Impact Factor
Theranostics 2015; 5(8):818-833. doi:10.7150/thno.11335 This issue Cite
Research Paper
A Light-Driven Therapy of Pancreatic Adenocarcinoma Using Gold Nanorods-Based Nanocarriers for Co-Delivery of Doxorubicin and siRNA
1. School of Electrical and Electronic Engineering, Nanyang Technological University, Singapore 639798, Singapore
2. Division of Structural Biology & Biochemistry, School of Biological Sciences, Nanyang Technological University, Singapore 639798, Singapore
3. Laboratory of Chemical Genetics, School of Chemical Biology and Biotechnology, Peking University Shenzhen Graduate School, Shenzhen, 518055, China
4. Nanomedicine Program and Institute of Biological Chemistry, Academia Sinica, Nankang, Taipei 115, Taiwan
5. The key lab of Biomedical Engineering and Research Institute of Uropoiesis and Reproduction, School of Medical Sciences, Shenzhen University, Shenzhen, 518060, China
6. CINTRA CNRS/NTU/THALES, UMI 3288, Research Techno Plaza, 50 Nanyang Drive, Border X Block, Singapore, 637553
7. School of Science, Changchun University of Science and Technology, Changchun, 130022, China
8. Institute of Research and Continuing Education, Hong Kong Baptist University (Shenzhen), Shenzhen 518057, China
9. Department of Genetic Engineering, College of Life Sciences, Kyung Hee University, Yongin-si Gyeonggi-do, 446-701, Republic of Korea
# These authors contributed equally to this work
Abstract
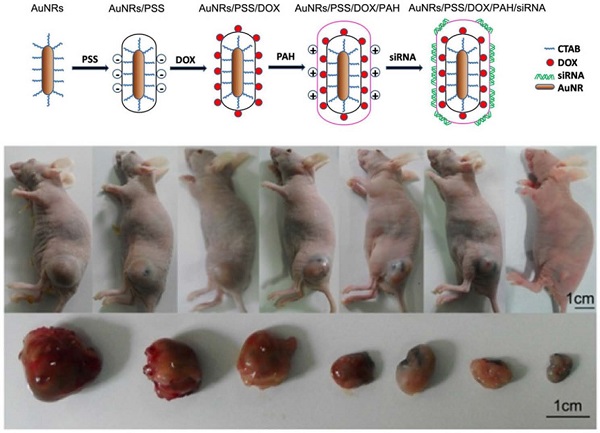
In this work, we report the engineering of polyelectrolyte polymers coated Gold nanorods (AuNRs)-based nanocarriers that are capable of co-delivering small interfering RNA (siRNA) and an anticancer drug doxorubicin (DOX) to Panc-1 cancer cells for combination of both chemo- and siRNA-mediated mutant K-Ras gene silencing therapy. Superior anticancer efficacy was observed through synergistic combination of promoted siRNA and DOX release upon irradiating the nanoplex formulation with 665 nm light. Our antitumor study shows that the synergistic effect of AuNRs nanoplex formulation with 665 nm light treatment is able to inhibit the in vivo tumor volume growth rate by 90%. The antitumor effect is contributed from the inactivation of K-Ras gene and thereby causing a profound synthesis (S) phase arrest in treated Panc-1 cells. Our study shows that the percentage of Panc-1 cells treated by nanoplex formulation with S phase is determined to be 35% and it is 17% much higher than that of Panc-1 cells without any treatments. The developed nanotherapy formulation here, that combines chemotherapy, RNA silencing and NIR window light-mediated therapy, will be seen to be the next natural step to be taken in the clinical research for improving the therapeutic outcomes of the pancreatic adenocarcinoma treatment.
Keywords: AuNRs, siRNA, K-Ras, Doxorubicin, Pancreatic adenocarcinoma, tumors
 Global reach, higher impact
Global reach, higher impact