Impact Factor
Theranostics 2016; 6(11):1792-1809. doi:10.7150/thno.14584 This issue Cite
Research Paper
Peripheral Blood Cell Gene Expression Diagnostic for Identifying Symptomatic Transthyretin Amyloidosis Patients: Male and Female Specific Signatures
1. Department of Molecular and Experimental Medicine, The Scripps Research Institute, 10550 N. Torrey Pines Road, La Jolla, CA 92037.
2. Hospital de Santo António, Porto, Portugal.
3. Department of Chemistry and The Skaggs Institute for Chemical Biology, The Scripps Research Institute, La Jolla, CA 92037.
Abstract
Background: Early diagnosis of familial transthyretin (TTR) amyloid diseases remains challenging because of variable disease penetrance. Currently, patients must have an amyloid positive tissue biopsy to be eligible for disease-modifying therapies. Endomyocardial biopsies are typically amyloid positive when cardiomyopathy is suspected, but this disease manifestation is generally diagnosed late. Early diagnosis is often difficult because patients exhibit apparent symptoms of polyneuropathy, but have a negative amyloid biopsy. Thus, there is a pressing need for an additional early diagnostic strategy for TTR-aggregation-associated polyneuropathy and cardiomyopathy.
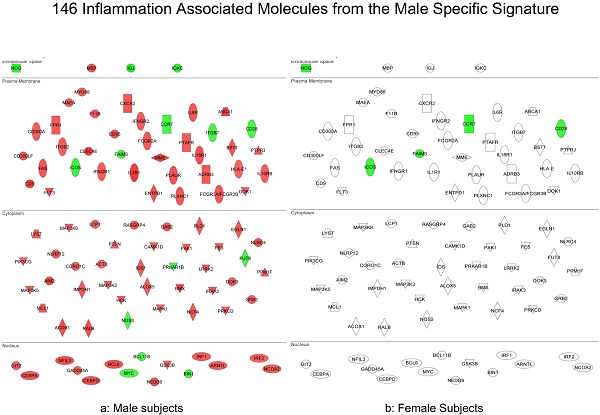
Methods and Findings: Global peripheral blood cell mRNA expression profiles from 263 tafamidis-treated and untreated V30M Familiar Amyloid Neuropathy patients, asymptomatic V30M carriers, and healthy, age- and sex-matched controls without TTR mutations were used to differentiate symptomatic from asymptomatic patients. We demonstrate that blood cell gene expression patterns reveal sex-independent, as well as male- and female-specific inflammatory signatures in symptomatic FAP patients, but not in asymptomatic carriers. These signatures differentiated symptomatic patients from asymptomatic V30M carriers with >80% accuracy. There was a global downregulation of the eIF2 pathway and its associated genes in all symptomatic FAP patients. We also demonstrated that the molecular scores based on these signatures significantly trended toward normalized values in an independent cohort of 46 FAP patients after only 3 months of tafamidis treatment.
Conclusions: This study identifies novel molecular signatures that differentiate symptomatic FAP patients from asymptomatic V30M carriers as well as affected males and females. We envision using this approach, initially in parallel with amyloid biopsies, to identify individuals who are asymptomatic gene carriers that may convert to FAP patients. Upon further validation, peripheral blood cell mRNA expression profiling could become an independent early diagnostic. This quantitative gene expression signature for symptomatic FAP could also become a biomarker to demonstrate significant disease-modifying effects of drugs and drug candidates. For example, when new disease modifiers are being evaluated in a FAP clinical trial, such surrogate biomarkers have the potential to provide an objective, quantitative and mechanistic molecular diagnostic of disease response to therapy.
Keywords: familial transthyretin
 Global reach, higher impact
Global reach, higher impact