Impact Factor
Theranostics 2017; 7(2):400-412. doi:10.7150/thno.16767 This issue Cite
Research Paper
Limited Tumor Tissue Drug Penetration Contributes to Primary Resistance against Angiogenesis Inhibitors
1. National Korányi Institute of Pulmonology, Budapest, Hungary;
2. Division of Thoracic Surgery, Department of Surgery, Comprehensive Cancer Center, Medical University of Vienna, Austria;
3. Clinical Protein Science&Imaging, Biomedical Center, Dept. of Biomedical Engineering, Lund University, Sweden;
4. Department of Thoracic Surgery, Semmelweis University and National Institute of Oncology, Budapest, Hungary;
5. Department of Pharmacology and Toxicology, University of Texas Medical Branch, Galveston, TX, USA;
6. Division of Oncology and Pathology, Department of Clinical Sciences, Lund University, Sweden;
7. Department of Experimental Pharmacology, National Institute of Oncology, Budapest, Hungary;
8. Department of Biomedical Imaging and Image-guided Therapy, Division of Molecular and Gender Imaging, Medical University of Vienna, Vienna, Austria;
9. Department of Thoracic Surgery, Ruhrlandklinik, University Clinic Essen, Germany;
10. Hungarian Academy of Sciences-Semmelweis University, Tumor Progression Research Group, Budapest, Hungary;
11. 1st Institute of Pathology and Experimental Cancer Research, Semmelweis University, Budapest, Hungary;
12. Center of Excellence in Biological and Medical Mass Spectrometry, Lund University, Sweden
GMV and BD contributed equally as senior authors to this study.
Abstract
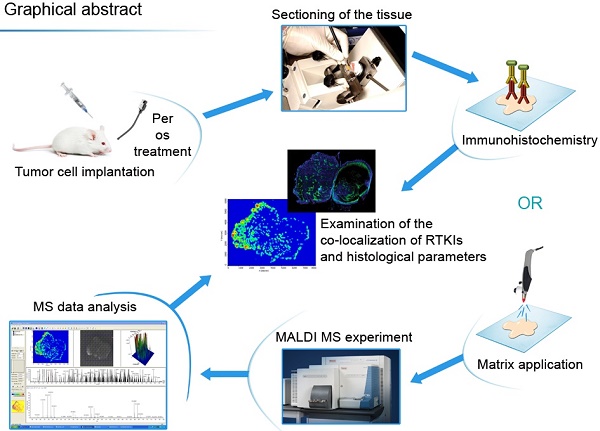
Resistance mechanisms against antiangiogenic drugs are unclear. Here, we correlated the antitumor and antivascular properties of five different antiangiogenic receptor tyrosine kinase inhibitors (RTKIs) (motesanib, pazopanib, sorafenib, sunitinib, vatalanib) with their intratumoral distribution data obtained by matrix-assisted laser desorption ionization mass spectrometry imaging (MALDI-MSI). In the first mouse model, only sunitinib exhibited broad-spectrum antivascular and antitumor activities by simultaneously suppressing vascular endothelial growth factor receptor-2 (VEGFR2) and desmin expression, and by increasing intratumoral hypoxia and inhibiting both tumor growth and vascularisation significantly. Importantly, the highest and most homogeneous intratumoral drug concentrations have been found in sunitinib-treated animals. In another animal model, where - in contrast to the first model - vatalanib was detectable at homogeneously high intratumoral concentrations, the drug significantly reduced tumor growth and angiogenesis. In conclusion, the tumor tissue penetration and thus the antiangiogenic and antitumor potential of antiangiogenic RTKIs vary among the tumor models and our study demonstrates the potential of MALDI-MSI to predict the efficacy of unlabelled small molecule antiangiogenic drugs in malignant tissue. Our approach is thus a major technical and preclinical advance demonstrating that primary resistance to angiogenesis inhibitors involves limited tumor tissue drug penetration. We also conclude that MALDI-MSI may significantly contribute to the improvement of antivascular cancer therapies.
Keywords: matrix assisted laser desorption ionization, imaging mass spectrometry, angiogenesis, receptor tyrosine kinase inhibitor, resistance, cancer
 Global reach, higher impact
Global reach, higher impact