Impact Factor
Theranostics 2021; 11(13):6393-6406. doi:10.7150/thno.53121 This issue Cite
Research Paper
Human endoglin-CD3 bispecific T cell engager antibody induces anti-tumor effect in vivo
1. National Center for International Research of Bio-targeting Theranostics, Guangxi Key Laboratory of Bio-targeting Theranostics, Guangxi Medical University, Nanning, Guangxi 530021, China. Collaborative Innovation Center for Targeting Tumor Diagnosis and Therapy, Guangxi Medical University, Nanning, Guangxi 530021, China.
2. The First People's Hospital of Changde City, Changde, Hunan 41500, China
3. Department of Oncology, The First Affiliated Hospital, Guangxi University of Chinese Medicine, Nanning, Guangxi 530023, China.
4. Department of Laboratory Medicine, The People's Hospital of Guangxi Zhuang Autonomous Region, Nanning, Guangxi 530021, China.
5. Department of Scientific Research, The Affiliated Fangchenggang Hospital, Guangxi University of Chinese Medicine, Fangchenggang, Guangxi 538001, China.
* These authors contributed equally to this work.
Abstract
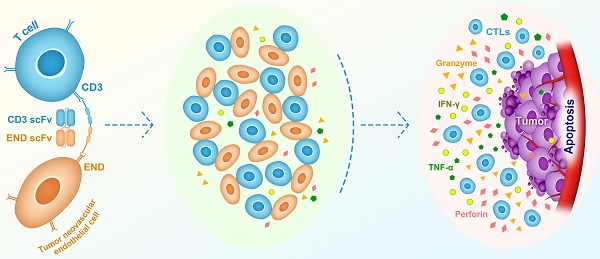
Rationale: Endoglin, also known as CD105, is a homo-dimeric membrane glycoprotein required for angiogenesis and serves as a marker for cancer vasculature. In this study, we constructed a bispecific T-cell engager (BiTE) antibody that targets human endoglin and CD3 (hEND-CD3/BiTE). We examined BiTE binding to endoglin-expressing cells and its effects on the cytolytic activity of T cells and cancer development.
Methods: The in vitro effects of hEND-CD3/BiTE, including binding to target cells, T-cell activation, proliferation, and cytotoxicity, were examined in endoglin-expressing 293T cells, human umbilical vascular endothelial cells, tumor-derived endothelial cells, and CD3+ T cells. An in vivo xenograft tumor model was established using A549 human lung cancer cells. The therapeutic efficacy of hEND-CD3/BiTE was assessed by monitoring tumor growth, angiogenesis, and mouse survival.
Results: hEND-CD3/BiTE specifically bound to endoglin-expressing cells and CD3+ T cells in vitro and stimulated T-cell activation, proliferation, and Th1 cytokine secretion, and promoted T-cell-mediated cytolysis of endoglin-expressing cells. The hEND-CD3/BiTE in vivo caused minimal toxicity to major organs, reduced tumor neoangiogenesis, inhibited tumor growth, and significantly improved mouse survival.
Conclusions: Our study demonstrated the therapeutic potential of hEND-CD3/BiTE and provided a novel approach to clinical cancer treatment.
Keywords: endoglin, bispecific T-cell engager antibody, neoangiogenesis, immune therapy.
 Global reach, higher impact
Global reach, higher impact