Impact Factor
Theranostics 2021; 11(12):5939-5954. doi:10.7150/thno.58160 This issue Cite
Research Paper
Crosstalk between coagulation and complement activation promotes cardiac dysfunction in arrhythmogenic right ventricular cardiomyopathy
1. Department of Cardiac Surgery, State Key Laboratory of Cardiovascular Disease, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, China.
2. Center of Basic Research, Biomedical Research Foundation, Academy of Athens, Athens, Greece.
3. Clinical, Experimental Surgery & Translational Research Center, Biomedical Research Foundation, Academy of Athens, Athens, Greece.
* Jie Ren, Konstantinos Tsilafakis, and Liang Chen contributed equally to this work.
Abstract
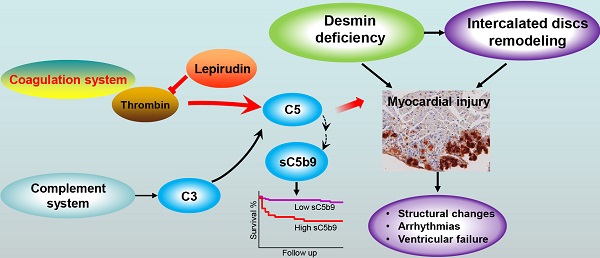
Aims: We previously found that complement components are upregulated in the myocardium of patients with arrhythmogenic right ventricular cardiomyopathy (ARVC), and inhibiting the complement receptor C5aR reduces disease severity in desmin knockout (Des-/-) mice, a model for ARVC. Here, we examined the mechanism underlying complement activation in ARVC, revealing a potential new therapeutic target.
Methods: First, immunostaining, RT-PCR and western blot were used to detect the expression levels of complement and coagulation factors. Second, we knocked out the central complement component C3 in Des-/- mice (ARVC model) by crossing Des-/- mice with C3-/- mice to explore whether complement system activation occurs independently of the conventional pathway. Then, we evaluated whether a targeted intervention to coagulation system is effective to reduce myocardium injury. Finally, the plasma sC5b9 level was assessed to investigate the role in predicting adverse cardiac events in the ARVC cohort.
Results: The complement system is activated in the myocardium in ARVC. Autoantibodies against myocardial proteins provided a possible mechanism underlying. Moreover, we found increased levels of myocardial C5 and the serum C5a in Des-/-C3-/- mice compared to wild-type mice, indicating that C5 is activated independently from the conventional pathway, presumably via the coagulation system. Crosstalk between the complement and coagulation systems exacerbated the myocardial injury in ARVC mice, and this injury was reduced by using the thrombin inhibitor lepirudin. In addition, we found significantly elevated plasma levels of sC5b9 and thrombin in patients, and this increase was correlated with all-cause mortality.
Conclusions: These results suggest that crosstalk between the coagulation and complement systems plays a pathogenic role in cardiac dysfunction in ARVC. Thus, understanding this crosstalk may have important clinical implications with respect to diagnosing and treating ARVC.
Keywords: Arrhythmogenic right ventricular cardiomyopathy, complement, coagulation, proteomics, serum biomarkers.
 Global reach, higher impact
Global reach, higher impact