Impact Factor
Theranostics 2016; 6(4):470-484. doi:10.7150/thno.14184 This issue Cite
Research Paper
Polydopamine-Based Surface Modification of Novel Nanoparticle-Aptamer Bioconjugates for In Vivo Breast Cancer Targeting and Enhanced Therapeutic Effects
1. The Shenzhen Key Lab of Gene and Antibody Therapy, and Division of Life and Health Sciences, Graduate School at Shenzhen, Tsinghua University, Shenzhen 518055, P.R. China;
2. School of Life Sciences, Tsinghua University, Beijing 100084, P.R. China.
3. Brigham and Women's Hospital, Harvard Medical School, Boston, Massachusetts 02115, United States.
4. Department of Biomedical Engineering, School of Engineering, Sun Yat-sen University, Guangzhou 510006, P.R. China.
5. Department of Molecular Microbiology and Immunology, University of Southern California, Los Angeles, CA 90033, USA.
* The two authors contributed equally to this work.
Abstract
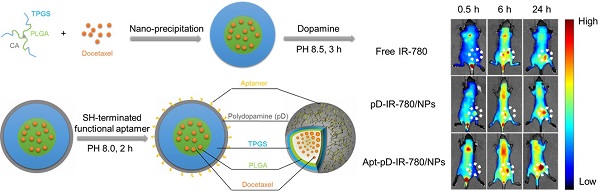
In this study, we reported a simple polydopamine (pD)-based surface modification method to prepare novel nanoparticle-aptamer bioconjugates (Apt-pD-DTX/NPs) for in vivo tumor targeting and enhanced therapeutic effects of breast cancer. With simple preparation procedures, the new functionalized Apt-pD-DTX/NPs could maximumly increase the local effective drug concentration on tumor sites, achieving enhanced treatment effectiveness and minimizing side effects. The dopamine polymerization and aptamer conjugation barely changed the characters of NPs. Both in vitro cell experiments (i.e. endocytosis of fluorescent NPs, in vitro cellular targeting and cytotoxicity assays) and in vivo animal studies (i.e. in vivo imaging, biodistribution and antitumor effects of NPs) demonstrated that the Apt-pD-DTX/NPs could achieve significantly high targeting efficiency and enhanced therapeutic effects compared with clinical Taxotere® and NPs without functional modification. Above all, the Apt-pD-DTX/NPs showed great potential as a promising nanoformulation for in vivo breast cancer therapy and the construction of pD-modified NP-aptamer bioconjugates could be of great value in medical use.
Keywords: Nanomedicine, Dopamine, Aptamer, Cancer targeting, Enhanced therapeutic effects.
 Global reach, higher impact
Global reach, higher impact