Impact Factor
Theranostics 2016; 6(11):1887-1898. doi:10.7150/thno.16358 This issue Cite
Research Paper
Targeting Negative Surface Charges of Cancer Cells by Multifunctional Nanoprobes
1. The Institute for Translational Nanomedicine, Shanghai East Hospital, The Institute for Biomedical Engineering & Nano Science, Tongji University School of Medicine, Shanghai, 200120, China;
2. Department of Biomaterials, College of Materials, Xiamen University, Xiamen 361005, China;
3. Institutes of Biomedical Sciences, Fudan University, Shanghai, 200032, China;
4. Public Health Sciences, University of Miami School of Medicine, Miami, FL 33136, USA;
5. Department of Geological Sciences, School of Earth, Energy & Environmental Sciences, Stanford University, Stanford, CA 94305-2115, USA;
6. Materials Science and Engineering Program, Department of Mechanical and Materials Engineering, College of Engineering and Applied Science, University of Cincinnati, Cincinnati, Ohio, USA;
7. Department of Pathology, Wake Forest University School of Medicine, Winston-Salem, North Carolina, USA.
* These authors contributed equally.
Abstract
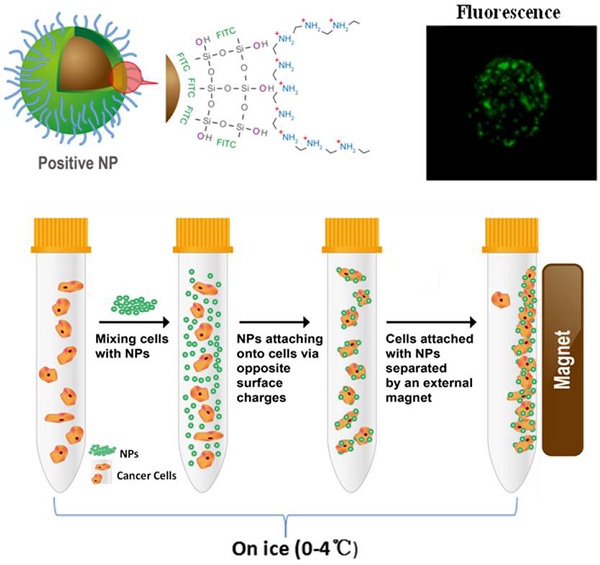
A set of electrostatically charged, fluorescent, and superparamagnetic nanoprobes was developed for targeting cancer cells without using any molecular biomarkers. The surface electrostatic properties of the established cancer cell lines and primary normal cells were characterized by using these nanoprobes with various electrostatic signs and amplitudes. All twenty two randomly selected cancer cell lines of different organs, but not normal control cells, bound specifically to the positively charged nanoprobes. The relative surface charges of cancer cells could be quantified by the percentage of cells captured magnetically. The activities of glucose metabolism had a profound impact on the surface charge level of cancer cells. The data indicate that an elevated glycolysis in the cancer cells led to a higher level secretion of lactate. The secreted lactate anions are known to remove the positive ions, leaving behind the negative changes on the cell surfaces. This unique metabolic behavior is responsible for generating negative cancer surface charges in a perpetuating fashion. The metabolically active cancer cells are shown to a unique surface electrostatic pattern that can be used for recovering cancer cells from the circulating blood and other solutions.
Keywords: Targeting, Biomarker, Nanoprobe, Surface Charge, Cell Metabolism, Lactate Secretion, Glycolysis.
 Global reach, higher impact
Global reach, higher impact