13.3
Impact Factor
Theranostics 2023; 13(8):2455-2470. doi:10.7150/thno.80494 This issue Cite
Research Paper
A strategy of local hydrogen capture and catalytic hydrogenation for enhanced therapy of chronic liver diseases
1. Key Laboratory of Major Diseases and Hydrogen Medical Translational Applications in Universities of Shandong Province & Key Laboratory of Hydrogen Biomedical Research of Health Commission of Shandong Province, The Second Affiliated Hospital of Shandong First Medical University & Shandong Academy of Medical Sciences, Tai'an 271000, China.
2. Taishan Institute for Hydrogen Biomedical Research, Shandong First Medical University & Shandong Academy of Medical Sciences, Tai'an 271000, China
3. School of Biomedical Engineering, Guangzhou Medical University, Guangdong, 511495 China.
4. Pathology Department of Tai'an City Central Hospital, Tai'an 271016, China.
5. Key Laboratory of Human-Machine-Intelligence Synergic System, Research Center for Neural Engineering, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, Guangdong, China.
6. Shanghai Key Laboratory of Hydrogen Science & Center of Hydrogen Science, School of Materials Science and Engineering, Shanghai Jiao Tong University, Shanghai 200240, China.
7. Shenzhen Research Institute, Shanghai Jiao Tong University, Shenzhen 518057, China.
# These authors contributed equally to this work.
Abstract
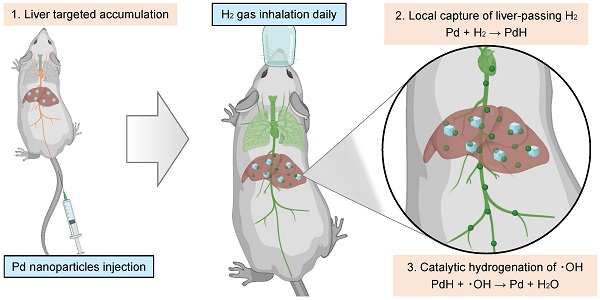
Background: Chronic liver diseases (CLD) frequently derive from hepatic steatosis, inflammation and fibrosis, and become a leading inducement of cirrhosis and hepatocarcinoma. Molecular hydrogen (H2) is an emerging wide-spectrum anti-inflammatory molecule which is able to improve hepatic inflammation and metabolic dysfunction, and holds obvious advantages in biosafety over traditional anti-CLD drugs, but existing H2 administration routes cannot realize the liver-targeted high-dose delivery of H2, severely limiting its anti-CLD efficacy.
Method: In this work, a concept of local hydrogen capture and catalytic hydroxyl radical (·OH) hydrogenation is proposed for CLD treatment. The mild and moderate non-alcoholic steatohepatitis (NASH) model mice were intravenously injected with PdH nanoparticles firstly, and then daily inhaled 4% hydrogen gas for 3 h throughout the whole treatment period. After the end of treatment, glutathione (GSH) was intramuscularly injected every day to assist the Pd excretion.
Results: In vitro and in vivo proof-of-concept experiments have confirmed that Pd nanoparticles can accumulate in liver in a targeted manner post intravenous injection, and play a dual role of hydrogen captor and ·OH filter to locally capture/store the liver-passing H2 during daily hydrogen gas inhalation and rapidly catalyze the ·OH hydrogenation into H2O. The proposed therapy significantly improves the outcomes of hydrogen therapy in the prevention and treatment of NASH by exhibiting a wide range of bioactivity including the regulation of lipid metabolism and anti-inflammation. Pd can be mostly eliminated after the end of treatment under the assistance of GSH.
Conclusion: Our study verified a catalytic strategy of combining PdH nanoparticles and hydrogen inhalation, which exhibited enhanced anti-inflammatory effect for CLD treatment. The proposed catalytic strategy will open a new window to realize safe and efficient CLD treatment.
Keywords: catalysis, controlled release, targeted delivery, nanomedicine, hydrogen therapy, chronic liver diseases
 Global reach, higher impact
Global reach, higher impact