), as well as a tandem of radio-labeled molecular probes, 99mTc-NC100692 and 99mTc-BRU-5921 respectively, designed to detect two key modulators of angiogenic activity, αVβ3 and HIF-1α , via scintigraphic imaging.
Results: The multimodal imaging strategy reveals a set of “landmarks”—key physiological and molecular events in the healing process—that can serve as a standardized framework for describing the impact of emerging PAD treatments. These landmarks span the entire process of neovascularization, beginning with the rapid decreases in perfusion and oxygenation associated with ligation surgery, extending through pro-angiogenic changes in gene expression driven by the master regulator HIF-1α , and ultimately leading to complete functional revascularization of the affected tissues.
Conclusions: This study represents an important step in the development of multimodal non-invasive imaging strategies for vascular research; the combined results offer more insight than can be gleaned through any of the individual imaging methods alone. Researchers adopting similar imaging strategies and will be better able to describe changes in the onset, duration, and strength of each of the landmarks of vascular recovery, yielding greater biological insight, and enabling more comprehensive cross-study comparisons. Perhaps most important, this study paves the road for more efficient translation of PAD research; emerging experimental treatments can be more effectively assessed and refined at the preclinical stage, ultimately leading to better next-generation therapies.
Keywords: Angiogenesis, hypoxia, hindlimb ischemia, 99mTc-NC100692, 99mTc-BRU-5921, perfusion, functional recovery, Power Doppler imaging.
 Global reach, higher impact
Global reach, higher impact