13.3
Impact Factor
Theranostics 2022; 12(14):6258-6272. doi:10.7150/thno.74808 This issue Cite
Research Paper
Intratumoral synthesis of transformable metal-phenolic nanoaggregates with enhanced tumor penetration and retention for photothermal immunotherapy
1. BMI Center for Biomass Materials and Nanointerfaces, College of Biomass Science and Engineering, Sichuan University, Chengdu, Sichuan 610065, China.
2. Collage of Material Science and Engineering, Sichuan University, Chengdu, Sichuan 610065, China.
3. Department of Biomass Chemistry and Engineering, College of Biomass Science and Engineering, Sichuan University, Chengdu, Sichuan 610065, China.
4. Key Laboratory of Birth Defects and Related of Women and Children of Ministry of Education, Department of Pediatrics, The Reproductive Medical Center, Department of Obstetrics and Gynecology, West China Second University Hospital, Sichuan University, Chengdu, Sichuan 610041, China.
5. Department of Bioengineering, School of Engineering, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-8656, Japan.
6. Institute for Tissue Engineering and Regenerative Medicine, The Chinese University of Hong Kong (CUHK), Shatin, Hong Kong SAR, China.
7. State Key Laboratory of Polymer Materials Engineering, Sichuan University, Chengdu, Sichuan 610065, China.
8. Bioproducts Institute, Department of Chemical and Biological Engineering, University of British Columbia, Vancouver, BC V6T 1Z4, Canada.
Abstract
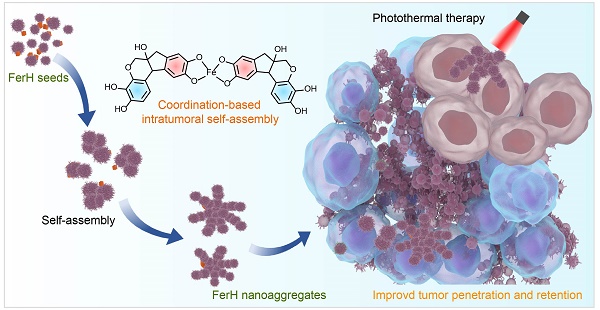
Rationale: Effective photothermal therapy (PTT) remains a great challenge due to the difficulties of delivering photothermal agents with both deep penetration and prolonged retention at tumor lesion spatiotemporally.
Methods: Here, we report an intratumoral self-assembled nanostructured aggregate named FerH, composed of a natural polyphenol and a commercial iron supplement. FerH assemblies possess size-increasing dynamic kinetics as a pseudo-stepwise polymerization from discrete nanocomplexes to microscale aggregates.
Results: The nanocomplex can penetrate deeply into solid tumors, followed by prolonged retention (> 6 days) due to the in vivo growth into nanoaggregates in the tumor microenvironment. FerH performs a targeting ablation of tumors with a high photothermal conversion efficiency (60.2%). Importantly, an enhanced immunotherapeutic effect on the distant tumor can be triggered when co-administrated with checkpoint-blockade PD-L1 antibody.
Conclusions: Such a therapeutic approach by intratumoral synthesis of metal-phenolic nanoaggregates can be instructive to address the challenges associated with malignant tumors.
Keywords: metal-phenolic coordination, intratumoral self-assembly, pseudo-stepwise kinetics, transformable materials, photothermal immunotherapy
 Global reach, higher impact
Global reach, higher impact