Impact Factor
Theranostics 2017; 7(7):2092-2107. doi:10.7150/thno.19884 This issue Cite
Research Paper
c-Myc is Required for BRAFV600E-Induced Epigenetic Silencing by H3K27me3 in Tumorigenesis
1. Department of Endocrinology, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, P.R. China;
2. Key Laboratory for Tumor Precision Medicine of Shaanxi Province, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, P.R. China;
3. Center for Translational Medicine, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, P.R. China;
4. Department of Neurosurgery, Tangdu Hospital, Fourth Military Medical University, Xi'an 710038, P.R. China.
Abstract
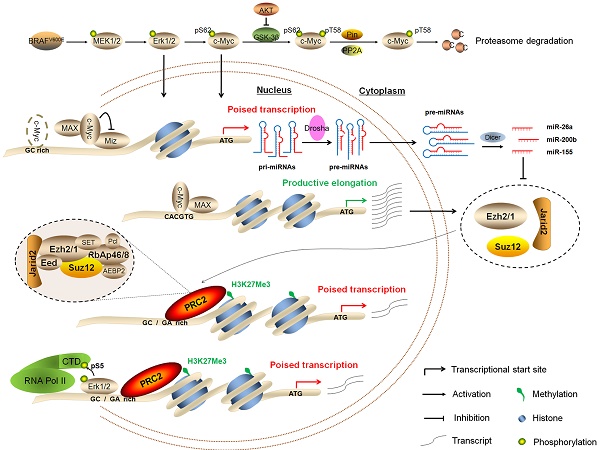
BRAFV600E mutation is frequently found in human cancers particularly thyroid cancer and melanoma, and is involved in the regulation of gene expression through activating MAPK/Erk signaling. Trimethylation of histone 3 lysine 27 (H3K27me3) is a critical epigenetic mark for the maintenance of gene silencing in tumorigenesis. However, molecular mechanism underlying the complex interplay between these two molecular events remains to be explored. In the present study, we conducted chromatin immunoprecipitation combined with next-generation sequencing (ChIP-Seq) and expression microarray analysis in NIH3T3 cells to explore the relationship between H3K27me3 and transcriptional regulation by BRAFV600E mutation. Our results showed that activated MAPK/Erk signaling by BRAFV600E mutation was a trigger of this epigenetic processing at many downstream target genes in cancer cell lines and BrafV600E-induced thyroid cancer of transgenetic mice. By integrating ChIP-Seq and gene expression microarray data, we identified 150 down-regulated loci with increased levels of H3K27me3 in BRAF-mutant cells relative to BRAF wild-type cells. Our data also demonstrated that c-Myc, a downstream key effector of BRAFV600E signaling, was required for BRAFV600E-induced changes in H3K27me3 through regulating the components of the polycomb repressive complex 2 (PRC2) genes Ezh2, Suz12 and Jarid2 at both transcriptional levels via direct binding to their regulatory elements and post-transcriptional levels via repressing the miR-26a, miR-200b and miR-155. In addition, BRAFV600E also caused gene silencing through Erk1/2-induced RNA polymerase II (RNAPII) poising and chromatin architecture. Collectively, our data uncover a previously unknown epigenetic mechanism in the tumorigenesis of BRAFV600E-driven cancers.
Keywords: BRAFV600E, c-Myc, H3K27me3, epigenetic silencing, tumorigenesis.
 Global reach, higher impact
Global reach, higher impact