Impact Factor
Theranostics 2017; 7(6):1650-1662. doi:10.7150/thno.17602 This issue Cite
Research Paper
Tumor-triggered drug release from calcium carbonate-encapsulated gold nanostars for near-infrared photodynamic/photothermal combination antitumor therapy
1. Institute of Nano Biomedicine and Engineering, Key Laboratory for Thin Film and Micro fabrication of the Ministry of Education, Department of Instrument Science and Engineering, School of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, P. R. China;
2. School of Biomedical Engineering, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, P. R. China;
3. Institute of Refrigeration and Cryogenics, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, P. R. China;
4. National Center for Translational Medicine, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, P. R. China;
5. School of Life Science and Technology, Xi'an Jiaotong University, 28 Xianning Xi Road, Shanxi 710049, P. R. China.
Abstract
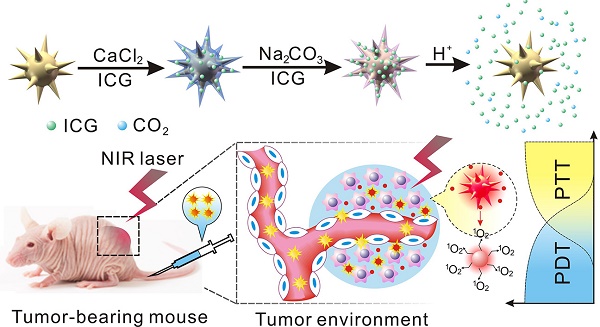
Different stimulus including pH, light and temperature have been used for controlled drug release to prevent drug inactivation and minimize side-effects. Herein a novel nano-platform (GNS@CaCO3/ICG) consisting of calcium carbonate-encapsulated gold nanostars loaded with ICG was established to couple the photothermal properties of gold nanostars (GNSs) and the photodynamic properties of indocyanine green (ICG) in the photodynamic/photothermal combination therapy (PDT/PTT). In this study, the calcium carbonate worked not only a drug keeper to entrap ICG on the surface of GNSs in the form of a stable aggregate which was protected from blood clearance, but also as the a pH-responder to achieve highly effective tumor-triggered drug release locally. The application of GNS@CaCO3/ICG for in vitro and in vivo therapy achieved the combined antitumor effects upon the NIR irradiation, which was superior to the single PDT or PTT. Meanwhile, the distinct pH-triggered drug release performance of GNS@CaCO3/ICG implemented the tumor-targeted NIR fluorescence imaging. In addition, we monitored the bio-distribution and excretion pathway of GNS@CaCO3/ICG based on the NIR fluorescence from ICG and two-photon fluorescence and photoacoustic signal from GNSs, and the results proved that GNS@CaCO3/ICG had a great ability for tumor-specific and tumor-triggered drug release. We therefore conclude that the GNS@CaCO3/ICG holds great promise for clinical applications in anti-tumor therapy with tumor imaging or drug tracing.
Keywords: Calcium carbonate, Gold nanostars, Tumor-triggered, Combination antitumor therapy.
 Global reach, higher impact
Global reach, higher impact