13.3
Impact Factor
Theranostics 2022; 12(5):2322-2334. doi:10.7150/thno.70568 This issue Cite
Research Paper
Turing milk into pro-apoptotic oral nanotherapeutic: De novo bionic chiral-peptide supramolecule for cancer targeted and immunological therapy
1. Department of Medical Oncology, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, PR. China.
2. Department of Talent Highland, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, PR. China.
3. Institute for Stem Cell & Regenerative Medicine, The Second Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710004, China
4. General Surgery Department, Tang Du Hospital, Fourth Military Medical University, 710032 Xi'an, Shaanxi, China
5. National & Local Joint Engineering Research Center of Biodiagnosis and Biotherapy, The Second Affiliated Hospital of Xi'an Jiaotong University, Xi'an, 710004, PR. China.
*These authors contributed equally.
Abstract
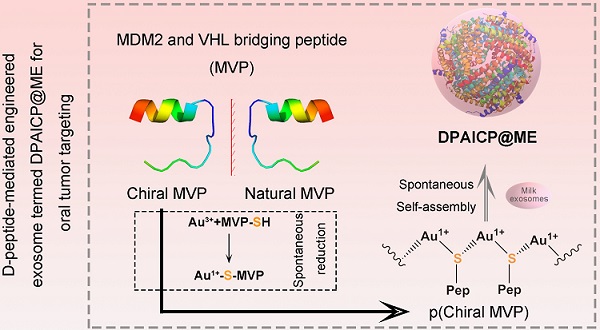
Chirality in biomolecules is ubiquitous in our world, but oral nanomedicines constructed from chiral peptides are extremely rare, principally because of the immature nanofabrication and inadequate bioavailability of chiral nanostructures.
Methods: To realize the oral administration of chiral peptides and break through their forbidden zone in intracellular space, a chiral-peptide supramolecular (DPAICP) camouflaging with the membrane from milk-derived extracellular vesicles (ME) was developed herein through an aqueous-based growth method of chiral peptide Au(I) infinite covalent polymer (DPAICP) involving in organothiol D-peptides and Au3+, and a feasible camouflage technology using ME.
Results: DPAICP@ME possessed favorable pharmaceutical properties to remain stable during the gastrointestinal absorption and blood circulation, and showed the satisfactory tumor accumulation through oral medication. Expectedly, oral DPAICP@ME played its predetermined role in vivo to restore p53 signaling pathway for cancer therapy in B16F10 homograft malignant melanoma model, LLC Lewis orthotopic transplantation model of lung cancer and patient-derived orthotopic xenograft (PDOX) mice model of colon cancer. Moreover, oral DPAICP@ME augmented the action of immunotherapy by Anti-PD1 through the further T-cell activation.
Conclusion: The de novo design of the bionic chiral-peptide supramolecule provides a practicable strategy for the construction of biomimetic chiral peptide-derived nanostructures that can be taken orally, and likely boosts chiral nanomedicine discovery efforts for a wider range of diseases including cancer.
Keywords: Chirality, Peptide, Biomimetic supramolecule, Oral medication and Cancer therapy
 Global reach, higher impact
Global reach, higher impact