Impact Factor
Theranostics 2018; 8(4):1159-1167. doi:10.7150/thno.23373 This issue Cite
Research Paper
Delivery of Sonic Hedgehog Gene Repressed Irradiation-induced Cellular Senescence in Salivary Glands by Promoting DNA Repair and Reducing Oxidative Stress
1. Institute for Regenerative Medicine, Molecular and Cellular Medicine Department, College of Medicine, Texas A&M University Health Science Center, College Station, Texas 77843, USA.
2. Department of Small Animal Clinical Sciences, College of Veterinary Medicine & Biomedical Sciences, Texas A&M University, College Station, Texas 77843, USA.
Abstract
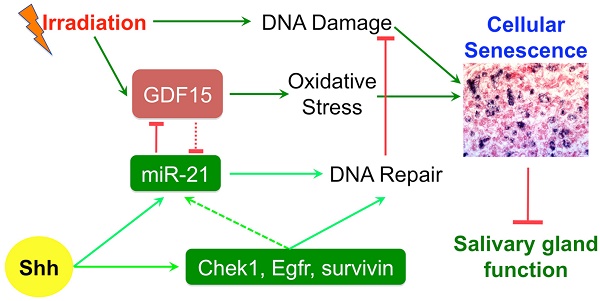
Rationale: Irreversible hypofunction of salivary glands or xerostomia is common in head and neck cancer survivors treated with radiotherapy even when various new techniques are applied to minimize the irradiation (IR) damage. This condition severely impairs the quality of life of patients and can only be temporarily relieved with current treatments. We found recently that transient expression of Sonic Hedgehog (Shh) in salivary glands after IR rescued salivary function, but the underlying mechanisms are not totally clear.
Methods: We generated a mouse model of IR-induced hyposalivation, and delivered adenoviral vectors carrying Shh or control GFP gene into submandibular glands (SMGs) via retrograde ductal instillation 3 days after IR. The cellular senescence was evaluated by senescence-associated beta-galactosidase assay and the expression of senescence markers. The underlying mechanisms were explored by examining DNA damage, oxidative stress, and the expression of related genes by qRT-PCR, Western blot and immunofluorescent staining.
Results: Shh gene transfer repressed IR-induced cellular senescence by promoting DNA repair and decreasing oxidative stress, which is mediated through upregulating expression of genes related to DNA repair such as survivin and miR-21 and repressing expression of pro-senescence gene Gdf15 likely downstream of miR-21.
Conclusion: Repressing cellular senescence contributes to the rescue of IR-induced hyposalivation by transient activation of Hh signaling, which is related to enhanced DNA repair and decreased oxidative stress in SMGs.
Keywords: Irradiation-induced hyposalivation, cellular senescence, Hedgehog signaling, DNA repair, oxidative stress
 Global reach, higher impact
Global reach, higher impact