Impact Factor
Theranostics 2016; 6(13):2337-2351. doi:10.7150/thno.14937 This issue Cite
Research Paper
NIR-Laser-Controlled Drug Release from DOX/IR-780-Loaded Temperature-Sensitive-Liposomes for Chemo-Photothermal Synergistic Tumor Therapy
1. Paul C. Lauterbur Research Center for Biomedical Imaging, Institute of biomedical and Health Engineering, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China;
2. Department of Ultrasound, Guangzhou General Hospital of Guangzhou Military Command, Guangzhou, China;
3. Department of Ultrasonography, The Third Affiliated Hospital of Southern Medical University (Academy of Orthopedics Guangdong Province), Guangzhou, China;
4. Department of Ultrasonography, Shenzhen People's Hospital & Second Clinical Medical College of Jinan University, Shenzhen, China;
5. Shenzhen Key Laboratory of Nanobiomechanics, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China.
*Both authors contributed equally to this paper.
Abstract
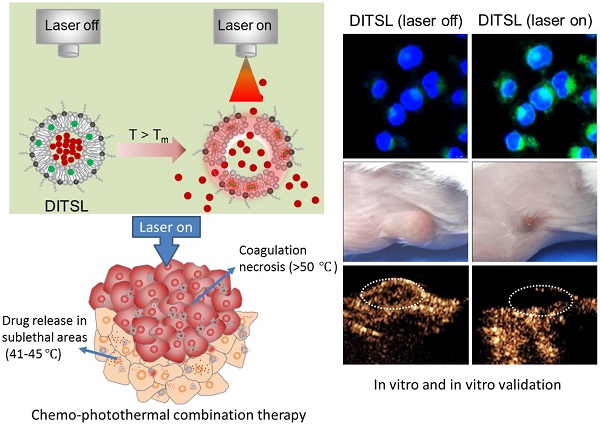
NIR laser-induced photothermal therapy (PTT) through near-infrared agents has demonstrated the great potential in solid tumor ablation. However, the nonuniform heat distribution over tumors from PTT makes it insufficient to kill all tumor cells, resulting in tumor recurrence and inferior outcomes. To improve the tumor treatment efficacy, it is highly desirable to develop the combinational treatment of PTT with other modalities, especially with chemotherapeutic agents. Here we report a smart DOX/IR-780-loaded temperature-sensitive-liposome (DITSL) which can achieve NIR-laser-controlled drug release for chemo-photothermal synergistic tumor therapy. In this system, the liposoluble IR-780 was incorporated into the temperature-sensitive lipid bilayer and the soluble chemotherapeutic doxorubicin (DOX) was encapsulated in the hydrophilic core. The resulting DITSL is proved to be physiologically stable and can provide a fast and laser irradiation-controllable DOX release in the PBS and cellular conditions. We further employed this nanoparticle for tumor treatment, demonstrating significantly higher tumor inhibition efficacy than that of DOX-loaded temperature-sensitive-liposome (DTSL) or IR780-loaded temperature-sensitive-liposome (ITSL) in the in vitro cells and in vivo animals. Histological analysis further revealed much more apoptotic cells, confirming the advantageous anti-tumor effect of DITSL over DTSL or ITSL. Our study provides a promising strategy to realize chemo-photothermal synergistic combination therapy for breast tumors.
Keywords: Photothermal therapy, Temperature-sensitive-liposome, IR-780, Doxorubicin, Breast tumors.
 Global reach, higher impact
Global reach, higher impact