Impact Factor
Theranostics 2021; 11(14):7029-7044. doi:10.7150/thno.60040 This issue Cite
Research Paper
Converged Rab37/IL-6 trafficking and STAT3/PD-1 transcription axes elicit an immunosuppressive lung tumor microenvironment
1. Department of Pharmacology, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
2. Department of Biochemistry, College of Medicine, National Cheng Kung University, Tainan, Taiwan College of Medicine, National Cheng Kung University, Tainan, Taiwan.
3. Institute of Basic Medical Sciences, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
4. Division of Oncology, Department of Internal Medicine, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
5. Department of Microbiology and Immunology, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
Abstract
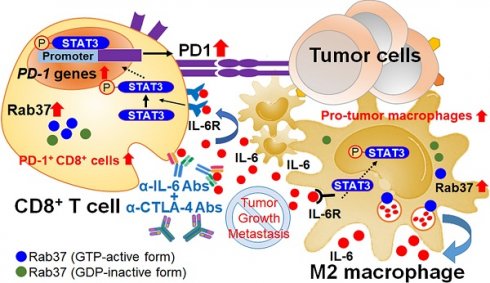
Background: Increased IL-6 level, M2 macrophages and PD-1+CD8+ T cells in tumor microenvironments (TME) have been identified to correlate with resistance to checkpoint blockade immunotherapy, yet the mechanism remains poorly understood. Rab small GTPase-mediated trafficking of cytokines is critical in immuno-modulation. We have previously reported dysregulation of Rab37 in lung cancer cells, whereas the roles of Rab37 in tumor-infiltrating immune cells and cancer immunotherapy are unclear.
Methods: The tumor growth of the syngeneic mouse allograft in wild type or Rab37 knockout mice was analyzed. Imaging analyses and vesicle isolation were conducted to determine Rab37-mediated IL-6 secretion. STAT3 binding sites at PD-1 promoter in T cells were identified by chromatin immunoprecipitation assay. Multiplex fluorescence immunohistochemistry was performed to detect the protein level of Rab37, IL-6 and PD-1 and localization of the tumor-infiltrating immune cells in allografts from mice or tumor specimens from lung cancer patients.
Results: We revealed that Rab37 regulates the secretion of IL-6 in a GTPase-dependent manner in macrophages to trigger M2 polarization. Macrophage-derived IL-6 promotes STAT3-dependent PD-1 mRNA expression in CD8+ T cells. Clinically, tumors with high stromal Rab37 and IL-6 expression coincide with tumor infiltrating M2-macrophages and PD1+CD8+ T cells that predicts poor prognosis in lung cancer patients. In addition, lung cancer patients with an increase in plasma IL-6 level are found to be associated with immunotherapeutic resistance. Importantly, combined blockade of IL-6 and CTLA-4 improves survival of tumor-bearing mice by reducing infiltration of PD1+CD8+ T cells and M2 macrophages in TME.
Conclusions: Rab37/IL-6 trafficking pathway links with IL-6/STAT3/PD-1 transcription regulation to foster an immunosuppressive TME and combined IL-6/CTLA-4 blockade therapy exerts potent anti-tumor efficacy.
Keywords: Rab37, IL-6, PD-1, transcription, tumor microenvironment
 Global reach, higher impact
Global reach, higher impact