Impact Factor
Theranostics 2020; 10(17):7889-7905. doi:10.7150/thno.45028 This issue Cite
Research Paper
Engineering blood exosomes for tumor-targeting efficient gene/chemo combination therapy
1. Tianjin Key Laboratory of Composite and Functional Materials, School of Material Science and Engineering, Tianjin University, Tianjin 300072, China.
2. Department of Neurosurgery, Tianjin Medical University General Hospital, Laboratory of Neuro-oncology, Tianjin Neurological Institute, Key Laboratory of Post-Neurotrauma Neuro-Repair and Regeneration in Central Nervous System, Ministry of Education and Tianjin City, Tianjin 300052, China.
3. Institute for Translational Medicine, Qingdao University, Qingdao, 266021, China.
#These authors contributed equally to this work.
Abstract
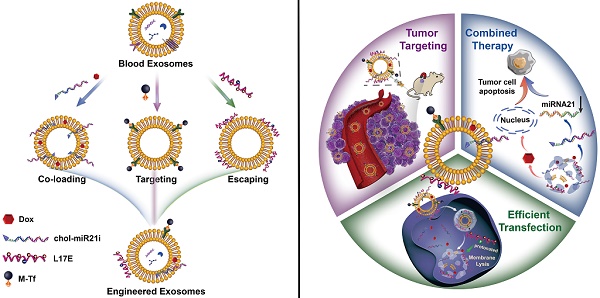
Rationale: Developing an effective nanoplatform to realize 'multi-in-one' is essential to broaden the therapeutic potential of combination therapy. Exosomes are ideal candidates since their intrinsic abilities of integrating multiple contents and functions. However, only limited efforts have been devoted to engineering exosomes to integrate the needed properties, also considering the safety and yield, for tumor-targeted and efficient gene/chemo combination therapy.
Methods: Herein, by manipulating the exosome membrane, blood exosomes with high abundance and safety are engineered as a versatile combinatorial delivery system, where the doxorubicin (Dox) and cholesterol-modified miRNA21 inhibitor (miR-21i) are co-embedded into the lipid bilayer of exosomes, and the magnetic molecules and endosomolytic peptides L17E are bind to the exosome membrane through ligand-receptor coupling and electrostatic interactions, respectively.
Results: It is proved that such engineering strategy not only preserves their intrinsic features, but also readily integrates multiple properties of tumor targeting, efficient transfection and gene/chemo combination therapy into blood exosomes. The lipid bilayer structure of exosomes allows them to co-load Dox and miR-21i with high-payloads. Moreover, profiting from the integration of magnetic molecules and L17E peptides, the engineered exosomes exhibit an enhanced tumor accumulation and an improved endosome escape ability, thereby specifically and efficiently delivering encapsulated cargos to tumor cells. As a result, a remarkable inhibition of tumor growth is observed in the tumor-bearing mice, and without noticeable side effects.
Conclusions: This study demonstrates the potential of engineered blood exosomes as feasible co-delivery nanosystem for tumor-targeted and efficient combination therapy. Further development by replacing the drugs combined regimens can potentially make this engineered exosome become a general platform for the design of safe and effective combination therapy modality.
Keywords: exosome, co-loading, tumor targeting, efficient transfection, combination therapy
 Global reach, higher impact
Global reach, higher impact