13.3
Impact Factor
Theranostics 2017; 7(16):3920-3932. doi:10.7150/thno.21389 This issue Cite
Research Paper
Decrease in Lymphoid Specific Helicase and 5-hydroxymethylcytosine Is Associated with Metastasis and Genome Instability
1. Key Laboratory of Carcinogenesis and Cancer Invasion (Ministry of Education), Xiangya Hospital, Central South University, Changsha, Hunan, 410008 China;
2. Department of Pathophysiology, Changzhi Medical College, Changzhi, Shanxi, 046000 China;
3. Cancer Research Institute, Central South University, Changsha, Hunan, 410078 China;
4. Institute of Medical Sciences, Xiangya Hospital, Central South University, Changsha, Hunan, 410008 China;
5. Department of Pathology, Xiangya Hospital, Central South University, Changsha, Hunan 410078 China;
6. Thoracic Surgery Section, Thoracic and GI Oncology Branch, Center for Cancer Research, National Cancer Institute, Bethesda, MD 20892, USA;
7. Department of Pharmacology, School of Pharmaceutical Sciences, Central South University, Changsha, Hunan 410078 China.
* equal contribution
Abstract
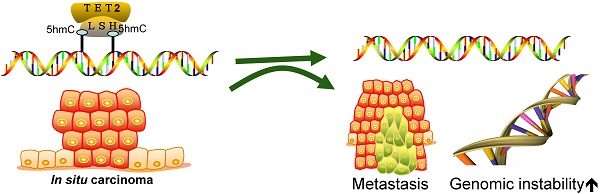
DNA methylation is an important epigenetic modification as a hallmark in cancer. Conversion of 5-methylcytosine (5-mC) to 5-hydroxymethylcytosine (5-hmC) by ten-eleven translocation (TET) family enzymes plays an important biological role in embryonic stem cells, development, aging and disease. Lymphoid specific helicase (LSH), a chromatin remodeling factor, is regarded as a reader of 5-hmC. Recent reports show that the level of 5-hmC is altered in various types of cancers. However, the change in 5-hmC levels in cancer and associated metastasis is not well defined. We report that the level of 5-hmC was decreased in metastatic tissues of nasopharyngeal carcinoma, breast cancer, and colon cancer relative to that in non-metastasis tumor tissues. Furthermore, our data show that TET2, but not TET3, interacted with LSH, whereas LSH increased TET2 expression through silencing miR-26b-5p and miR-29c-5p. Finally, LSH promoted genome stability by silencing satellite expression by affecting 5-hmC levels in pericentromeric satellite repeats, and LSH was resistant to cisplatin-induced DNA damage. Our data indicate that 5-hmC might serve as a metastasis marker for cancer and that the decreased expression of LSH is likely one of the mechanisms of genome instability underlying 5-hmC loss in cancer.
Keywords: LSH, 5-hmC, TET2, satellites, genome instability.
 Global reach, higher impact
Global reach, higher impact