Impact Factor
Theranostics 2021; 11(12):5955-5969. doi:10.7150/thno.58164 This issue Cite
Research Paper
Enhanced anti-PD-1 therapy in hepatocellular carcinoma by tumor vascular disruption and normalization dependent on combretastatin A4 nanoparticles and DC101
1. Department of Radiotherapy, The Second Hospital of Jilin University, Changchun 130041, P. R. China.
2. Key Laboratory of Polymer Ecomaterials, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun 130022, P. R. China.
3. Department of Thyroid, The Second Hospital of Jilin University, Changchun, 130041, P. R. China.
4. Department of Nephropathy, The Second Hospital of Jilin University, Changchun, 130041, P. R. China.
Abstract
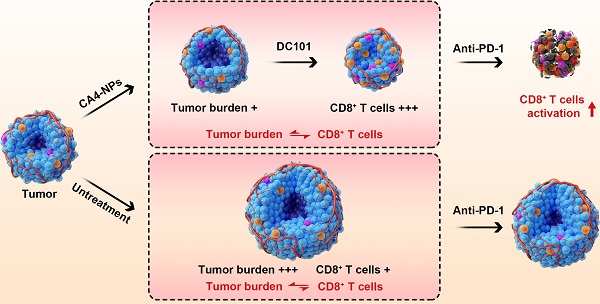
Anti-programmed cell death protein 1 (PD-1) therapy has shown promising efficacy in hepatocellular carcinoma (HCC), but its response rates in advanced HCC are lower than 20%. A critical reason for this is the imbalance between CD8+ T cells and tumor burden. Here, a novel concept of vascular disruption and normalization dependent on a polymeric vascular disrupting agent (VDA) poly (L-glutamic acid)-graft-methoxy poly (ethylene glycol)/combretastatin A4 (CA4-NPs) + a vascular endothelial growth factor (VEGF)/VEGF receptor 2 (VEGFR2) inhibitor DC101 is applied to improve anti-PD-1 therapy, wherein CA4-NPs reduce tumor burden and DC101 simultaneously increases the number of intratumoral CD8+ T cells, successfully regulating the abovementioned imbalance in an H22 tumor model.
Methods: Blood vessel density, tumor cell proliferation, and necrosis were evaluated to reveal the effects on reducing tumor burden by CA4-NP treatment. Pericyte coverage of blood vessels, tumor blood vessel perfusion, tumor hypoxia, and intratumoral immune cells were examined to verify their role in vascular normalization and immune cell homing of DC101. Furthermore, the effects of CA4-NPs + DC101 on reducing tumor burden and increasing the number of immune cells were studied. Finally, tumor suppression, intratumoral CD8+ T cell activation, and the synergistic effects of anti-PD-1 combined with CA4-NPs + DC101 were verified.
Results: The tumor inhibition rate of anti-PD-1 antibody combined with CA4-NPs + DC101 reached 86.4%, which was significantly higher than that of anti-PD-1 (16.8%) alone. Importantly, the Q value reflecting the synergy between CA4-NPs + DC101 and anti-PD-1 was 1.24, demonstrating a strong synergistic effect. Furthermore, CA4-NPs + DC101 improved anti-PD-1 therapy by increasing the number of intratumoral CD8+ T cells (anti-PD-1, 0.31% vs triple drug combination, 1.18%).
Conclusion: These results reveal a novel approach to enhance anti-PD-1 therapy with VDAs + VEGF/VEGFR2 inhibitors in HCC.
Keywords: combretastatin A4, nanoparticles, DC101, anti-PD-1 antibody, hepatocellular carcinoma
 Global reach, higher impact
Global reach, higher impact