13.3
Impact Factor
Theranostics 2016; 6(4):533-544. doi:10.7150/thno.14315 This issue Cite
Research Paper
Mifepristone Suppresses Basal Triple-Negative Breast Cancer Stem Cells by Down-regulating KLF5 Expression
1. Kunming Institute of Zoology, Chinese Academy of Sciences, Key Laboratory of Animal Models and Human Disease Mechanisms of Chinese Academy of Sciences & Yunnan Province, Kunming, 650223, China;
2. Department of Neurology, First Affiliated Hospital of Kunming Medical University, Kunming, 650032, China;
3. Department of Breast surgery, the 3rd affiliated Hospital of Kunming Medical University, Kunming, Yunnan, 650118, China;
4. College of Chemistry and Chemical Engineering, Fuzhou University, Fuzhou, 350108, China;
5. Department of the second medical oncology, the 3rd affiliated Hospital of Kunming Medical University, Kunming, Yunnan, 650118, China;
6. Institute for Cancer Research, CAS Key Laboratory of Innate Immunity and Chronic Disease, School of Life Sciences and Medical Center, University of Science & Technology of China, Hefei, Anhui, 230027, China.
Abstract
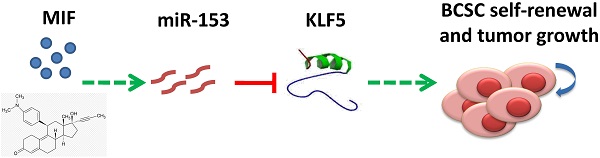
Triple-negative breast cancer (TNBC) is currently the most malignant subtype of breast cancers without effective targeted therapies. Mifepristone (MIF), a drug regularly used for abortion, has been reported to have anti-tumor activity in multiple hormone-dependent cancers, including luminal type breast cancers. In this study, we showed that MIF suppressed tumor growth of the TNBC cell lines and patient-derived xenografts in NOD-SCID mice. Furthermore, MIF reduced the TNBC cancer stem cell (CSC) population through down-regulating KLF5 expression, a stem cell transcription factor over-expressed in basal type TNBC and promoting cell proliferation, survival and stemness. Interestingly, MIF suppresses the expression of KLF5 through inducing the expression of miR-153. Consistently, miR-153 decreases CSC and miR-153 inhibitor rescued MIF-induced down-regulation of the KLF5 protein level and CSC ratio. Taken together, our findings suggest that MIF inhibits basal TNBC via the miR-153/KLF5 axis and MIF may be used for the treatment of TNBC.
Keywords: Triple-negative Breast Cancer, Mifepristone, Cancer Stem Cell, KLF5, miR-153.
 Global reach, higher impact
Global reach, higher impact