Impact Factor
Theranostics 2021; 11(3):1473-1492. doi:10.7150/thno.51245 This issue Cite
Research Paper
H3K27 acetylation activated-COL6A1 promotes osteosarcoma lung metastasis by repressing STAT1 and activating pulmonary cancer-associated fibroblasts
1. Sun Yat-sen University Cancer Center; State Key Laboratory of Oncology in South China; Collaborative Innovation Center for Cancer Medicine, Guangzhou 510060, China.
2. Department of Pathology, Sun Yat-sen University Cancer Center, Guangzhou 510060, China.
3. Department of Orthopedics, First Affiliated Hospital of Shantou University Medical College, No.57 Changping Road, Shantou, Guangdong 515041, China.
4. Department of Orthopedics, Sun Yat-sen University Cancer Center, Guangzhou 510060, China.
#These authors contributed equally to this work.
Abstract
Background: Collagen type VI alpha 1 (COL6A1) has been found to be dysregulated in several human malignancies. However, the role of COL6A1 in osteosarcoma (OS) progression remains largely unclear. Here, we aimed to explore the clinical significance and biological involvement of COL6A1 in the OS cell migration and invasion.
Material and Methods: We used immunohistochemistry, qRT-PCR and western blot to detect the expression of COL6A1 in 181 OS patient samples. Chromatin immunoprecipitation (ChIP) and PCR were carried out to verify the regulatory interaction of p300, c-Jun and COL6A1 promoter. The invasion and migration function of COL6A1 in OS was detected in vitro and in vivo. RNA sequence was performed to detect the downstream pathway of COL6A1, and then co-immunoprecipitation (co-IP), ubiquitination assays and rescue experiments were performed to determine the regulatory effect of COL6A1 and signal transducers and activators of transcription (STAT1). Exosomes derived from OS cell lines were assessed for the ability to promote cancer progression by co-cultured assay and exosomes tracing.
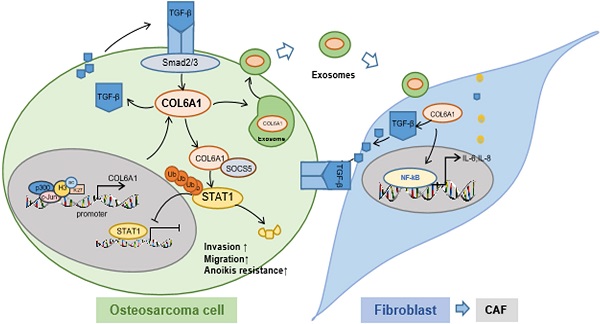
Results: COL6A1 was commonly upregulated in OS tissues, especially in lung metastasis tissues, which was associated with a poor prognosis. c-Jun bound p300 increased the enrichment of H3K27ac at the promoter region of the COL6A1 gene, which resulted in the upregulation of COL6A1 in OS. Overexpression of COL6A1 promoted OS cell migration and invasion via interacting with SOCS5 to suppress STAT1 expression and activation in an ubiquitination and proteasomal degradation manner. Most interestingly, we found that exosomal COL6A1 derived from OS cells convert normal fibroblasts to cancer-associated fibroblasts (CAFs) by secreting pro-inflammatory cytokines, including IL-6 and IL-8. The activated CAFs could promote OS cell invasion and migration by mediating TGF-β/COL6A1 signaling pathway.
Conclusion: Our data demonstrated that upregulation of COL6A1 activated by H3K27 acetylation promoted the cell migration and invasion by suppressing STAT1 pathway in OS cells. Moreover, COL6A1 can be packaged into OS cell-derived exosomes and activate CAFs to promote OS metastasis.
Keywords: COL6A1, osteosarcoma, STAT1, metastasis, cancer-associated fibroblasts
 Global reach, higher impact
Global reach, higher impact