Impact Factor
Theranostics 2026; 16(9):5016-5043. doi:10.7150/thno.130380 This issue Cite
Review
Carbon dots penetrating the blood-brain barrier for central nervous system nanomedicine
1. Department of Animal Science & Technology and BET Research Institute, Chung-Ang University, Anseong, Gyeonggi-do 17546, Republic of Korea.
2. Department of Chemistry, College of Natural Science, Wollo University, P.O. Box 1145, Dessie 1000, Ethiopia.
3. Nanochemistry Laboratory Center, Department of Chemical Engineering, National Taiwan University of Science and Technology, Taipei 106335, Taiwan.
4. Department of Chemistry, Faculty of Natural Science, Wolaita Sodo University, Wolaita Sodo, Ethiopia.
5. Department of Chemistry, College of Natural and Computational Sciences, University of Gondar, Gondar 196, Ethiopia.
6. Advanced Membrane Materials Research Centre, Graduate Institute of Applied Science and Technology, National Taiwan University of Science and Technology, Taipei 106335, Taiwan.
7. Department of Chemical Engineering, National Taiwan University, Taipei 106319, Taiwan.
Received 2025-12-22; Accepted 2026-2-3; Published 2026-2-26
Abstract
Central nervous system (CNS) diseases are challenging to treat because of the blood-brain barrier (BBB), formed by tight junctions that limit the transcellular transport of therapeutic drugs. Carbon dots (CDs) have emerged as versatile nanotheranostic platforms for the targeting, diagnosis, and treatment of CNS diseases owing to their ultrasmall size, intrinsic photoluminescence, tunable surface chemistry, and biocompatibility. Surface modifications of CDs with targeting ligands, polymer coatings, biomimetic membranes, and exosome-like molecules enable BBB penetration and selective brain accumulation. CDs also support multimodal imaging techniques, such as fluorescence, magnetic resonance, and photoacoustic imaging, for early disease detection and real-time therapeutic monitoring. In addition, their ability to deliver drugs, genes, and therapeutic agents, combined with their antioxidant, anti-inflammatory, photothermal, photodynamic, and sonodynamic properties, highlights their potential for the integrated diagnosis and treatment of CNS diseases. This review systematically summarizes the background of CDs, the design of BBB-penetrating CDs, and their applications in tumor diagnosis, treatment, and imaging-guided cooperative therapies for CNS diseases. Finally, current obstacles and future perspectives are discussed. This review provides a valuable reference for the rational design of BBB-penetrating CDs for the precise treatment of neurological disorders and brain cancers.
Keywords: blood-brain barrier, central nervous system, carbon dots, imaging, therapy
1. Introduction
Central nervous system (CNS) diseases pose a substantial threat to human health and place a substantial burden on society [1]. Although the incidence and mortality rates of peripheral diseases have declined, mortality associated with brain disorders remains elevated. As the population ages, addressing brain diseases has become increasingly critical [2, 3]. Alzheimer's disease, Parkinson's disease, epilepsy, schizophrenia, traumatic brain injury, stroke, cerebral ischemia, lysosomal storage diseases, anxiety, depression, and multiple sclerosis are major neurological disorders that affect the CNS and present formidable challenges in the development of effective treatments [4-7]. Neurodegenerative disorders (e.g., Alzheimer's and Parkinson's diseases) are characterized by abnormal protein accumulation in the brain and deterioration of cognitive and behavioral functions, resulting in cognitive impairment, neuronal death, and imbalanced glial cell activity, posing a major threat to the health and well-being of the older population [8, 9]. Brain cancer is among the most aggressive and lethal primary intracranial tumors of the CNS and is known for its poor prognosis, elevated recurrence risk, and minimal survival rates [10].
The main challenge in treating CNS disorders is the presence of a blood-brain barrier (BBB), a highly selective and tightly regulated interface comprising endothelial cells, microglia, pericytes, astrocytes, and a basement membrane. Strong interactions at tight junctions bind these cells and limit transcellular transit [11]. The BBB is essential for maintaining the CNS microenvironment because it separates circulating blood and brain interstitial fluids. It preserves brain homeostasis, facilitates communication between the central and peripheral systems, and regulates potential infections and immunological reactions [5, 12-14]. However, the tight BBB structure presents a significant obstacle to the effective delivery of therapeutic agents, limiting the treatment of CNS disorders [8]. During tumor progression, the integrity of the BBB is compromised, forming the blood-tumor barrier (BTB) [15]. Although the BTB is more permeable than the normal BBB, its uneven permeability to both small and large molecules, along with irregular blood perfusion, often leads to insufficient drug accumulation within brain tumors. Consequently, the BBB and its pathological form, the BTB, remain the major limiting factors in achieving effective clinical treatment [12, 16]. Patients with neurological disorders often require long-term drug administration, which can lead to side effects in non-target organs. Moreover, numerous drugs that are effective in treating these conditions lose their therapeutic potential because of the restrictive nature of the BBB. Consequently, treatment options for patients with neurodegenerative diseases and brain tumors remain limited [17, 18].
In recent decades, various invasive and noninvasive strategies have been developed to overcome the restrictive nature of the BBB and effectively target specific diseased areas within the brain [4, 19, 20]. Researchers have attempted to design effective nanomedicines to overcome the obstacles in the treatment of CNS diseases. The clinical application of nanomedicine has opened new avenues for selective and targeted drug delivery systems. Nanoformulations, especially carbon dots (CDs), have become a significant center of attraction to treating major CNS disorders [21]. The enhanced therapeutic efficacy of CDs is attributed to their high drug-loading capacity, long-term stability in biological fluids, biocompatibility, and ability for controlled drug release. Their ultrasmall size and unique surface chemistry enable them to effectively cross the BBB and deliver neurological drugs to the CNS [22-24]. Table 1 compares CDs with established nanocarrier systems, such as liposomes, polymer nanoparticles, inorganic nanoparticles, and exosomes, in terms of their advantages, limitations, and features that may favor BBB transport, while also acknowledging current limitations and translational considerations.
Comparison of the advantages and limitations of various nanocarriers: CDs, liposomes, polymer nanoparticles, inorganic nanoparticles, and exosomes, focusing on CNS delivery and translational feasibility
| Nanocarrier type | Advantages | Limitations | CNS delivery relevance and translational potential | Ref. |
|---|---|---|---|---|
| CDs | Ultra small size allowing BBB penetration High biocompatibility and low toxicity Intrinsic fluorescence useful for imaging Functional groups allow conjugation of various drugs and ligands | Aggregation and reduced stability in biological media Incomplete in vivo safety and clearance data Lack of clinical validation. | CDs can cross the BBB in animal models, but their CNS translation is limited by inadequate pharmacokinetic, neurotoxicity, biodegradation data, and lack of regulatory precedent. | [11, 86, 100, 110] |
| Liposomes | Excellent biocompatibility and safety profile Able to encapsulate both hydrophilic and hydrophobic drugs Several FDA-approved liposomal drugs show clinical success. | Poor stability and payload leakage Limited BBB penetration without modification Rapid reticuloendothelial system clearance and immune activation. High cost and cold-chain-dependent manufacturing. | Liposomes have poor inherent BBB permeability and require PEGylation or ligand targeting for CNS delivery, increasing complexity and limiting clinical translation. | [92, 94, 145] |
| Polymer nanoparticles | Tunable size, degradability, and release profiles Cargo protection and extended circulation Potential immunogenicity | Complex synthesis and limited reproducibility Limited BBB permeability due to size, requiring active transport | Certain polymer nanoparticles can cross the BBB via ligand- or transport-mediated mechanisms, but CNS clinical translation remains challenging. | [146, 147] |
| Inorganic nanoparticles | Excellent for imaging (optical/magnetic) and multifunctional systems Certain nanoparticles, like gold/silica, can be engineered to facilitate BBB transport. | Potential organ accumulation and toxicity Agglomeration and rapid immune clearance Unclear long-term neural effects | Although functionalized inorganic nanoparticles can cross the BBB, concerns over chronic accumulation and neurotoxicity limit their long-term CNS use. | [13, 94, 148] |
| Exosomes | Highly biocompatible, cell-derived vesicles with targeting potential Low immunogenicity as endogenous communication carriers | Non-standardized isolation and purification Storage and stability limitations Biological heterogeneity complicates dosing and regulation. | Show promise for CNS delivery due to their BBB-crossing ability, but clinical translation is limited by issues related to scale-up, batch consistency, regulation, and dose control. | [109, 149, 150] |
BBB: blood-brain barrier; CDs: carbon dots; CNS: central nervous disease; FDA: food and drug administration.
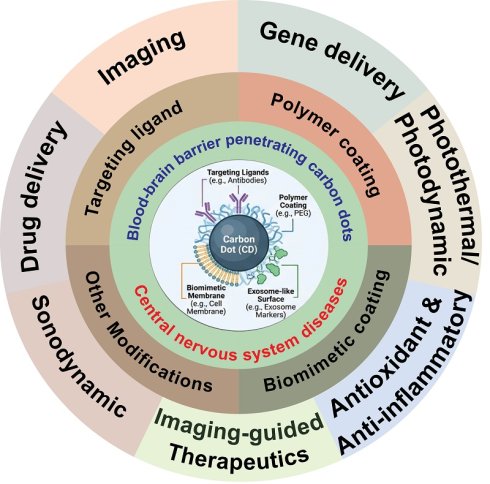
In this review, we summarize the synthesis strategies, characterizations, and properties of CDs, discuss recent advances in the design of BBB-crossing CD-based nanomedicines for targeting CNS diseases, including surface modifications using targeting ligands, polymers, biomimetic membranes, and exosomes. We briefly discuss the modification strategies for CDs to facilitate BBB penetration, enabling targeted diagnosis and treatment of CNS diseases through modalities such as imaging, chemotherapy, gene therapy, antioxidant and anti-inflammatory treatments, photothermal therapy, photodynamic therapy, sonodynamic therapy and imaging-guided therapeutics (Figure 1). Finally, we highlight the current challenges and future perspectives of using CD-based nanomedicines for treating neurological disorders and brain cancer. This review offers a focused overview of recent advancements in CD-based nanomedicines for neurotheranostics.
Schematic illustration of CD functionalization strategies to facilitate BBB penetration for imaging, therapy, or theranostic applications in CNS disease treatment. Created with BioRender.com.
2. Carbon dots
CDs were first discovered in 2004 by Xu et al. as a fluorescent nanomaterial formed as a byproduct during the purification of carbon nanotubes, where their unexpected fluorescence revealed a new class of zero-dimensional carbon-based nanomaterial [25]. Numerous researchers have explored various strategies for CD synthesis and surface engineering, as well as their widespread applications [26-28]. Early studies basically focused on their optical properties, positioning CDs as a cost-effective photostable alternative to traditional semiconductor quantum dots for imaging and sensing applications [29]. Subsequent research advanced CD synthesis through particle-size control, surface functionalization, and heteroatom doping, improving quantum yield and emission tunability [27] and marking the transition of CDs from incidental fluorescent probes to rationally designed nanoplatforms. In recent years, CDs have been explored as multifunctional nanodrug delivery systems owing to their ultrasmall size, aqueous dispersibility, tunable photoluminescence, low toxicity, and emerging capability to cross biological barriers such as the BBB, expanding their role from traditional imaging agents to theranostic platforms [11, 23, 30, 31].
In this section, we introduce representative synthesis methods, characterization techniques, and optical properties of CDs relevant to surface functionalization. A detailed discussion of other aspects—such as fluorescence mechanisms, particle size, surface charge, and other properties—is beyond the scope of this review. Readers interested in a comprehensive coverage of these topics are referred to the existing review articles by Yue et al. [32], Ge et al. [33], Girum et al. [34], Alafeef et al. [35], Yan et al. [36], Sharma et al. [37] and related works.
2.1. Synthesis methods of CDs
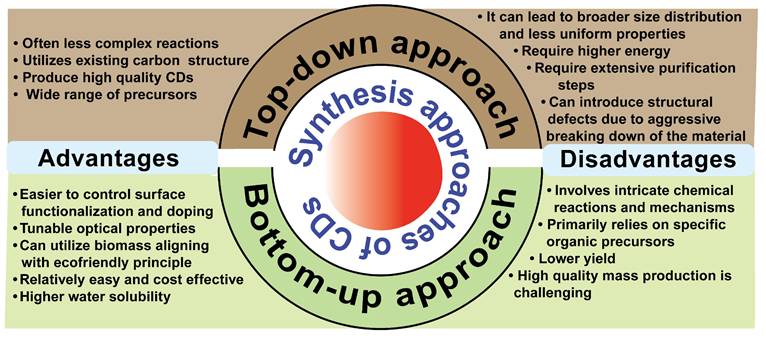
Understanding the synthesis methods is important for tailoring CDs for specific biomedical applications. CDs can be synthesized through top-down or bottom-up approaches. Top-down approaches, including arc-discharge [25], plasma treatment [38], and laser ablation [39], break bulk carbon materials into small nano fragments, where the particle size and structure of CDs are controlled by adjusting physical parameters. However, its disadvantages include using harsh chemicals and physical conditions, producing uneven size distributions, irregular surface functionalities, and requirement for expensive equipment in some techniques [40]. In contrast, bottom-up approaches, including pyrolysis microwave [30], hydrothermal [41], and ultrasonication [42], enable eco-friendly production, tunable composition, and tailored surface functional groups by the choice of precursors and reaction conditions. It still suffers from broad size distributions and batch-to-batch variability in fluorescence and quantum yield [40]. We summarize the advantages and disadvantages of top-down and bottom-up methods in Figure 2.
Advantages and disadvantages of top-down and bottom-up synthesis approaches of CDs.
In the synthesis of CDs, multiple interconnected parameters should be finely tuned to achieve a desirable size, photoluminescence, surface chemistry, and biocompatibility [43]. Accordingly, the choice of synthesis method governs how these parameters are integrated and controlled during CD formation, as different approaches regulate precursor conversion, carbonization pathways, dopant incorporation, and surface passivation, ultimately determining the size distribution, quantum yield, and emission behavior of CDs [44]. For example, hydrothermal, solvothermal, and pyrolysis methods determine reaction pathways and nucleation, which indirectly affect the optical properties and biological performances of CDs [45-47].
Dopant incorporation, solvent selection, temperature, and surface engineering are strongly governed by selection of synthesis strategies and together determine the structural and optical properties of CDs [48]. For example, controlled incorporation of heteroatoms such as nitrogen, sulfur, or phosphorus during carbonization can modulate the electronic structures, defect states, and photoluminescence properties of CDs, enabling precise tuning of their overall properties [49-51]. On the other hand, high boiling solvents commonly used in hydrothermal or solvothermal routes promote efficient dehydration, polymerization, and carbon core formations, ultimately leading to complete carbonization and enhanced optical properties [52]. The thermal profile intrinsic to each specific synthesis method further regulates the degree of carbonization and passivation of surface groups, thereby determining particle uniformity and emission characteristics of the resulting CDs [49]. In general, the surface groups on CDs, introduced either from precursors or via post-synthesis modification, govern their hydrophilicity, stability, and biofunctionalization capability. Therefore, the choice of synthesis technique serves as a central framework to tailor CDs for biomedical applications [33]. Because numerous comprehensive reviews cover each synthesis approach in depth [33, 35, 40, 53], detailed synthesis approaches are not discussed here.
2.2. Characterization techniques of CDs
To evaluate the physical and chemical properties of CDs, various types of characterization techniques have been employed, including spectroscopic and microscopic methods [54]. Prior to characterization, CDs must be purified via centrifugation, filtration, and dialysis to remove large aggregates, residual precursors, and small molecular fluorophores. The optical properties of CDs are analyzed using the clarified supernatant in quartz cuvettes to minimize scattering artifacts. Fluorescence properties of CDs are acquired from their excitation-emission spectra and three-dimensional photoluminescence mapping. Quantum yield of CDs is determined either using standards such as quinine sulphate or by absolute measurements employing a fluorescence spectrometer equipped with integrating sphere accessories [30, 55]. Transit optical properties and excited state dynamics are evaluated using time-resolved approaches, including time-correlated single-photon counting, fluorescence upconversion, and transient absorption spectroscopy, to determine carrier lifetime and relaxation pathways [56]. UV-visible absorption spectroscopy is routinely used to correlate the absorption properties of CDs with their emission behaviors.
Structural and morphological analyses of CDs are predominantly performed using high-resolution transmission electron microscopy (HR-TEM) and selected area electron diffraction, typically after depositing diluted CDs on an ultrathin grid [57]. Atomic force microscopy (AFM) is used as a complementary approach to assess height profiles and dispersion properties of CDs [21]. HR-TEM or AFM analysis generally indicates the particle size and size distribution of CDs in the range of 1-10 nm. Compositions and bonding environments are investigated using X-ray photoelectron spectroscopy (XPS), energy dispersive X-ray spectroscopy, Fourier transform infrared spectroscopy (FTIR), and Raman spectroscopy. Nuclear magnetic resonance, mass spectroscopy, and chromatographic techniques are additionally used to elucidate molecular composition and heterogeneity, particularly for CDs with molecular fluorophore-like characteristics [58, 59].
Despite extensive advanced analytical approaches, the characterizations of CDs still face challenges that require targeted improvements. One of the major challenges is the lack of standardized protocols for measuring key properties such as quantum yield, size, and lifetime, which leads to variability across studies and complicates direct comparison. Conventional methods for size distribution analysis, including TEM and AFM, often provide localized information and may not be accurate to reflect the hydrodynamic size states of CDs in biological media. Furthermore, characterization by FTIR and XPS is qualitative, which limits precise correlation between functional group density and optical features. Time-resolved spectroscopic investigations are not consistently used across studies, resulting in an incomplete understanding of charge-carrier dynamics and emission mechanisms. Future studies should focus on developing unified characterization standards, quantitative surface analysis methods, and in situ techniques to monitor dynamic structural and optical changes of CDs under physiological conditions. These efforts are essential for establishing structure-property relationships and enhancing the reproducibility required for downstream biomedical applications.
2.3. Properties of CDs
CDs are quasi-spherical nanomaterials featuring size-dependent and excitation-tunable photoluminescence, higher water solubility, excellent photostability, and abundant surface functional groups [60, 61]. Their diverse properties make them suitable for a wide range of applications, such as sensing, bioimaging, drug delivery, photocatalysis, and optoelectronic devices [62]. The structure of CDs generally consists of a carbon core and numerous functional groups on the surface, such as hydroxyl (-OH), carboxyl (-COOH), and amine (-NH2) groups [61]. CDs are predominantly characterized by the coexistence of sp2 and sp3 hybridized carbon domains, which govern their fundamental physicochemical and optical properties [63]. These intrinsic features underpin the use of CDs in biomedical fields, including bioimaging and bioanalysis. The sp2 hybridized carbon typically forms graphitic or polyaromatic domains that contribute to 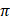 -conjugation, electronic delocalization, and light absorption, while sp3 hybridized carbon constitutes amorphous regions and defect sites that are frequently decorated with oxygen- and nitrogen-containing functional groups [64, 65]. Due to the presence of sp2 hybridized carbon, CDs are prone to π- π stacking, which can promote aggregation, quench fluorescence, and reduce stability [66]. These interactions further affect protein corona formation and cellular uptake. Therefore, surface functionalization was commonly applied to maintain physicochemical and optical properties and balance electronic functionality with aggregation control in CDs design for biomedical applications.
-conjugation, electronic delocalization, and light absorption, while sp3 hybridized carbon constitutes amorphous regions and defect sites that are frequently decorated with oxygen- and nitrogen-containing functional groups [64, 65]. Due to the presence of sp2 hybridized carbon, CDs are prone to π- π stacking, which can promote aggregation, quench fluorescence, and reduce stability [66]. These interactions further affect protein corona formation and cellular uptake. Therefore, surface functionalization was commonly applied to maintain physicochemical and optical properties and balance electronic functionality with aggregation control in CDs design for biomedical applications.
Structural studies confirmed that most reported CDs consist of sp2-hybridized carbon domains embedded within an amorphous sp3-rich carbon framework functionalized with nitrogen- and oxygen-containing groups. X-ray diffraction studies reflect a high density of structural defects and partial disorder. These defects are related to functional moieties such as hydroxyl, carboxyl, and epoxy groups, playing critical roles in defining the electronic properties of CDs. Optical absorption and emission are governed by σ/n→σ* and n/π→π* transitions, where π-states originate from aromatic sp² carbon domains and n-states from lone pairs at structural defects [67]. Variations in domain size and defect density modulate the effective energy gap, giving rise to size- and surface-dependent photoluminescence behaviors.
The optical properties of CDs are influenced by factors such as functionalization, particle aggregation, and solid concentrations [68]. Heteroatom doping, such as nitrogen, can tune the optical transitions and affect the photoluminescence kinetics and lifetime of CDs [69, 70]. The photoluminescence properties arise from a combination of quantum confinement in small sp2 domains, surface trap/defect states, and molecular fluorophore-like species [59]. Surface engineering, particularly the introduction of oxygen-, nitrogen-, sulfur-, and phosphorus-containing functional groups, is also critical for tuning the properties of CDs for specific applications in sensing and biomedicine [71, 72]. These functional groups profoundly influence the solubility, surface charge, bioconjugation capabilities, and optical behaviors of CDs [71]. Oxygen-containing functional groups, such as -COOH, -OH, carbonyl (C=O), and ether (C-O-C) groups, are commonly found on the surface of CDs [71, 72]. For instance, zirconium-doped CDs and pristine CDs exhibit prominent O-H, C=O, and C-O stretching vibrations in their infrared absorption spectra. These groups enhance the hydrophilicity of CDs, which is essential for maintaining stability and dispersibility in aqueous biological environments. Furthermore, these oxygen-containing functional groups provide reactive sites for subsequent chemical modifications, enabling the attachment of targeting ligands, therapeutic molecules, or biocompatible polymers for specific biomedical applications [71]. In addition, surface defects and functional groups on CDs can generate tunable fluorescence across various wavelengths, including long-wavelength emissions that are advantageous for deep tissue bioimaging due to reduced scattering and absorption by biological tissues. The functionalization can also enhance the photostability of CDs, ensuring consistent performance in demanding biological environments [73, 74].
Rational design and precise control over oxygen-, nitrogen-, sulfur-, and phosphorus-containing functional groups through synthesis or post-functionalization are central to optimizing CDs for diverse biomedical applications. This surface engineering allows fine-tuning of solubility, charge, bioconjugation sites, and optical properties, ultimately advancing the development of highly effective CDs for sensing, diagnostics, and targeted therapeutic delivery.
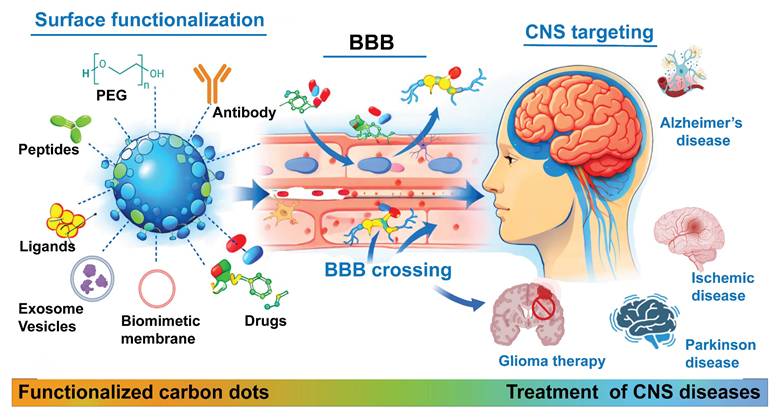
3. Design of BBB-crossing CDs for CNS targeting
The BBB poses a significant obstacle to effective drug delivery to treat CNS disorders. Although the BBB remains intact in most neurodegenerative cases, it can undergo subtle disruptions that aggravate disease progression and restrict the delivery of therapeutic agents [75]. Conversely, brain cancers frequently induce disrupted and uneven structures such as the BTB, which is more permeable than the normal BBB but heterogeneous. This irregular permeability and uneven blood flow hamper the uniform delivery of therapeutic agents [76]. Intravenously administered nanoparticles accumulate in damaged brain tissue, despite their payload mechanisms. However, this inconsistent accumulation reduces the effectiveness of nanoparticle-based therapies for CNS disorders [77, 78]. Therefore, the design of advanced drug delivery nanoplatforms capable of penetrating the BBB and targeting brain lesions is essential to effectively treat CNS disorders.
CDs are nanocarbon-based materials with excellent aqueous dispersibility, low cytotoxicity, and tailored surface properties [30, 79]. Their tunable functional groups and high surface areas enable high drug loading, and their adjustable photoluminescence allows for drug tracking [80]. Moreover, ultrasmall sizes (< 5 nm) can effectively cross the BBB between endothelial cells with 4-6 nm gaps [81, 82].
Various strategies have been adopted to effectively deliver CDs to the brain. For example, conjugation of CDs with peptides or ligands, such as angiopep-2 [83], transferrin (TfR) [84], rabies virus glycoprotein 29 (RVG29) [85], TfR-binding peptide T7, or glucose analogs [86], can engage receptors such as low-density lipoprotein receptor-related protein 1 (LRP1) [87], TfR receptor, glucose transporter 1 (GLUT1), or L-type amino acid transporter 1 (LAT1) [88] on brain endothelial cells. These functionalization strategies facilitate receptor-mediated transcytosis (RMT) for the delivery of therapeutic CDs to the brain [89]. Cationic CDs can be delivered to the brain via adsorption-mediated transcytosis (AMT) through electrostatic interactions with the negatively charged endothelial glycocalyx. However, this mechanism is less specific and increases off-target uptake [90]. In pathological contexts, such as tumors or neuroinflammation, BBB/BTB distraction allows passive accumulation but with diminished targeting selectivity. Physical interventions, such as ultrasound, can temporarily open the BBB, allowing CDs to cross more effectively. Furthermore, intranasal injections provide a BBB-bypassing route, delivering CDs to the brain through the olfactory and trigeminal nerve pathways for efficient accumulation [91]. However, intranasal delivery is influenced by mucociliary clearance and nasal residence time, as species-dependent anatomical differences between rodents and humans can affect clinical translation relevance. Brain accumulation following intranasal administration is typically assessed using fluorescence imaging, quantitative biodistributions, and histological analyses to confirm nose-to-brain transport [92, 93].
Research on CDs, specifically on BBB penetration, remains ongoing. CDs have been modified using various strategies to enhance their BBB penetration. This section outlines recent strategies for modifying CD surfaces to facilitate BBB penetration and target neurodegenerative diseases and brain cancer for effective diagnosis and treatment. Table 2 summarizes the general requirements for brain delivery, highlighting that the optimal design of CDs should be tailored to disease-specific alterations of the BBB and local pathophysiology. In neurodegenerative disorders such as Alzheimer's disease, the BBB largely exhibits reduced transporter efficiency, chronic inflammation, and impaired clearance mechanisms [94]. In this context, ultrasmall CDs (less than 5 nm) with neural or mildly negative surface charge and targeting ligands are essential for RMT, while minimizing nonspecific immune activation. Additionally, antioxidants and anti-inflammatory CD formulations are relevant for mitigating oxidative stress and microglial overactivation associated with amyloid pathology [21, 95, 96].
Summary of various modification strategies of CDs for BBB crossing in CNS disease diagnostics and theranostics
| Functionalization strategies | Size of CDs (nm) | Targeting mechanisms | Disease model | Applications | Ref. | |
|---|---|---|---|---|---|---|
| Ligand functionalization | TfR-conjugation of CDs | 2-6 | TfR-mediated endocytosis | Glioma | Imaging and drug delivery | [116] |
| Angiopep-2 decoration of CDs | 4 | LRP1-mediated transcytosis | Glioma | Glioma targeting with imaging capability | [151] | |
| Glucose-derived CDs | 3.77 | GLUT1 transporter-mediated transport | Neurodegeneration & glioma | BBB crossing and neuronal imaging | [86] | |
| Tryptophan-derived CDs | 4-7 | LAT1 transporter-mediated transport | Neurodegeneration & glioma | BBB crossing and glioblastoma cell uptake | [152] | |
| IL-6 peptide-functionalized polymer-coated CDs | 5-10 | IL-6 receptor targeting | Glioma | Glioma targeting, pH-triggered drug release, and imaging | [99] | |
| Aptamer-functionalized CDs | 5 | Aβ-targeting via aptamer and photomodulation | Alzheimer's disease | Target Aβ, red-light-induced denaturation, and plaque reduction | [153] | |
| Polymer coating strategies | Ginkgolide B-loaded carbonized CDs | 5 | Delivery through a polymer matrix | Ischemic stroke | Decrease infarct size, reduce neuronal apoptosis and inflammation | [154] |
| Metformin CDs | 2.95 | Passive diffusion | Alzheimer's disease | Promote neurogenesis, reduce Aβ deposition, and provide neuroprotection | [155] | |
| PEG- Lf Coated CDs | 4.5 | Lf receptor-mediated transportation | Parkinson's disease | Nitric oxide-releasing CDs reduce ROS, chelate Fe²⁺, and suppress α-synuclein aggregation | [103] | |
| Polydopamine-coated CDs in macrophage-derived exosomes | 3.15 | Exosome-mediated delivery | Parkinson's disease | Reduce oxidative stress, alleviate symptoms through anti-inflammatory/photothermal therapy | [156] | |
| Triple-functionalized HSA-conjugated CDs (TfR + RVG29 + Angiopep-2) | 2-4 | RMT via three ligands | Alzheimer's disease | Detect Aβ aggregates, prevent aggregation, scavenge ROS, enable in vivo imaging | [23] | |
| Biomimetic membrane/ exosome coating | Macrophage-exosome-coated CDs | 2 | Exosome-mediated homing | Glioma | Precise boron delivery for BNCT, in vivo survival | [106] |
| Exosome-coated polydopamine CDs | 2-4 | Biomimetic membrane homing and receptor-mediated uptake | Parkinson's disease | BBB crossing, target neuroinflammation to restore injured neurons by eliminating oxidative stress | [107] | |
| Microglia membrane-wrapped caffeic acid-CDs | 5 | Biomimetic homing and nasal route to bypass the BBB | Alzheimer's disease | Microglial membrane enhances brain targeting, reduces neuroinflammation, and improves cognition | [133] | |
| Other modification strategies | Vitamin C-derived CDs | 4 | Passive diffusion | Alzheimer's disease | Inhibit Aβ aggregation, ROS scavenging | [129] |
| Aspartic acid-derived CDs | 2.28 | Self-targeting via glioma affinity | Brain cancer | Intrinsic glioma targeting and imaging | [108] | |
| Quercetin-derived red-emission CDs (R-CD-75) | 3.66 | Passive diffusion, hydrophobic/π-π/H-bond interactions | Alzheimer's disease | Inhibit Aβ aggregation, red fluorescence imaging | [157] | |
| Memantine-loaded CDs | 2.2 | Passive diffusion | Alzheimer's (tau aggregation) | Deliver memantine and inhibit tau aggregation | [118] | |
| Crinis Carbonisatus-derived CDs | 3.2-8.8 | Passive diffusion | Ischemic stroke (cerebral I/R injury) | Reduce infarct volume, inflammation, and neuroprotection | [158] | |
| Fucoidan-derived CDs with sulfate groups | 1.83 | Negative-charge mediated transcytosis | Parkinson's disease | Antioxidant/anti-inflammatory effects | [21] | |
| CDs with amphiphilic surface | 3 | Passive diffusion | Alzheimer's disease | Inhibit APP & Aβ overexpression and allow BBB penetration | [95] | |
| Endogenous nutrient-based CDs | 6.21 | Passive diffusion | Glioma | Visible/NIR imaging and drug delivery | [142] | |
TfR: Transferrin, Aβ: β-amyloid, APP: Amyloid precursor protein, BNCT: Boron neutron capture therapy, GLUT1: Glucose transporter 1, IL-6: Interleukin-6, LAT1: L-type amino acid transporter 1, LRP1: Low-density lipoprotein receptor-related protein 1, RVG29: Rabies virus glycoprotein 29; PEG-Lf: polyethylene glycol Lactoferrin
In contrast, brain glioma presents highly heterogenous BBB regions that are partially disrupted, with abnormal vasculature and an immunosuppressive tumor microenvironment. For glioma treatment, CDs can be designed with targeting ligands such as epidermal growth factor receptor (EGFR) and TfR, as well as stimuli-responsive triggered drug release mechanism, to exploit the acidic and oxidative tumor milieu [97].
In ischemic stroke, BBB disruption is acute and transient, accompanied by elevated levels of reactive oxygen species (ROS) and inflammatory cytokines and increased vascular permeability. CDs formulated for treating this condition prioritize rapid BBB crossing, ROS scavenging capacity, and short systemic retention to reduce secondary injury. More specifically, biodegradable CDs with strong ROS scavenging and anti-inflammatory activity are suited for this setting, supporting neuroprotection and functional recovery while ensuring efficient renal clearance [98].
Overall, disease-specific considerations underscore that the design of BBB-penetrating CDs should not follow a universally applicable approach but should be tailored to the dynamic and pathological features of each neurological condition. Explicitly aligning CD physicochemical properties with disease-specific BBB alterations is essential for improving therapeutic efficiency and accelerating clinical translation.
3.1. Surface modification with a targeting ligand
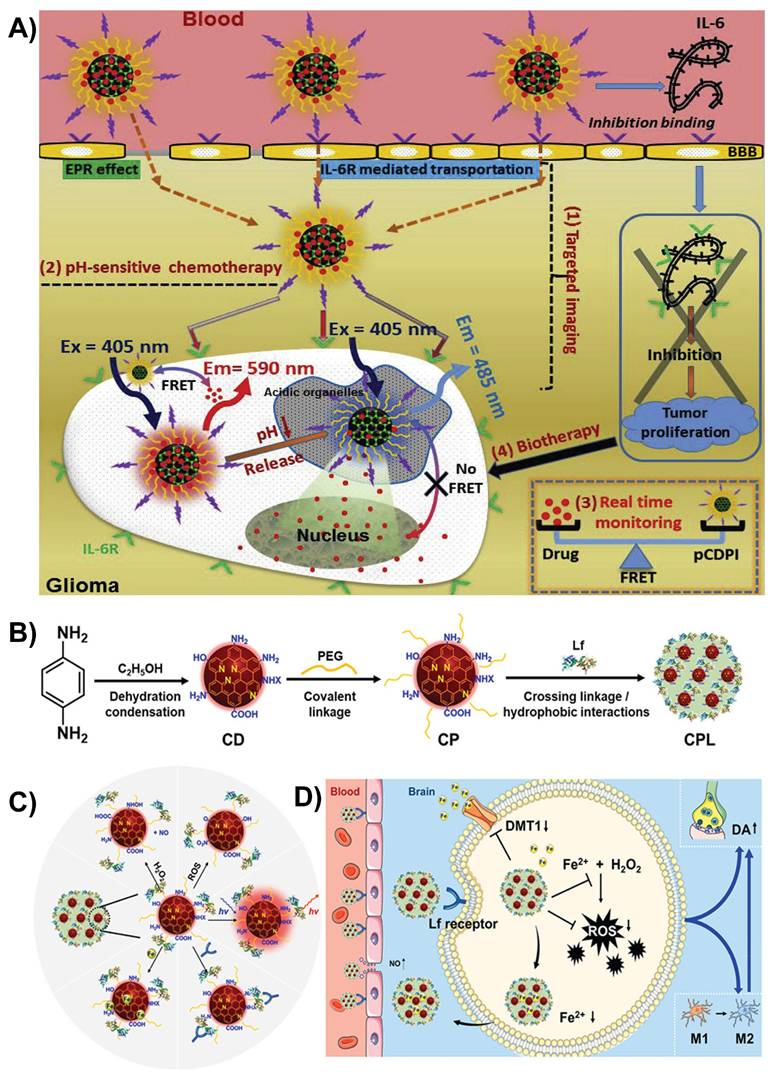
By modifying CDs with peptides or ligands, their ability to connect with receptors on endothelial cells can be enhanced. The addition of functional groups, such as -COOH, -OH, -C=O, or imine (-C=N), facilitates their passage across the BBB through receptor-mediated transport, allowing for better targeting of disease-related lesions [81, 90]. Wang et al. developed CDs linked with interleukin-6 (IL-6) peptide fragments and doxorubicin (DOX) to target IL-6 receptors for targeted drug delivery. This dual-targeting approach helps the CDs cross the BBB efficiently via RMT and reach deep into the glioma tissues. These smart CDs remained stable at the normal body pH but released DOX in the acidic tumor environment, making drug delivery more precise (Figure 3A). The fluorescence of CDs also allows real-time tracking of the drug location. In animal studies, these CDs could effectively cross the BBB, target gliomas, and release drugs in a controlled manner, paving the way for potential imaging-guided brain cancer treatments [99].
A) Schematic illustration of the in vivo delivery of multifunctional CDs for targeted imaging, pH-responsive chemotherapy, real-time monitoring, and biotherapy. Reproduced with permission [99] Copyright 2017, Elsevier. B) Schematic illustration of the synthesis of different nanoformulations. C) Multiple functions of Lf-modified CDs. D) Illustration of iron ion-sequestering and antioxidative CD-based nano formulation with NO release for Parkinson's disease treatment. Reproduced with permission from [103], copyright 2024, Elsevier.
Recent advances have shown that CDs modified with ligands/peptides demonstrate substantially improved ability to penetrate the BBB via RMT. For example, TfR-conjugated CDs (TfR-CDs) selectively bind to TfR receptors on endothelial cells, allowing uptake in zebrafish models, whereas unmodified CDs do not cross the BBB [84]. Similarly, glucose-derived CDs (GluCDs) with inherent -OH and -C=O functional groups use GLUT1-mediated transport to penetrate the BBB in zebrafish and rat models, allowing the delivery of fluorescent payloads without additional ligand conjugation [86]. Recently, Chang et al. developed curcumin-derived CDs (Cur-CDs) using an eco-friendly one-step dry-heating method to enhance the delivery of neuroprotective agents across the BBB for Alzheimer's disease treatment. By retaining curcumin's phenolic -OH and β-diketone groups, Cur-CDs offer high solubility, biocompatibility, and improved BBB permeability due to their small size and functionalized surfaces. In SH-SY5Y cells, these CDs were nontoxic and effectively inhibited Aβ fibrillization and tau hyperphosphorylation, which are key pathologies of Alzheimer's disease. This study highlights surface-functionalized CDs derived from natural compounds as a scalable platform for crossing the BBB and targeting multiple pathways involved in Alzheimer's disease [100]. Furthermore, this work exemplifies that rational selection of precursors and synthesis approaches can transform CDs from fluorescent materials to disease-specific biological platforms. Incorporating such disease-informed synthesis strategies will be essential for bridging the gap between laboratory-scale CD development and meaningful clinical translation.
3.2. Polymer coating strategy
Coating CDs with biocompatible polymers, such as polyethylene glycol (PEG) or chitosan, can make them more stable, less toxic, and better at crossing the BBB through both active and passive mechanisms [101]. For example, CDs coated with chitosan or albumin can undergo AMT to cross the BBB. However, efflux pumps in the brain may reduce the retention time of these CDs in the brain [102].
Huang et al. designed red-emissive CDs by modifying them with PEG-lactoferrin (PEG-Lf) to improve brain delivery of the drug to treat Parkinson's disease. This modification allows CDs to cross the BBB in two ways: by temporarily opening the barrier with nitric oxide (NO) and via lactoferrin RMT (Figure 3B and C). Once in the brain, CDs exert multifaceted neuroprotective effects by neutralizing harmful ROS owing to their antioxidant properties, release NO to reduce neuroinflammation, and bind excess iron to prevent oxidative stress via the Fenton-like reaction. These PEG-Lf-modified CDs also supported real-time imaging and showed promise in mouse models of Parkinson's disease by improving motor deficits and reducing neurodegeneration (Figure 3D). The PEG-Lf coating modulated the surface charge of the CDs to be more neutral, which minimized unwanted interactions, boosted the uptake by lactoferrin receptors, and lowered toxicity [103].
Apart from promoting BBB penetration, this strategy improves the circulation time, stability, biocompatibility, and targeting capability of CDs to treat CNS diseases. The most common polymers used to coat CDs for crossing the BBB are PEG, chitosan, polyethyleneimine (PEI), polysorbate 80 (Tween 80), and poly(lactic-co-glycolic acid). These coatings enhance hydrophilicity, help evade immune clearance, provide mucoadhesion, promote AMT and RMT, and enable controlled drug release.
3.3. Modification with biomimetic membranes and exosomes
CDs can be enveloped in membranes sourced from cells (such as macrophages or mesenchymal stem cells) to replicate biological structures, improving their capacity to penetrate the BBB via cell-mediated transcytosis or immune cell infiltration [104]. Xie et al. developed a biomimetic nanoplatform for the targeted treatment of Alzheimer's disease. Nitrogen-doped CDs (N-CDs) are enclosed within macrophage cell membranes, allowing them to cross the BBB and specifically target Aβ plaques. The N-CDs perform dual roles in regulating metal ion homeostasis to disrupt toxic metal Aβ interactions and delivering photothermal therapy via near infrared (NIR) irradiation to disaggregate Aβ plaques. This approach enhances therapeutic precision, reduces off-target effects, and shows promise in targeting Aβ plaques and modulating the Alzheimer's disease microenvironment [105]. While the macrophage membrane camouflage provides targeting and BBB crossing, potential immunogenicity, long-term biosafety, and large-scale production of the nanoplatform remain to be established in future preclinical studies.
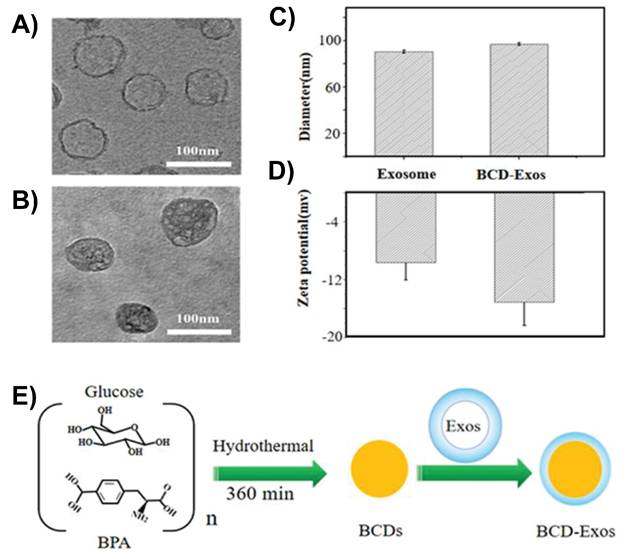
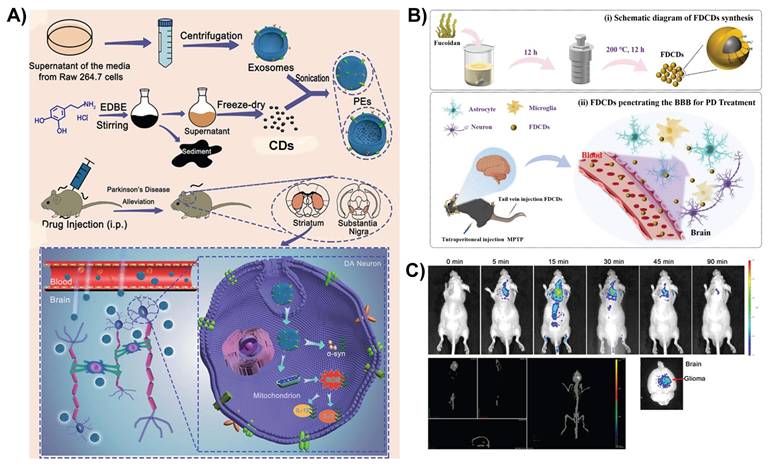
Exosomes are natural nanocarriers with low immunogenicity and inherent BBB-crossing capabilities. CDs can be incorporated into exosomes to deliver drugs and imaging agents to the brain. Boron-enriched CDs (10BCDs) were encapsulated within macrophage-derived exosomes to create a biomimetic nanoplatform (BCD-exosome) for targeted glioma therapy using boron neutron capture therapy (BNCT). Exosome coating considerably enhanced the ability of CDs to cross the BBB by leveraging the natural targeting and transcytosis mechanisms of macrophage vesicles. As shown in Figure 4A-D, TEM and dynamic light scattering analysis confirmed successful encapsulation of BCDs in exosomes, yielding a size of approximately 100 nm. Upon neutron irradiation, BCD-exosomes (Figure 4E) delivered a localized dose (~8.4 Gy), resulting in complete tumor ablation and 100% survival of the treated mice [106].
TEM images of A) Exosomes and B) BCD-Exosomes. C) Hydrodynamic size distributions and D) zeta potentials of BCDs and BCD-Exosomes in PBS. E) BCD-Exosome preparation. Reproduced with permission from [106], copyright 2021, Wiley-VCH.
Similarly, Yang et al. reported macrophage-derived exosome-coated polydopamine-CDs (PCDs) of 2-4 nm in size for formulating a biomimetic nanoplatform (PCDs@Exosomes) for Parkinson's disease therapy. Exosomes facilitate the BBB-crossing ability of CDs by exploiting the uptake mechanisms of macrophage extracellular vesicles and their homing nature to target inflamed neural tissues (Figure 5A). After crossing the BBB, PCDs effectively mitigate both motor and non-motor symptoms in Parkinson's disease mouse models by exerting antioxidant and anti-inflammatory effects, decreasing α-synuclein aggregation, and promoting neuronal viability in areas such as the substantia nigra, striatum, and prefrontal cortex [107]. In general, modifying CDs with biomimetic membranes or exosomes can enhance BBB penetration by utilizing cell-mediated transport for targeted homing. These modifications also promote immune evasion, prolong circulation time, and improve biocompatibility.
A) Synthesis and modification of PCDs@Exosomes, along with the proposed mechanism for treating Parkinson's disease. Reproduced with permission. [107] Copyright 2025, Elsevier. B) Schematic illustration of fucoidan-derived CDs crossing the BBB for Parkinson's disease treatment. Reproduced with permission from [21], copyright 2025, Elsevier. C) In vivo and ex vivo imaging of glioma-bearing mice following tail vein injection of CD-Asp; whole-body distribution over time, 3D reconstruction of brain localization at 20 min post-injection, and ex vivo brain imaging at 90 min post-injection. Reproduced with permission from [108], copyright 2015, American Chemical Society.
3.4. Other modification strategies for CDs
In addition to surface modification in CDs, other strategies have been developed to improve the capacity of CD-based nanomedicines to traverse the BBB and treat neurodegenerative disorders. Fucoidan-derived CDs penetrate the BBB via various complementary pathways. Due to their extremely small size (less than 4-6 nm), they can passively diffuse through nanometer-sized gaps between endothelial cells via paracellular transport. This mechanism works well when the BBB is compromised, which is frequently observed in neurodegenerative conditions, such as Parkinson's disease, where inflammation and oxidative stress increase BBB permeability (Figure 5B). Furthermore, the negative charge on the CD surface, resulting from the sulfate groups of fucoidan, induces electrostatic interactions with positively charged sites on the BBB or carrier proteins, thus promoting AMT and improving penetration. The combination of their ultrasmall size, surface charge, and altered state of BTB supports the effective delivery of fucoidan-derived CDs into the brain, establishing them as promising options for the targeted treatment of Parkinson's disease and other CNS disorders [21]. Zhang et al. found that CDs made from D-glucose and L-aspartic acid (CD-Asp) can cross the BBB and accumulate in glioma tissues without external targeting agents. The small size of these CDs allows them to passively cross the BBB. Their surface chemistry, which includes -NH2 and -COOH groups from L-aspartic acid, may increase glioma cell uptake. In vivo fluorescence imaging demonstrated that 15 min after intravenous injection, CD-Asp showed a substantially greater signal in glioma than in normal brain regions (Figure 5C). Control CDs containing D-glucose or L-glutamic acid alone showed negligible brain targeting, underlining the unique self-targeting capabilities of CD-Asp for noninvasive glioma diagnosis [108].
A targeted theranostic platform for Alzheimer's disease was developed by conjugating CDs to triple-functionalized human serum albumin (HSA) to create a multifunctional nanocomposite. Three ligands known to promote transport across the BBB—angiopep-2 (a BBB-penetrating ligand), neurotropic peptide (RVG29), and TfR (a TfR receptor ligand)—were added to the HSA. These surface changes facilitate RMT and allow CDs to pass through the BBB and accumulate in the brain. After entering, the CDs offer fluorescent imaging capabilities and collaborate with HSA to scavenge ROS, bind metal ions, and prevent Aβ aggregation. This multitarget strategy is suitable for simultaneous diagnosis and treatment of Alzheimer's disease [23].
3.5. Mechanistic insights into CD functionalization strategies for disease-specific CNS pathology
Table 2 summarizes representative functionalization strategies of CDs and their relative efficiency, selectivity, and translational limitations. However, more explicit comparisons of existing strategies are warranted in the context of disease-specific pathology. Ligand-mediated functionalization strategies offer higher targeting specificity via RMT in multiple preclinical models. However, this method is constrained by receptor saturation, competitive binding with endogenous ligands, and disease-dependent variability in receptor expression in neurodegenerative conditions [99]. These limitations highlight the need to align ligand selection and density with the pathological state of the BBB. In contrast, polymer-based surface modification could enhance circulation stability and reduce nonspecific protein adsorption rather than conferring active targeting. Despite their improved pharmacokinetics and reduced immune clearance, the BBB-crossing efficacy of polymer-coated CDs is generally lower than that of ligand-functionalized CDs and relies largely on passive diffusion or nonspecific endocytosis. Moreover, excessive polymer shielding may hinder cellular uptake and intercellular trafficking [81]. Such approaches may therefore be suitable for conditions involving transient BBB disruption, such as ischemic stroke, rather than diseases with intact BBB.
Biomimetic functionalization approaches provide a multifactorial mechanism for BBB penetration by combining immune evasion, prolonged circulation, and endogenous receptor engagement. These approaches display superior biological mimicry and enhanced accumulation in brain tissue [107]. However, they face significant challenges related to batch-to-batch variation, complex preparation, and limited scalability, which hinder their clinical translation [109].
Importantly, the effectiveness of the above functionalization strategies is closely related to upstream synthesis preference, including precursors and reaction parameters, which can determine the overall physicochemical properties of CDs. The lack of connection among synthesis approaches, functionalization mechanisms, and disease-specific pathology could be a major bottleneck for the clinical translation of CDs. No single functionalization strategy is universally optimal. Future CD design should prioritize hybrid, or modular functionalization approaches that balance targeting precision, biological stability, and manufacturability, while being guided by the pathological features of specific CNS diseases.
4. Diagnosis and treatment of brain diseases using BBB-penetrating CDs
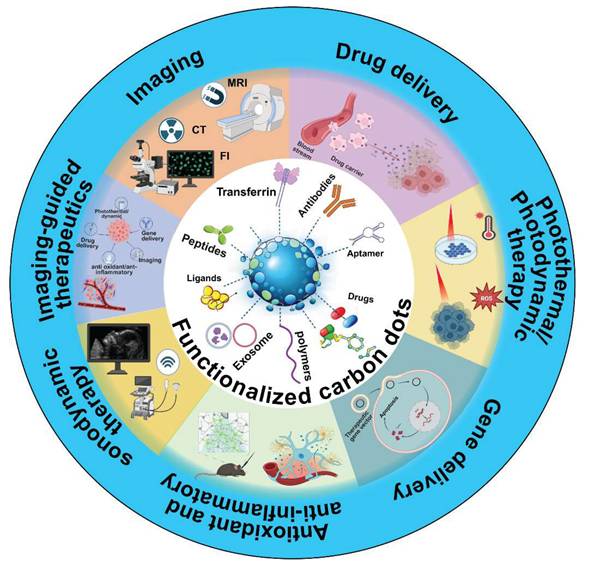
Building upon the above CD design strategies to enable BBB penetration, various nanomedicine formulations have been developed for imaging, treatment, and theranostic management of brain disorders. In this section, we summarize the key advancements in CD-based nanomedicines, focusing on their applications in imaging, drug delivery, gene delivery, photothermal therapy, photodynamic therapy, sonodynamic therapy, antioxidant and anti-inflammatory treatment, and theranostic approaches for CNS diseases (Figure 6). A comprehensive overview of the mechanism of action of CDs in different treatments and diagnostic strategies for neurodegenerative diseases and brain cancer is presented in Table 3. Although some of these applications were briefly mentioned in the previous sections, a more detailed examination of their roles and potential in the management of CNS diseases is presented below.
Schematic overview of functionalized CDs capable of crossing the BBB for theranostic applications in CNS diseases. Created with BioRender.com.
Summary of the main CNS diseases, their brain pathologies, their symptoms, and mechanisms of CDs in treatment
| Representative CNS Disease | Targeted brain elements | Pathological roles | Mechanism of treatment with CDs |
|---|---|---|---|
| Alzheimer's disease | Microglia, Aβ receptors (RAGE, TREM2), Synapses [159] | Neuroinflammation via microglial activation; Aβ plaque accumulation | Inhibit Aβ aggregation, reduce ROS, conjugate with drugs (e.g., memantine) for targeted delivery, and fluorescent imaging of Aβ plaques for early diagnosis [160, 161]. |
| Parkinson's disease | Dopamine receptors (D1/D2), Microglia [162] | Dopaminergic neuron degeneration, neuroinflammation | Scavenge ROS, promote neurodegeneration, deliver neuroprotective agents (e.g., dopamine precursors), fluorescent tracking of drug distribution, and imaging Lewy bodies [163]. |
| Brain tumor (e.g., Glioblastoma) | TME, EGFR, BBB [164] | Tumor growth, BBB disruption, resistance to therapy | Deliver chemotherapeutic drugs, induce photodynamic therapy via ROS generation, target tumor cells with conjugated drugs (e.g., doxorubicin), and enable fluorescent imaging for tumor localization and treatment monitoring [87, 165]. |
| Traumatic brain injury | Tight junctions (occludin, claudins), ROS, Microglia [166] | BBB disruption, oxidative damage, neuroinflammation | Scavenge ROS, reduce inflammation, promote neuronal repair, deliver anti-inflammatory or neuroprotective agents (e.g., minocycline), and use fluorescent imaging to monitor injury sites and enhance treatment efficacy [167, 168]. |
| Ischemic stroke | Glutamate receptors (NMDA), ROS, Endothelial cells [169] | Excitotoxicity, BBB breakdown, oxidative stress | Scavenge ROS, mitigate excitotoxicity, reduce inflammation, deliver thrombolytic or neuroprotective drugs (e.g., tissue plasminogen activator), and image ischemic areas for real-time monitoring of tissue damage and drug distribution [158, 170]. |
| Depression | Serotonin receptors (5-HT1A/2A), BDNF [171] | Neurotransmitter imbalance, neuronal atrophy | Modulate oxidative stress, enhance neurogenesis, reduce inflammation, deliver antidepressants (e.g., SSRIs like fluoxetine) to specific brain regions, imaging to study neurotransmitter pathways or monitor treatment response [130, 172]. |
BDNF: brain-derived neurotrophic factor, RAGE: receptor for advanced glycation end products, TREM2: triggering receptor expressed on myeloid cells 2, EGFR: epidermal growth factor receptor, ROS: reactive oxygen species, TME: tumor microenvironment, 5-HTA1A/2A: 5-hydroxytryptamine (serotonin) receptors 1A and 2A, NMDA: N-methyl-D-aspartate receptor.
4.1. Imaging
CDs are nontoxic and display excellent photoluminescence and transverse BBB permeability, making them promising candidates for noninvasive imaging of the brain. Surface functionalization, doping, and biomimetic coating can enhance their imaging performance [110]. Manganese-doped CDs (Mn-CDs) were synthesized via a one-pot microwave-assisted method, resulting in ultrasmall particles exhibiting excitation-dependent fluorescence and exceptionally high T₁ magnetic resonance imaging (MRI) relaxivity. Following systemic administration in a mouse glioma model, Mn-CDs enabled in vivo T₁-weighted MRI to distinctly enhance the contrast within the glioma region and reveal tumor margins. Ex vivo fluorescence imaging of resected brain tissue confirmed the selective accumulation of Mn-CDs in brain tumors, as evidenced by localized bright photoluminescence. Owing to their low cytotoxicity, strong MRI contrast, and effective optical imaging capabilities, Mn-CDs have strong potential as dual-modal nanoprobes for the precise detection and intraoperative localization of small brain gliomas [111]. Gadolinium-ytterbium co-doped CDs (Yb/Gd-CDs) were created via a one-pot hydrothermal method with an ultrasmall size, excellent colloidal stability, strong fluorescence, and robust computed tomography (CT) and MRI contrast. In vivo imaging in mice demonstrated bright fluorescence signals (Figure 7A and B), clear CT visualization of tissues at high X-ray attenuation (~40 HU/g/L), and T₁-weighted MRI with a T₁ relaxivity of ~11.16 mM⁻¹ s⁻¹. This multimodal platform supports precise anatomical localization (MRI/CT) along with sensitive optical tracking, which is ideal for detecting brain tumors, such as gliomas [57]. In addition, Gd-doped CDs (~2-8 nm) derived from starch showed high MRI relaxivity and stable red emission.
A) Confocal fluorescent microscopic images of 4T1 cells incubated with PBS and Yb/Gd-CDs. B) In vivo fluorescence images nude mice and major organs (heart, liver, spleen, lungs, and kidneys) after intravenous injection of PBS and Yb/Gd-CDs (excitation: 430 nm; emission: 500 nm). Reproduced with permission from [57], copyright 2024, Elsevier. C) Schematic diagram of the preparation of NCD coupled with clinical Gd-DTPA and its major biological effects. D) T1-MR images of a representative nude mouse pre-injected with NCDDG (left panel); T1-MR signal-to-background ratio for tumor margin and center (upper right); fluorescence images of the mouse at various time points (middle right); fluorescence images of major organs (brain, heart, liver, spleen, lung, and kidney) and two cross-sections of the brain. Reproduced with permission from [113], copyright 2018, Elsevier.
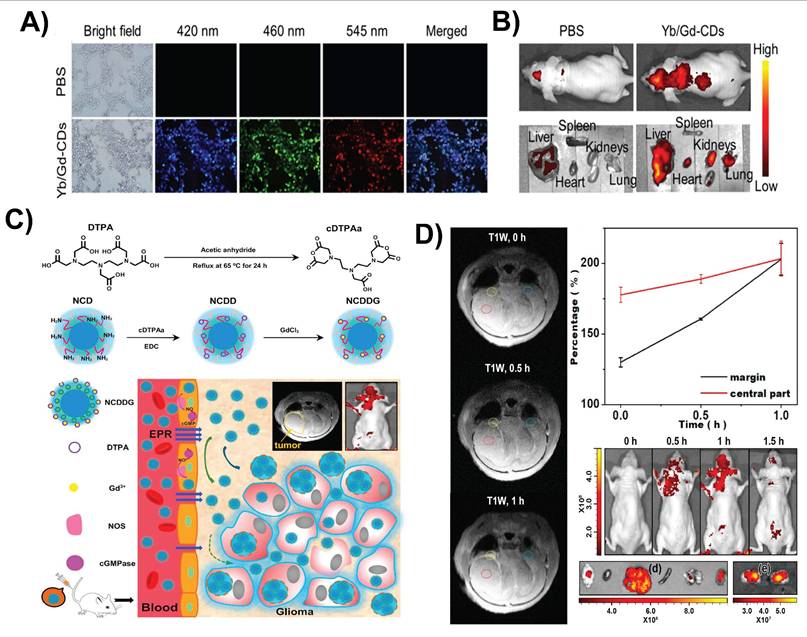
In cellular models, they are localized within the cytoplasm and produce robust contrast enhancement, highlighting their potential for brain mapping and imaging gliomas. These CDs also exhibit bright and stable fluorescence, ultrasmall size (<10 nm), and optimized surface characteristics, enabling them to cross the BBB and selectively target brain tumors. Their high MRI relaxivity enhances the imaging contrast, whereas real-time optical imaging provides dynamic visualization. These features support multimodal imaging capabilities (fluorescence imaging + MRI + CT), improve detection accuracy, and allow cross-validation across different imaging modalities [112]. Similarly, polymer-coated N-CDs covalently linked with clinical-grade Gd-DTPA formed a dual-modal nanoprobe for MRI and fluorescence imaging of gliomas (Figure 7C). The polymer shell enhances biocompatibility, whereas its ultrasmall size (~5-10 nm) facilitates BBB penetration and tumor targeting via the enhanced permeability and retention effect. Compared with free Gd-DTPA, Gd-CDs exhibit higher T₁ relaxivity and stronger fluorescence. As shown in Figure 7D, glioma-bearing mice displayed a clear, time-dependent MRI signal enhancement, with fluorescence imaging of brain sections confirming precise tumor localization [113]. This platform offers high-resolution, biocompatible glioma imaging with strong MRI and optical contrast.
4.2. Drug delivery
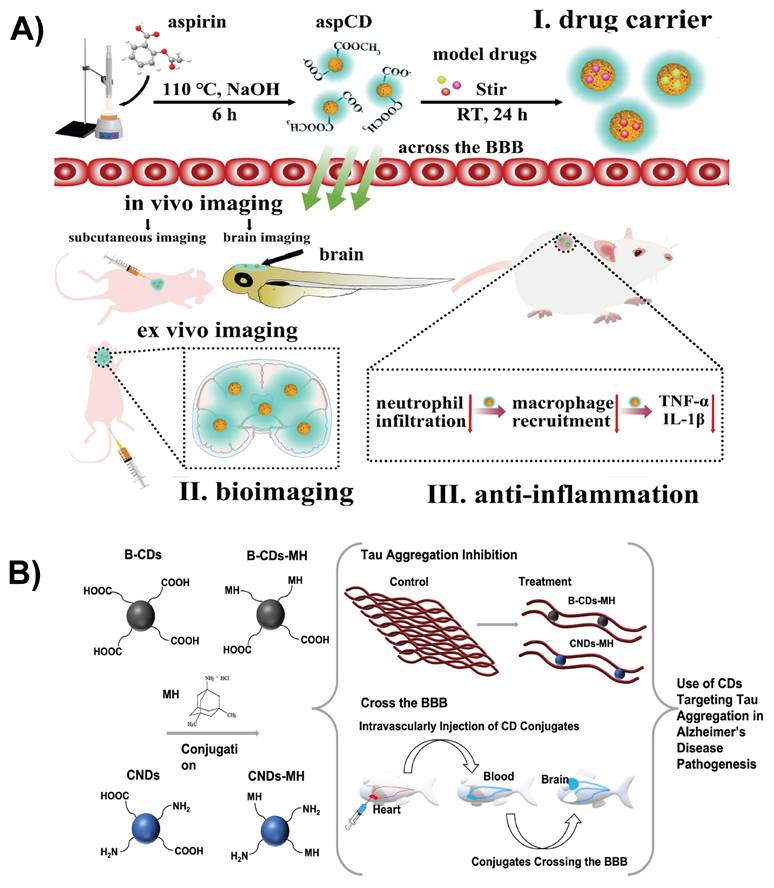
Drug delivery systems are designed to transport drugs to specific sites in the body for effective drug interaction. Nanostructured materials improve drug delivery by enhancing the absorption, distribution, and elimination of drugs. Nanotechnology enables targeted delivery, improved solubility of poorly water-soluble drugs, co-delivery of multiple therapies, transport of large macromolecules, and real-time monitoring using imaging agents [114]. Triple-conjugated CDs functionalized with TfR and loaded with two chemotherapeutic drugs, epirubicin and temozolomide, were designed for targeted brain delivery. The TfR on CDs facilitates RMT across the BBB, enhancing their accumulation in glioblastoma cells. This dual-drug delivery system induces synergistic chemotherapeutic effects at lower doses, improving tumor inhibition while minimizing the off-target toxicity. The small size and surface modifications of CDs promote cellular uptake and controlled drug release within the tumor microenvironment, making them promising nanocarriers to effectively treat brain cancer [115]. TfR is highly expressed in the BBB and in many brain tumors; therefore, many nanoplatforms engage TfR-mediated endocytosis for cellular uptake. Li et al. formulated a nanocarrier in which CDs are covalently conjugated with the targeting ligand TfR and the drug DOX for brain tumor treatment. The conjugated CDs showed a more significant uptake in pediatric brain tumor cell lines compared to the free DOX because the TfR moiety facilitated receptor recognition and internalization [116]. In another study, the small size and surface chemical properties of CDs promoted BBB penetration in both zebrafish and mouse models. CDs fabricated by hydrothermal carbonizations of aspirin exhibited a stable photoluminescence for imaging while maintaining good biosafety both in vitro and in vivo (Figure 8A). In addition to imaging, it functioned as an anti-inflammatory agent with the ability to load and deliver drugs of varying polarities [117]. Zhang et al. employed CDs as a carrier to deliver the Alzheimer's drug memantine across the BBB in vivo. They synthesized carbon nitride CDs and black CDs functionalized with memantine using N-ethyl-N′-(3-dimethylaminopropyl)carbodiimide/N-hydroxysuccinimide amidation (Figure 8B). Due to their diverse surface functionalities and small size, these conjugates facilitated efficient transport across the BBB in the zebrafish model. After penetrating the BBB, CDs not only delivered memantine but also inhibited tau protein aggregation. These findings indicate that CDs serve as nanocarriers for drug delivery across the BBB and as functional inhibitors of disease-related biomolecular processes in the brain [118].
A) Schematic representative of the synthesis and application of aspirin-derived CDs and their applications in drug loading, bioimaging, and anti-inflammation. Reproduced with permission from [117], copyright 2021, Elsevier. B) Schematic representation of CDs conjugated with memantine and their applications in delivering memantine to treat Alzheimer's disease. Reproduced with permission from [118], copyright 2022, Elsevier.
4.3. Gene delivery
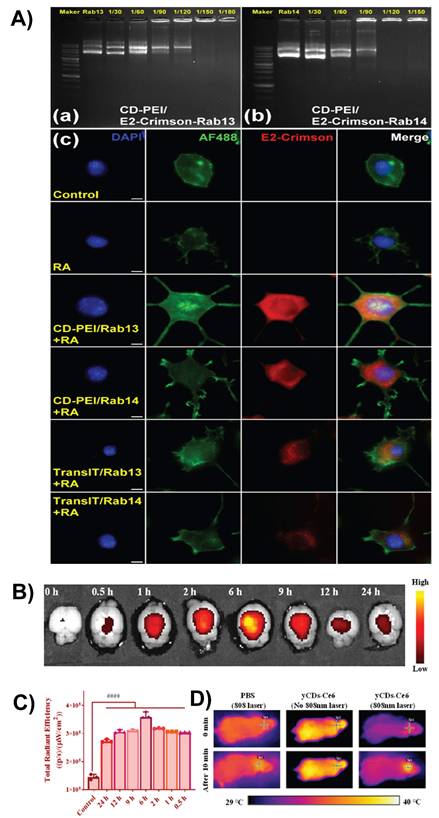
Gene delivery requires transporting genetic material (such as DNA, siRNA, or mRNA) across the BBB. This strategy aims to correct defective genes, silence harmful genes, and promote the expression of therapeutic proteins. It holds promise for treating conditions such as gliomas, Parkinson's disease, and Alzheimer's disease [17, 102, 119]. CDs offer a safe and effective nonviral platform for gene delivery. Functionalized CDs, especially those with amines or glucose, can bind to genetic materials and cross the BBB with minimal toxicity. Their small size, tunable surface, and intrinsic fluorescence also allow tracking of delivery and therapeutic effects [86]. Zhang et al. prepared ultrasmall, fluorescent, and positively charged pentaethylenehexamine-modified CDs (PCDs) using a facile microwave method and noncovalently bound them to plasmid DNA at a 2:1 mass ratio, achieving high transfection efficiency and notably lower cytotoxicity than conventional polyethylenimine (PEI)-based vectors. Crucially, these PCDs crossed the BBB in zebrafish models, demonstrating potential for non-viral gene delivery and bioimaging in CNS applications, such as neurodegenerative disease treatment and glioma therapy [119]. Researchers synthesized CDs using nitric acid reflux and subsequently coated them with PEI. The modified CDs possess a positive charge and strong fluorescence and serve as both imaging probes and gene-delivery vehicles. Gel retardation analysis suggests that these CDs successfully bind to Rab13-Q67L and Rab14 plasmids (Figure 9A) and are readily internalized by neurons. Fluorescence images of N2a cells demonstrate cellular uptake and strong expression of Rab13-Q67L and Rab14 (E2-crimson, red), with minimal background in controls (Figure 9A). These results are consistent with the reported high transfection efficiency of up to 97%, while exhibiting minimal cytotoxicity. Following delivery, CDs significantly enhanced the expression of Rab13 and Rab14, leading to notable neurite outgrowth, with over 56% of neurons displaying neurites longer than twice the diameter of their cell bodies. This demonstrates the potent capacity of CDs to promote neurite sprouting and elongation. This study presents a highly effective fluorescent nanocarrier system for non-viral gene delivery to the nervous system [120].
A) Agarose gel analysis of CD-PEI/plasmid complexes for (a) E2-Crimson-Rab13-Q67L and (b) E2-Crimson-Rab14 at DNA:CD-PEI ratios of 1/30-1/180. (c) Fluorescence images of N2a cells after 4 days showing expression of Rab13-Q67L or Rab14 delivered by TransIT (positive control) or CD-PEI. E2-Crimson (red), nuclei (DAPI, blue), and F-actin (Alexa Fluor 488, green). Scale bar: 10 μm. Reproduced with permission. [120] Copyright 2023, American Chemical Society. B) Fluorescence signals observed in the brain tissues of mice 24 h after tail vein injection of yCDs-Ce6 and C) Corresponding fluorescence signals of yCDs-Ce6. D) Thermal graphs of mice brain tissues treated with 808 nm laser irradiation for 10 min after intravenous injection of PBS and yCDs-Ce6. Reproduced with permission from [124], copyright 2022, American Chemical Society.
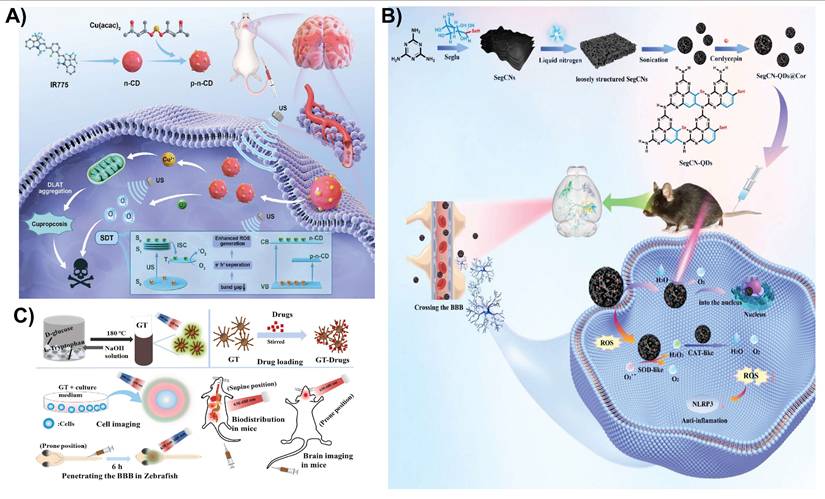
4.4. Photothermal and photodynamic therapy
Photothermal and photodynamic therapies are light-activated treatment strategies that can be enhanced using CDs for brain diseases, such as glioma. In photothermal therapy, CDs with strong NIR absorption convert light into localized heat, inducing hyperthermia and targeted tumor cell death [121]. In photodynamic therapy, CDs act as or carry photosensitizers (e.g., Ce6 or porphyrin), which, upon red or NIR light activation, trigger apoptosis [122]. CDs can effectively cross the BBB, accumulate in brain lesions, and offer dual functionality for both therapy and fluorescence imaging, enabling precise image-guided treatment with minimal damage to the surrounding healthy tissue. Recently, Fan et al. developed CDs with dual photodynamic and photothermal therapy capabilities for brain applications. These CDs are engineered to cross the BBB, allowing targeted treatment of Aβ plaques associated with Alzheimer's disease. Under laser irradiation, the CDs generate ROS for photodynamic therapy-mediated oxidative degradation of Aβ plaques, while also producing localized heat for photothermal therapy, enhancing plaque disruption and clearance. Their multifunctional properties, which combine BBB permeability, ROS generation, and thermal effects, make them promising noninvasive therapeutic tools for targeting neurodegenerative pathologies through combined photothermal/photodynamic therapy mechanisms [123]. Yan et al. reported a multifunctional nanoplatform composed of CDs and a photosensitizer (Ce6), forming nanoassemblies (CD-Ce6) with anti-amyloid, antimicrobial, and BBB-penetrating properties. Fluorescence imaging and brain tissue section analysis in mice demonstrated that CD-Ce6 exhibited strong accumulation in the brain regions after systemic administration, in contrast to free Ce6, which showed negligible brain penetration (Figure 9B-D). This confirms that the CD component effectively enhances BBB penetration, likely because of its small size, surface properties, and biointeractions, making CD-Ce6 a promising platform for brain-targeted phototherapeutic interventions [124].
4.5. Sonodynamic therapy
Sonodynamic therapy is a noninvasive treatment that uses low-intensity ultrasound to activate sonosensitizers, generating ROS that induce tumor cell apoptosis. It is effective for deep-seated tumors, such as glioblastomas, owing to its tissue-penetrating ability [125]. CDs are emerging as promising nanomaterials for sonodynamic therapy in the treatment of brain diseases, particularly glioblastomas. Recently, Cheng et al. demonstrated a novel sonosensitizer using copper-doped CDs (Cu-CDs) for sonodynamic therapy of glioblastoma multiforme. The Cu-CDs were transformed into p-n-type semiconductors with a 1.58 eV bandgap and a prolonged 10.7 µs lifespan, enhancing ROS production by improving the electron-hole separation efficiency under low-intensity ultrasound. This sonodynamic effect, combined with the induction of cuproptosis (copper-mediated cell death) via the upregulation of dihydrosulfanyl transacetylase, synergistically killed glioblastoma multiforme cells (Figure 10A). Cu-CDs demonstrate excellent BBB permeability and potent antitumor activity, inhibiting glioblastoma multiforme tumor growth and increasing survival rates in mouse models, offering a promising approach for noninvasive glioblastoma multiforme treatment with high safety and efficacy [126]. In another recent report, rose bengal (RB)-modified CDs functionalized with RGD peptides, termed RB-CDs@RGD, selectively targeted integrin αvβ3 receptors that are highly expressed on glioma cells, facilitating their accumulation in brain tumors. Upon ultrasound activation during sonodynamic therapy, these nanoplatforms produce ROS, triggering oxidative stress-induced apoptosis of glioma cells. This approach offers a noninvasive treatment modality with deep-tissue penetration and high therapeutic efficacy. In vivo experiments revealed potent antitumor activity and excellent biocompatibility. In addition, the intrinsic fluorescence of RB-CDs@RGD supports fluorescence-guided surgery, accurately delineating tumor margins and reducing the risk of recurrence [127]. In this modality, CDs absorb acoustic energy and convert it into chemical energy to produce singlet oxygen (1O₂), hydroxyl radicals (•OH), and other ROS. The presence of functional groups on CDs, along with doping elements such as nitrogen, sulfur, and metals, can significantly enhance ROS production. Combined with other therapies, CD-based sonodynamic therapy offers a noninvasive method to penetrate deep brain tissues, overcoming the depth limitations associated with photodynamic therapy.
A) Schematic illustration of Cu-doped carbon dots (Cu-CDs) featuring a long-lived triplet excited state (T₁) and a p-n-type heterojunction, designed for brain imaging and sonodynamic cancer therapy. Reproduced with permission. [126] Copyright 2024, Wiley-VCH. B) Schematic illustration of SegCN-QDs@Cor-mediated microglia-targeted delivery to the mouse substantia nigra and striatum with 808 nm light assistance, along with its molecular mechanism. Reproduced with permission. [136] Copyright 2024, Elsevier. C) Overall schematic of the chemical synthesis of the multi-emission fluorescence platform and its biomedical applications. Reproduced with permission from [142], copyright 2023, Elsevier.
4.6. Antioxidant and anti-inflammatory
CDs with antioxidants and anti-inflammatory properties hold significant promise for treating CNS diseases characterized by oxidative stress and chronic neuroinflammation [128]. In conditions such as Alzheimer's disease, Parkinson's disease, traumatic brain injury, ischemic stroke, and multiple sclerosis, excessive production of ROS leads to mitochondrial dysfunction, lipid peroxidation, DNA damage, and neuronal apoptosis [129-131]. CDs, particularly those doped with heteroatoms (e.g., nitrogen, sulfur, and phosphorus) or derived from antioxidant-rich precursors, exhibit strong free radical scavenging capabilities because of their abundant surface functional groups (e.g., -OH, -COOH, and -NH2) [132]. These CDs can directly neutralize ROS and modulate redox balance within neural tissues. In parallel, CDs exert anti-inflammatory effects by downregulating pro-inflammatory cytokines (e.g., TNF-α, IL-1β, and IL-6) and inhibiting the activation of microglia and astrocytes, which are central mediators of neuroinflammation [133]. Moreover, CDs can suppress the NF-κB and MAPK signaling pathways, which are commonly overactivated during CNS inflammation [134]. Recently, CDs have been reported to enhance the delivery and efficacy of epigallocatechin-3-gallate (EGCG), a natural antioxidant, by improving its stability and brain penetration. In the neural system, EGCG-CDs act as antioxidants by scavenging ROS and as anti-inflammatory agents by inhibiting the NLRP3 inflammasome, a key driver of neuroinflammation. This dual action protects neurons, reduces oxidative stress, and suppresses inflammatory cytokines, making EGCG-CDs effective for treating brain injuries and neurodegenerative conditions [135]. Se-doped CDs (Se-CDs) act as artificial nanozymes that mimic the activities of superoxide dismutase and catalase, which are key antioxidant enzymes. In neural systems, especially under Parkinson's disease-like conditions, these nanozymes actively scavenge ROS and mitigate oxidative stress. In addition, they inhibit the NF-κB/NLRP3 inflammasome signaling pathway, which is a critical mediator of neuroinflammation. This combined action reduced both ROS accumulation and inflammatory cytokine production, protecting dopaminergic neurons from degeneration and supporting the recovery of neurological function (Figure 10B). NIR activation further enhances the therapeutic efficacy by boosting ROS clearance in the deep brain tissues [136]. Yang et al. engineered amine-functionalized aspirin-derived CDs with strong neuroprotective properties to address oxidative stress and neuroinflammation. These CDs function as efficient iron chelators, eliminating excess iron that contributes to ROS production after intracerebral hemorrhage, and act as antioxidants to directly scavenge ROS. Their application significantly reduced iron accumulation in the brain, alleviated meningeal inflammation, and enhanced neurological recovery in mice, underscoring their potential as a dual-function therapeutic approach for brain injury [137]. More recently, Wang et al. reported a rational co-assembly strategy using L-glutamate-derived CDs (LCDs) and ferulic acid (FA) to form multifunctional nanoassemblies (FA-LCDs). Notably, the enhanced BBB transport of FA-LCDs was attributed to their physicochemical properties, including ultrasmall size and surface chemistry, rather than a receptor-targeting mechanism. Once across the BBB, the FA-LCDs showed specific affinity interactions with Aβ species, inhibiting Aβ fibrillation, disassembling existing aggregates, and clearing excessive ROS. These multifunctional activities alleviated Aβ burden, oxidative stress, neuronal damage, and cognitive deficits in Alzheimer's disease mouse models, highlighting the therapeutic potential of engineered CDs for CNS disorders [138].
When functionalized with brain-targeting ligands, coated with membrane-like biological species, or rationally designed, CDs can efficiently cross the BBB and accumulate in diseased brain areas. Targeted delivery enhances therapeutic efficacy and reduces systemic side effects. Antioxidant and anti-inflammatory CDs offer a multifunctional nanoplatform for neuroprotection, capable of mitigating oxidative damage, restoring neuronal homeostasis, and modulating immune responses in various CNS pathologies.
4.7. Imaging-guided therapeutics
Imaging-guided therapeutics represent a transformative approach to treat CNS diseases, enabling real-time visualization, precise drug delivery, and monitoring of therapeutic efficacy in complex brain environments [11]. CDs are ideal candidates for theranostic systems because of their intrinsic photoluminescence, biocompatibility, and modifiability [139]. When designed for dual or multimodal imaging, such as fluorescence, photoacoustic, or MR, CD-based nanoplatforms can be used to track biodistribution and accumulation at target sites (e.g., gliomas and Alzheimer's plaques) and to monitor pathological changes during therapy [121]. For instance, CDs can be conjugated with targeting ligands (e.g., TfR, folic acid, and peptides) or cloaked in biomimetic membranes to cross the BBB and selectively accumulate in diseased areas [108]. Upon reaching the target, these nanosystems can deliver drugs or genes or induce therapeutic effects via photothermal or photodynamic therapy with laser or NIR light activation, guided by imaging feedback [140]. This allows visualization of the lesion, confirms nanocarrier accumulation, and precisely activates therapy at the desired location, minimizing damage to healthy tissues [141]. In diseases such as glioblastoma, stroke, and neurodegenerative disorders, imaging-guided CD-based therapeutics improve treatment precision, reduce systemic toxicity, and allow personalized and timely interventions by integrating diagnostics and therapy into a single, intelligent nanoplatform. Yang et al. synthesized CDs from endogenous nutrient precursors that exhibited both visible- and NIR fluorescence. These ultrasmall CDs (<5 nm) can effectively cross the BBB through size-dependent passive diffusion. Once in the brain, NIR fluorescence enables deep tissue bioimaging, and NIR laser irradiation activates the CDs for photothermal therapy. Upon NIR exposure, the CDs convert light energy into localized heat, offering a minimally invasive therapeutic approach to damage pathological tissues, such as gliomas, without harming the surrounding healthy cells (Figure 10C). The dual functionality of noninvasive imaging and NIR-triggered therapy makes these CDs promising tools for both the diagnosis and treatment of CNS diseases [142]. Highly crystalline multicolor CDs (HCCDs) have been effectively applied in dual-modal imaging-guided photothermal therapy for gliomas. These ultrasmall CDs (~6-8 nm) exhibit bright multicolor fluorescence and excellent photothermal conversion efficiencies under NIR light. Their crystalline structures enhance stability and light absorption, allowing them to generate localized heat upon NIR exposure. After systemic administration in mice bearing intracranial U87 glioma tumors, HCCDs successfully crossed the BBB and selectively accumulated in tumor tissues. Real-time tumor imaging was achieved using both fluorescence and photoacoustic modalities, guiding precise NIR laser irradiation. Upon laser exposure, localized heating leads to effective tumor cell ablation with minimal damage to the surrounding healthy brain tissue. This approach demonstrates high therapeutic efficacy, minimal toxicity, and potential for clinical translation in the treatment of brain cancers such as glioma [121]. In another report, gadolinium-doped CDs (Gd-CDs) were used as multimodal agents for photothermal therapy and tumor monitoring in brain cancer. These biocompatible CDs absorbed NIR light and generated heat to ablate glioblastoma cells via photothermal therapy. Their gadolinium content enhances MRI contrast for tumor visualization, whereas their fluorescence enables optical imaging. Small-sized Gd-CDs penetrate the BBB and achieve effective tumor reduction in vivo with low toxicity, making them promising theranostic platforms for brain cancer treatment and monitoring [143].
CDs represent a versatile class of nanotheranostic agents for CNS pathologies that integrate multimodal diagnostic capabilities with targeted therapy delivery. Their intrinsic photoluminescence facilitates high-resolution, non-invasive neuroimaging, while their tunable physicochemical properties and capacity for BBB translocation permit the precise delivery of pharmacological, genetic, or molecular payloads [11, 86]. Coupling real-time imaging feedback with therapeutic activation allows spatiotemporal control of photothermal, photodynamic, sonodynamic, chemotherapeutic, or combinatorial interventions, maximizing on-target efficacy and minimizing collateral damage. Preclinical investigations have demonstrated the utility of CDs as both intrinsic and vectorized photo/sonosensitizers in gliomas and other brain tumor models, as well as their capacity to elicit photothermal therapy, ROS-mediated oxidative stress, antioxidant modulation, and anti-inflammatory responses (Figure 11A and B) [128, 133]. These multifunctional attributes make CDs a next-generation platform for precision neurotherapeutics.
A) Schematic diagram of the preparation of three herbal CDs and their applications as dynamic ROS nanomodulators for anti-inflammatory and antibacterial purposes. Reproduced with permission from [128], copyright 2024, American Chemical Society. B) Preparation process and action mechanism of caffeic acid-CDs coated with microglia membrane (CDs-CA-MGs). Reproduced with permission from [133], copyright 2024, Springer Nature.
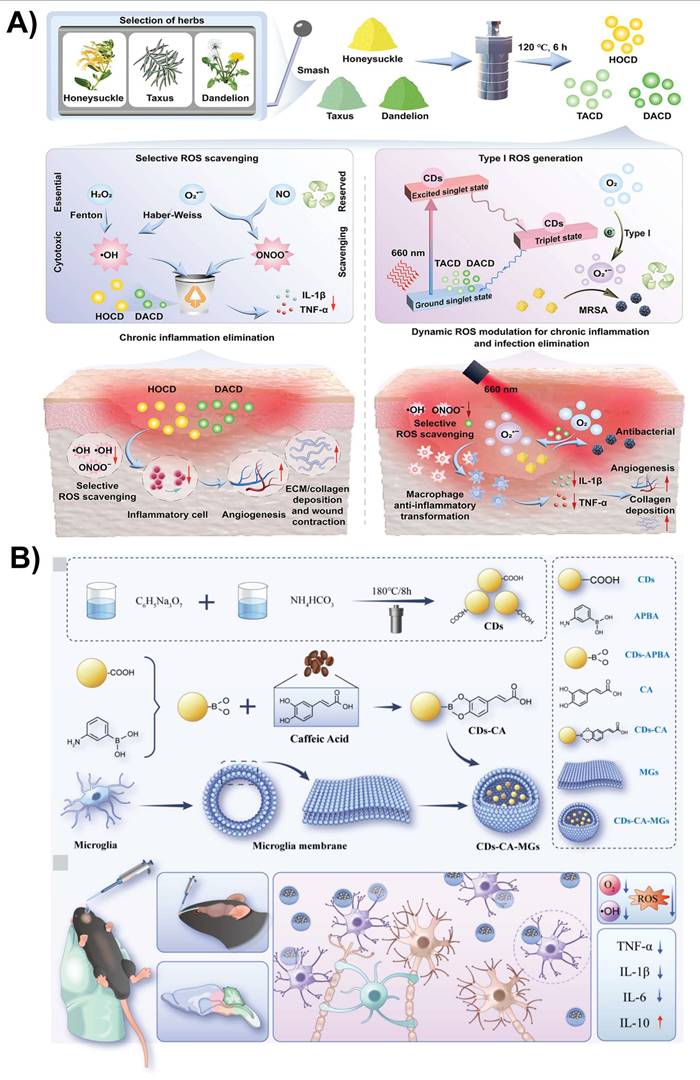
Owing to their ultrasmall size, surface chemistry, and BBB penetration ability, CDs can interact with key components of the tumor microenvironment and modulate immune responses through both direct and indirect mechanisms. Functionalized CDs have shown to influence macrophage and microglial polarizations, promoting a shift from immunosuppressive (M2-type) phenotypes toward pro-inflammatory, anti-tumor (M1-type) states, thereby enhancing local immune activation. Furthermore, CD-based nanoplatforms could facilitate antigen presentation by dendritic cells and improve T-cell priming via delivery of immunogenic cargos or adjuvants, conceptually aligning with established cancer immunotherapy strategies [144]. For example, Hu et al. [133] functionalized CDs with bioactive molecules, which exhibited immunomodulatory effects in a brain disease model by attenuating neuroinflammation rather than directly activating adaptive immunity. The formulated CDs penetrated the BBB and reduced pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 in the brain tissue, suggesting pathological immune activation (Figure 11B). This was accompanied by modulation of microglial activation and reduction of oxidative stress-driven inflammatory signaling. These findings suggest that, while current CD-based approaches function primarily through innate immune modulation rather than classical immune checkpoint or T-cell-based immunotherapy, they provide a mechanistic foundation for developing CD nanoplatforms that integrate neuroimmune regulation into future CNS immunotherapeutic strategies.
To be effective in brain theranostics, CDs must meet several design requirements, which are summarized in Table 4. By fulfilling these design criteria, CDs have the potential to be used as next-generation nanotheranostic agents for the personalized diagnosis and treatment of complex CNS diseases.
Summary of CDs for theranostic applications in brain diseases and their design requirements
| Stages (↓) | Biological target /barrier | CD properties/modifications required | Predicted function | Ref. |
|---|---|---|---|---|
| Administration & circulation | Systemic circulation (IV injection) | High colloidal stability Long circulation time Biocompatibility & non-toxicity | Avoid immune clearance and reach brain vasculature | [21, 173] |
| BBB penetration | Tight junctions of endothelial cells | Less than 10 nm in size Surface functionalization with TfR, ApoE, TAT peptide, and exosome coating Neutral/positive surface charge | Transcytosis / receptor-mediated transport across the BBB | [91, 95] |
| Brain targeting | Neurons, glia, tumor cells | Ligand-receptor binding (e.g., EGFR, TfR) pH-responsive or ROS-triggered release Stimuli-responsive targeting | Accumulate at pathological sites (tumors, plaques) | [116, 151, 152] |
| Theranostic action | Cellular level | Fluorescence/MR imaging Photothermal therapy Photodynamic therapy Drug/gene delivery Antioxidant & anti-inflammatory activity | Combined diagnosis & treatment | [11] |
| Biodegradation & clearance | Metabolism and excretion | Biodegradable core Renal clearance (<5.5 nm) | Safe elimination from the body | [174, 175] |
ApoE: Apolipoprotein E; BBB: Blood-brain barrier; CDs: carbon dots; EGFR: Epidermal Growth Factor Receptor; IV: intravenous; MRI: Magnetic resonance imaging; ROS: Reactive oxygen species; TAT: Trans-activating transcriptional activator; TfR: Transferrin Receptor.
5. Current limitations and future perspectives
In general, the translation of CDs to clinical settings is hindered by several critical interrelated obstacles, including a lack of synthesis standardizations, physicochemical instability, unclear biological fate, and incomplete safety evaluation. A major challenge is the lack of scalable and reproducible manufacturing protocols. Batch-to-batch variability in precursor composition, reaction condition, and post-synthesis processing often results in heterogeneous size distributions and inconsistent photoluminescence properties, which significantly compromise reproducibility and regulatory acceptance.
Surface functionalization, while essential for targeting and drug loading, can introduce additional complexity. In many cases, adding targeting ligands or functional groups induces aggregation, causes surface oxidation, and deteriorates the emission profile. Furthermore, weakly bound surface moieties may detach from the carbon core before reaching the target, reducing the therapeutic efficacy of CDs. Although CDs have been reported to have lower toxicity compared with semiconductor quantum dots, information regarding their long-term effects, pharmacokinetics, and body clearance remains inadequate, particularly under repeated dosing and chronic administration. The absence of standardized metrics for dimensional characterization (hydrodynamic vs. core size), surface chemistry quantification, and photophysical property assessment contribute to inconsistent reporting and complicated data interpretation across studies. The in vivo fate of CDs, including renal versus hepatobiliary clearance pathways, biodegradation, organ accumulation, immunogenicity, and potential genotoxicity, requires comprehensive long-term evaluation. Even with high-affinity targeting ligands, only a small portion of systemically administered CDs reaches therapeutically relevant concentrations in the brain parenchyma, with considerable off-target accumulation in clearance organs such as the liver and spleen.
In contrast, several FDA-approved nanomedicines, such as liposomal formulations and polymer-based nanoparticles, have well-defined physicochemical specifications, pharmacokinetic parameters, clearance pathways, and safety profiles established through rigorous preclinical and clinical evaluation. Compared with clinically approved platforms, CDs lack standardized and comprehensive pharmacokinetic parameters, such as quantitative brain-to-blood concentration ratios, administered dose accumulated in the brain, organ-level biodistribution profiles, clearance pathways, and long-term immunotoxicity assessments. Therefore, direct comparison with established nanocarriers, as well as among different CD formulations, is difficult, hindering the rational positioning of CDs within the existing nanotherapeutics landscape.
Live cell-based delivery has recently emerged as a promising approach for brain targeting. Integrating CDs with living carriers, such as immune or stem cells, may synergistically combine active homing and BBB-crossing capabilities with CD-based theranostics, although this strategy remains at an early proof-of-concept stage and requires further investigation to address biosafety and translational challenges.
To advance clinical translation, future research should prioritize the rational design of ultrasmall (<10 nm) CDs with engineered NIR emission for deep-tissue imaging and rapid renal clearance. Developing stimuli-responsive nanoplatforms such as pH-, enzyme-, ROS-, or ultrasound-responsive linkers could enable spatiotemporally controlled drug release while minimizing off-target effects. Equally important is the adoption of harmonized characterization standards. Comprehensive toxicological studies, including repeat-dose, reproductive, and genotoxic endpoints, coupled with biodistribution and clearance data, are essential for accelerating clinical translation. Systematic comparison of CDs with clinically approved nanomedicines, using consistent dosing, administration routes, and evaluation endpoints, will be essential to assess their translational potential and to position CDs within the broader landscape of clinically viable nanotherapeutics.
6. Summary
This review summarizes recent advances in the design of CDs that cross the BBB for targeting, diagnosis, and treatment of CNS diseases, or for theranostics. To facilitate effective BBB crossing, CDs can be functionalized with targeting ligands, coated with polymers, or modified with biomimetic membranes, exosomes, or other ligands, allowing them to target diseased cells or biomarkers. The collective engineering of these functional groups supports precise control over the surface charge of CDs, which is pivotal for stability against aggregation in physiological solutions and for directing cell-specific targeting. Surface charges affect how CDs interact with cell membranes, dictating internalization pathways and ultimately their intracellular fate. CDs have emerged as highly promising nanomaterials for the diagnosis and treatment of neurodegenerative diseases and brain cancers owing to their unique physicochemical and biological properties. Their ultrasmall size, tunable surface functionalities, excellent photoluminescence, and biocompatibility enable them to cross the BBB efficiently and selectively accumulate in brain tissue. CDs can serve as multimodal imaging agents that offer fluorescence, MRI, or photoacoustic imaging, facilitating early detection and real-time monitoring of brain diseases. In addition, they can be engineered to deliver drugs, genes, or therapeutic agents specifically to diseased areas, reducing off-target effects. Their antioxidants, anti-inflammatory, photothermal, and photodynamic therapeutic properties further enhance their potential as theranostic agents. These attributes collectively position CDs as a transformative platform for the integrated diagnosis and targeted therapy of complex CNS disorders. Hybrid nanotherapeutic strategies that integrate CDs carriers with targeting peptides, receptor-specific ligands, PEG coatings, or stimuli-responsive modalities (light, ultrasound, and ROS) have demonstrated enhanced BBB penetration and disease-specific payload release. Biomimetic cloaking, such as macrophage membrane coating, introduces endothelial adhesion proteins (e.g. vascular cell adhesion molecule-1 ligands) that facilitate the selective recognition of the inflamed brain vasculature, further improving targeting precision. Despite these advances, significant translation barriers exist. CDs synthesized from diverse precursors and fabrication routes exhibit substantial heterogeneity in terms of particle size, surface functionality, quantum yield, and residual impurities, hindering their cross-study comparability and complicating regulatory validation.
Overall, the rapid progress in CD engineering strategies has established a strong foundation for their application in CNS targeting, diagnosis, and therapy. Continued refinement in synthesis control and surface design is expected to improve reproducibility and translational robustness, reinforcing the potential of CDs as versatile and clinically relevant nanoplatforms for brain diseases.
Abbreviations
5-HTA1A/2A: 5-hydroxytryptamine (serotonin) receptors 1A and 2A; AMT: adsorption-mediated transcytosis; APP: amyloid precursor protein; AFM: atomic force microscopy; Aβ: β-amyloid; BDNF: brain-derived neurotrophic factor; BBB: blood-brain barrier; BTB: blood-tumor barrier; BNCT: boron neutron capture therapy; 10BCDs: boron-enriched CDs; CDs: carbon dots; CNS: central nervous system; Cur-CDs: curcumin-derived CDs; DOX: doxorubicin; EGFR: epidermal growth factor receptor; EGCG: epigallocatechin-3-gallate; FA: ferulic acid; FTIR: Fourier transform infrared spectroscopy; GLUT1: glucose transporter 1; HCCDs: highly crystalline multicolor CDs; HR-TEM: high-resolution transmission electron microscopy; IL-6: interleukin-6; LAT1: L-type amino acid transporter 1; LCDs: L-glutamate-derived CDs; LRP1: low-density lipoprotein receptor-related protein 1; LAT1: L-type amino acid transporter 1 (LAT1); NIR: near infrared; N-CDs: nitrogen-doped CDs (N-CDs); NMDA: N-methyl-D-aspartate receptor; PEG-Lf: polyethylene glycol lactoferrin; PCDs: polydopamine-CDs; PEG: polyethylene glycol; PEI: polyethyleneimine; RVG29: rabies virus glycoprotein 29; RAGE: receptor for advanced glycation end products; RMT: receptor-mediated transcytosis; ROS: reactive oxygen species; TfR: transferrin; TfR-CDs: TfR-conjugated CDs; TME: tumor microenvironment; TREM2: triggering receptor expressed on myeloid cells 2; XPS: X-ray photoelectron spectroscopy.
Acknowledgements
This study was financially supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), supported by the Ministry of Education, under grant NRF-2018R1A6A1A03025159. We also thank the BT Research facility center at Chung-Ang University.
Contributions
Wubshet Mekonnen Girma: Writing - original draft, review & editing, Investigation, Conceptualization. Girum Getachew Demissie: Investigation, Data curation, Writing - review & editing. Shewaye Lakew Mekuria: Investigation, Writing - review & editing. Shamsa Kizhepat: Visualization, Data curation. T.M. Subrahmanya: Validation, formal analysis. Akash S. Rasal: Formal analysis, Data curation. Binyam Abdu Berhe: Data curation, Software. Gangaraju Gedda: Writing - review & editing. Yoo-Jin Park: Writing - review & editing. Jia-Yaw Chang: Writing - review & editing. Myung-Geol Pang: Writing - review & editing, Resources, Supervision, Funding acquisition.
Competing interests
The authors have declared that no competing interest exists.
References
1. Khan AR, Yang X, Fu M, Zhai G. Recent progress of drug nanoformulations targeting to brain. J Control Release. 2018;291:37-64
2. Collaborators GBDN. Global, regional, and national burden of neurological disorders, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:459-80
3. Collaborators GBDNSD. Global, regional, and national burden of disorders affecting the nervous system, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024;23:344-81
4. Khan AR, Liu M, Khan MW, Zhai G. Progress in brain targeting drug delivery system by nasal route. J Control Release. 2017;268:364-89
5. Dai W, Zhan M, Gao Y, Sun H, Zou Y, Laurent R. et al. Brain delivery of fibronectin through bioactive phosphorous dendrimers for Parkinson's disease treatment via cooperative modulation of microglia. Bioact Mater. 2024;38:45-54
6. Selkoe DJ, Hardy J. The amyloid hypothesis of Alzheimer's disease at 25 years. EMBO Mol Med. 2016;8:595-608
7. Menon DK, Schwab K, Wright DW, Maas AI, Demographics, Clinical Assessment Working Group of the I. et al. Position statement: definition of traumatic brain injury. Arch Phys Med Rehabil. 2010;91:1637-40
8. Wang J, Wang Z, Zhang G, Rodrigues J, Tomas H, Shi X. et al. Blood-brain barrier-crossing dendrimers for glioma theranostics. Biomater Sci. 2024;12:1346-56
9. Li L, Lu Y, Ye X, Zhang C, Liu J, Yang Z. et al. Hydrophobic Carbon Dots Prevent α-Synucleinopathy and Suppress Neuroinflammation to Treat Parkinson's Disease. Aggregate. 2024;6:e711
10. Xiao T, He M, Xu F, Fan Y, Jia B, Shen M. et al. Macrophage Membrane-Camouflaged Responsive Polymer Nanogels Enable Magnetic Resonance Imaging-Guided Chemotherapy/Chemodynamic Therapy of Orthotopic Glioma. ACS Nano. 2021;15:20377-90
11. Ashrafizadeh M, Mohammadinejad R, Kailasa SK, Ahmadi Z, Afshar EG, Pardakhty A. Carbon dots as versatile nanoarchitectures for the treatment of neurological disorders and their theranostic applications: A review. Adv Colloid Interface Sci. 2020;278:102123
12. Arvanitis CD, Ferraro GB, Jain RK. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat Rev Cancer. 2020;20:26-41
13. Zhou Y, Peng Z, Seven ES, Leblanc RM. Crossing the blood-brain barrier with nanoparticles. J Control Release. 2018;270:290-303
14. Yalamandala BN, Moorthy T, Liu Z-H, Huynh TMH, Iao HM, Pan W-C. et al. A Self-Cascading Catalytic Therapy and Antigen Capture Scaffold-Mediated T Cells Augments for Postoperative Brain Immunotherapy. Small. 2025;21:2406178
15. Steeg PS. The blood-tumour barrier in cancer biology and therapy. Nat Rev Clin Oncol. 2021;18:696-714
16. Sarkaria JN, Hu LS, Parney IF, Pafundi DH, Brinkmann DH, Laack NN. et al. Is the blood-brain barrier really disrupted in all glioblastomas? A critical assessment of existing clinical data. Neuro Oncol. 2018;20:184-91
17. Pardridge WM. The blood-brain barrier: bottleneck in brain drug development. NeuroRx. 2005;2:3-14
18. Si Y, Cha R, Zhang P, Zhou F, Yuan W, Jiang X. CD-MOF Based pH/ROS Sensitive 5-ASA Delivery System for Ulcerative Colitis Therapy. Adv Healthc Mater. 2025;14:2501160
19. Yalamandala BN, Chen Y-J, Lin Y-H, Huynh TMH, Chiang W-H, Chou T-C. et al. A Self-Cascade Penetrating Brain Tumor Immunotherapy Mediated by Near-Infrared II Cell Membrane-Disrupting Nanoflakes via Detained Dendritic Cells. ACS Nano. 2024;18:18712-28
20. Moorthy T, Chen C-K, Liu Z-H, Yalamandala BN, Huynh TMH, Iao HM. et al. Wireless chargeable gold Yarnball-mediated mitochondrial depolarization for dendritic cell detainment in programmed brain tumor immunotherapy. Nano Today. 2025;65:102838
21. Han M, Yi B, Song R, Wang D, Huang N, Ma Y. et al. Fucoidan-derived carbon dots as nanopenetrants of blood-brain barrier for Parkinson's disease treatment. J Colloid Interface Sci. 2025;680:516-27
22. Zhang W, Smith N, Zhou Y, McGee CM, Bartoli M, Fu S. et al. Carbon dots as dual inhibitors of tau and amyloid-beta aggregation for the treatment of Alzheimer's disease. Acta Biomater. 2024;183:341-55
23. Wang W, Lin X, Dong X, Sun Y. A multi-target theranostic nano-composite against Alzheimer's disease fabricated by conjugating carbon dots and triple-functionalized human serum albumin. Acta Biomater. 2022;148:298-309
24. Yan C, Shao X, Wang Y, Tang S, Li S, Wang C. et al. Application of carbon dots-based nanomaterials in amyloid aggregation disease. Carbon. 2025;234:119971
25. Xu X, Ray R, Gu Y, Ploehn HJ, Gearheart L, Raker K. et al. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J Am Chem Soc. 2004;126:12736-7
26. Sun Y-P, Zhou B, Lin Y, Wang W, Fernando KS, Pathak P. et al. Quantum-sized carbon dots for bright and colorful photoluminescence. J Am Chem Soc. 2006;128:7756-7
27. Chiu S-H, Gedda G, Girma WM, Chen J-K, Ling Y-C, Ghule AV. et al. Rapid fabrication of carbon quantum dots as multifunctional nanovehicles for dual-modal targeted imaging and chemotherapy. Acta Biomater. 2016;46:151-64
28. Cui L, Ren X, Wang J, Sun M. Synthesis of homogeneous carbon quantum dots by ultrafast dual-beam pulsed laser ablation for bioimaging. Mater Today Nano. 2020;12:100091
29. Yuan F, Li S, Fan Z, Meng X, Fan L, Yang S. Shining carbon dots: Synthesis and biomedical and optoelectronic applications. Nano Today. 2016;11:565-86
30. Yao Y-Y, Gedda G, Girma WM, Yen C-L, Ling Y-C, Chang J-Y. Magnetofluorescent carbon dots derived from crab shell for targeted dual-modality bioimaging and drug delivery. ACS Appl Mater Interfaces. 2017;9:13887-99
31. Dehvari K, Chiu S-H, Lin J-S, Girma WM, Ling Y-C, Chang J-Y. Heteroatom doped carbon dots with nanoenzyme like properties as theranostic platforms for free radical scavenging, imaging, and chemotherapy. Acta Biomater. 2020;114:343-57
32. Yue J-s, Yuan F-y, Qiu H-x, Li Y, Li J, Xue Y-h. et al. A review of fluorescent carbon dots: synthesis, photoluminescence mechanism, solid-state photoluminescence and applications in white light-emitting diodes. New Carbon Mater. 2023;38:478-92
33. Ge G, Li L, Wang D, Chen M, Zeng Z, Xiong W. et al. Carbon dots: synthesis, properties and biomedical applications. J Mater Chem B. 2021;9:6553-75
34. Girma WM, Mekuria SL, Gedda G, Shi X, Park Y-J, Pang M-G. Carbon dot-based nanohybrids for tumor microenvironment-activated cancer theranostics. Coord Chem Rev. 2026;548:217236
35. Alafeef M, Srivastava I, Aditya T, Pan D. Carbon Dots: From Synthesis to Unraveling the Fluorescence Mechanism. Small. 2024;20:2303937
36. Yan F, Jiang Y, Sun X, Bai Z, Zhang Y, Zhou X. Surface modification and chemical functionalization of carbon dots: a review. Microchim Acta. 2018;185:424
37. Sharma AS, Lee NY. Comprehensive review on fluorescent carbon dots and their applications in nucleic acid detection, nucleolus targeted imaging and gene delivery. Analyst. 2024;149:4095-115
38. Jiang H, Chen F, Lagally MG, Denes FS. New strategy for synthesis and functionalization of carbon nanoparticles. Langmuir. 2010;26:1991-5
39. Yang S, Zeng H, Zhao H, Zhang H, Cai W. Luminescent hollow carbon shells and fullerene-like carbon spheres produced by laser ablation with toluene. J Mater Chem. 2011;21:4432-6
40. Liu H, Zhong X, Pan Q, Zhang Y, Deng W, Zou G. et al. A review of carbon dots in synthesis strategy. Coord Chem Rev. 2024;498:215468
41. Ye Z, Li G, Lei J, Liu M, Jin Y, Li B. One-step and one-precursor hydrothermal synthesis of carbon dots with superior antibacterial activity. ACS Appl Bio Mater. 2020;3:7095-102
42. Li H, He X, Liu Y, Huang H, Lian S, Lee S-T. et al. One-step ultrasonic synthesis of water-soluble carbon nanoparticles with excellent photoluminescent properties. Carbon. 2011;49:605-9
43. Lesani P, Lu Z, Singh G, Mursi M, Mirkhalaf M, New EJ. et al. Influence of carbon dot synthetic parameters on photophysical and biological properties. Nanoscale. 2021;13:11138-49
44. García-Salcedo ÁJ, Giraldo-Pinto LÁ, Márquez-Castro DJ, Tirado-Mejía L. Influence of synthesis parameters on the optical properties of carbon dots. Carbon Trends. 2024;17:100403
45. Hu W, Zhao S, Sun C, Song X, Madadi M, Chiani E. et al. Structural and functional divergence of lignin-derived carbon dots and their nucleation pathways under varying alcohol-solvothermal treatments. Green Chem. 2025;27:11093-106
46. Newman Monday Y, Abdullah J, Yusof NA, Abdul Rashid S, Shueb RH. Facile hydrothermal and solvothermal synthesis and characterization of nitrogen-doped carbon dots from palm kernel shell precursor. Appl Sci. 2021;11:1630
47. Xue S, Li P, Sun L, An L, Qu D, Wang X. et al. The formation process and mechanism of carbon dots prepared from aromatic compounds as precursors: a review. Small. 2023;19:2206180
48. Moniruzzaman M, Kim J. Synthesis and post-synthesis strategies for polychromatic carbon dots toward unique and tunable multicolor photoluminescence and associated emission mechanism. Nanoscale. 2023;15:13858-85
49. Reckmeier CJ, Wang Y, Zboril R, Rogach AL. Influence of Doping and Temperature on Solvatochromic Shifts in Optical Spectra of Carbon Dots. J Phys Chem C. 2016;120:10591-604
50. Devi S, Luwang MN. Engineering Luminescent Carbon Dot Emission through Surface State Functional Group via Heteroatom Doping and Unveiling the Effect of Solvents. Langmuir. 2025;41:13751-62
51. Xu Q, Kuang T, Liu Y, Cai L, Peng X, Sreeprasad TS. et al. Heteroatom-doped carbon dots: synthesis, characterization, properties, photoluminescence mechanism and biological applications. J Mater Chem B. 2016;4:7204-19
52. Sun Z, Yan F, Xu J, Zhang H, Chen L. Solvent-controlled synthesis strategy of multicolor emission carbon dots and its applications in sensing and light-emitting devices. Nano Res. 2022;15:414-22
53. Li L, Dong T. Photoluminescence tuning in carbon dots: surface passivation or/and functionalization, heteroatom doping. J Mater Chem C. 2018;6:7944-70
54. Rooj B, Mandal U. A review on characterization of carbon quantum dots. Vietnam J Chem. 2023;61:693-718
55. Lin Y, Wang C, Li L, Wang H, Liu K, Wang K. et al. Tunable fluorescent silica-coated carbon dots: a synergistic effect for enhancing the fluorescence sensing of extracellular Cu2+ in rat brain. ACS Appl Mater Interfaces. 2015;7:27262-70
56. Strauss V, Margraf JT, Dolle C, Butz B, Nacken TJ, Walter J. et al. Carbon nanodots: toward a comprehensive understanding of their photoluminescence. J Am Chem Soc. 2014;136:17308-16
57. Shu G, Jiang TT, Zhang XN, Zhao HX. Gadolinium and Ytterbium-Doped carbon dots for Fluorescence/Magnetic Resonance/Computed tomography multimodal imaging. Chem Eng J. 2024;494:153086
58. Baker SN, Baker GA. Luminescent Carbon Nanodots: Emergent Nanolights. Angew Chem, Int Ed. 2010;49:6726-44
59. Zhang Q, Wang R, Feng B, Zhong X, Ostrikov K. Photoluminescence mechanism of carbon dots: triggering high-color-purity red fluorescence emission through edge amino protonation. Nat Commun. 2021;12:6856
60. Roy P, Chen P-C, Periasamy AP, Chen Y-N, Chang H-T. Photoluminescent carbon nanodots: synthesis, physicochemical properties and analytical applications. Mater Today. 2015;18:447-58
61. Falara PP, Zourou A, Kordatos KV. Recent advances in Carbon Dots/2-D hybrid materials. Carbon. 2022;195:219-45
62. Priya u, Chaudhary R. Historical review and recent advances in the field of carbon dots with their applications. Mater Chem Phys. 2025;4:100028
63. Saleh M, Gul A, Nasir A, Moses TO, Nural Y, Yabalak E. Comprehensive review of Carbon-based nanostructures: Properties, synthesis, characterization, and cross-disciplinary applications. J Ind Eng Chem. 2025;146:176-212
64. Wang B, Lu S. The light of carbon dots: From mechanism to applications. Matter. 2022;5:110-49
65. Ozyurt D, Kobaisi MA, Hocking RK, Fox B. Properties, synthesis, and applications of carbon dots: A review. Carbon Trends. 2023;12:100276
66. Ru Y, Waterhouse GIN, Lu S. Aggregation in carbon dots. Aggregate. 2022;3:e296
67. Wang P, Chen C, Ren H, Duan E. A review of carbon dots in synthesis strategies, photoluminescence mechanisms, and applications in wastewater treatment. Chin Chem Lett. 2024: 110725.
68. Gonzalez-Vera AS, Perrin G, Botsoa J, Ntsoenzok E, Chazaro-Ruiz LF, Rangel-Mendez JR. et al. Analyzing photoluminescence emission quenching effects in colloidal aqueous suspensions of carbon dots. Colloids Surf, A. 2026;728:138806
69. Ferreyra DD, Rodríguez Sartori D, Ezquerra Riega SD, Rodríguez HB, Gonzalez MC. Tuning the nitrogen content of carbon dots in carbon nitride nanoflakes. Carbon. 2020;167:230-43
70. Wang Y, Hu A. Carbon quantum dots: synthesis, properties and applications. J Mater Chem C. 2014;2:6921-39
71. Gong X, Wu Y, Li J, Zhang S, Wu M. Rational design of long wavelength solid-state fluorescent carbon dots based on zirconium. Carbon. 2024;226:119208
72. Li D, Wang D, Zhao X, Xi W, Zebibula A, Alifu N. et al. Short-wave infrared emitted/excited fluorescence from carbon dots and preliminary applications in bioimaging. Mater Chem Front. 2018;2:1343-50
73. Seo G, Kim B-s, Lim H, Choi J, Kim M, Lee H. et al. Biomedical applications and future perspectives of carbon dots and their hybrid nanomaterials. Mater Adv. 2026
74. Ding H, Yu S-B, Wei J-S, Xiong H-M. Full-Color Light-Emitting Carbon Dots with a Surface-State-Controlled Luminescence Mechanism. ACS Nano. 2016;10:484-91
75. Song G, Plumlee P, Ahn JY, Wong ST, Zhao H. Translational strategies and systems biology insights for blood-brain barrier opening and delivery in brain tumors and Alzheimer's disease. Biomed Pharmacother. 2023;167:115450
76. Upton DH, Ung C, George SM, Tsoli M, Kavallaris M, Ziegler DS. Challenges and opportunities to penetrate the blood-brain barrier for brain cancer therapy. Theranostics. 2022;12:4734-52
77. Bharadwaj VN, Rowe RK, Harrison J, Wu C, Anderson TR, Lifshitz J. et al. Blood-brainbarrier disruption dictates nanoparticle accumulation following experimental brain injury. Nanomedicine. 2018;14:2155-66
78. Houston ZH, Bunt J, Chen KS, Puttick S, Howard CB, Fletcher NL. et al. Understanding the Uptake of Nanomedicines at Different Stages of Brain Cancer Using a Modular Nanocarrier Platform and Precision Bispecific Antibodies. ACS Cent Sci. 2020;6:727-38
79. Girma WM, Zhu Z, Guo Y, Xiao X, Wang Z, Mekuria SL. et al. Synthesis and Characterization of Copper-Crosslinked Carbon Dot Nanoassemblies for Efficient Macrophage Manipulation. Macromol Rapid Commun. 2024;45:e2400511
80. Chan KK, Yap SHK, Yong KT. Biogreen Synthesis of Carbon Dots for Biotechnology and Nanomedicine Applications. Nanomicro Lett. 2018;10:72
81. Zhang W, Sigdel G, Mintz KJ, Seven ES, Zhou Y, Wang C. et al. Carbon Dots: A Future Blood-Brain Barrier Penetrating Nanomedicine and Drug Nanocarrier. Int J Nanomedicine. 2021;16:5003-16
82. Shao X, Cao X, Fan T, Yan C, Wang C, Wang J. et al. Tannic acid-derived carbon dots: Dual-function inhibitors against β-amyloid fibrosis and bacterial infections. Int J Biol Macromol. 2025;322:146516
83. Habib S, Singh M. Angiopep-2-Modified Nanoparticles for Brain-Directed Delivery of Therapeutics: A Review. Polymers. 2022 14
84. Li S, Peng Z, Dallman J, Baker J, Othman AM, Blackwelder PL. et al. Crossing the blood-brain-barrier with transferrin conjugated carbon dots: A zebrafish model study. Colloids Surf, B. 2016;145:251-6
85. You L, Wang J, Liu T, Zhang Y, Han X, Wang T. et al. Targeted Brain Delivery of Rabies Virus Glycoprotein 29-Modified Deferoxamine-Loaded Nanoparticles Reverses Functional Deficits in Parkinsonian Mice. ACS Nano. 2018;12:4123-39
86. Seven ES, Seven YB, Zhou Y, Poudel-Sharma S, Diaz-Rucco JJ, Kirbas Cilingir E. et al. Crossing the blood-brain barrier with carbon dots: uptake mechanism and in vivo cargo delivery. Nanoscale Adv. 2021;3:3942-53
87. Muhammad P, Hanif S, Li J, Guller A, Rehman FU, Ismail M. et al. Carbon dots supported single Fe atom nanozyme for drug-resistant glioblastoma therapy by activating autophagy-lysosome pathway. Nano Today. 2022;45:101530
88. Vallejo FA, Sigdel G, Veliz EA, Leblanc RM, Vanni S, Graham RM. Carbon Dots in Treatment of Pediatric Brain Tumors: Past, Present, and Future Directions. Int J Mol Sci. 2023 24
89. Parikh K, Seven E, Luca E, Mitchell G, LeBlanc R, Seven Y. Glucose Carbon Dots Cross the Blood-Brain Barrier and Preferentially Target Spinal Neurons. Physiology. 2023;38:5733000
90. Zha S, Liu H, Li H, Li H, Wong K-L, All AH. Functionalized Nanomaterials Capable of Crossing the Blood-Brain Barrier. ACS Nano. 2024;18:1820-45
91. Araújo C, Rodrigues RO, Bañobre-López M, Silva AMT, Ribeiro RS. Carbon Dots as a Fluorescent Nanosystem for Crossing the Blood-Brain Barrier with Plausible Application in Neurological Diseases. Pharmaceutics. 2025 17
92. Koo J, Lim C, Oh KT. Recent Advances in Intranasal Administration for Brain-Targeting Delivery: A Comprehensive Review of Lipid-Based Nanoparticles and Stimuli-Responsive Gel Formulations. Int J Nanomedicine. 2024;19:1767-807
93. Formica ML, Real DA, Picchio ML, Catlin E, Donnelly RF, Paredes AJ. On a highway to the brain: A review on nose-to-brain drug delivery using nanoparticles. Appl Mater Today. 2022;29:101631
94. Liu J, Wang T, Dong J, Lu Y. The blood-brain barriers: novel nanocarriers for central nervous system diseases. J Nanobiotechnol. 2025;23:146
95. Zhou Y, Liyanage PY, Devadoss D, Rios Guevara LR, Cheng L, Graham RM. et al. Nontoxic amphiphilic carbon dots as promising drug nanocarriers across the blood-brain barrier and inhibitors of beta-amyloid. Nanoscale. 2019;11:22387-97
96. Tripathi S, Singh R, Jaiswal S, Sethi M, Rana R, Mishra K. et al. Targeting Alzheimer's pathophysiology with carbon dots: from mechanisms to therapy. J Mater Chem B. 2026;14:389-422
97. Calabrese G, De Luca G, Nocito G, Rizzo MG, Lombardo SP, Chisari G. et al. Carbon Dots: An Innovative Tool for Drug Delivery in Brain Tumors. Int J Mol Sci. 2021;22:11783
98. Niu Y, Tan H, Li X, Zhao L, Xie Z, Zhang Y. et al. Protein-Carbon Dot Nanohybrid-Based Early Blood-Brain Barrier Damage Theranostics. ACS Appl Mater Interfaces. 2020;12:3445-52
99. Wang S, Li C, Qian M, Jiang H, Shi W, Chen J. et al. Augmented glioma-targeted theranostics using multifunctional polymer-coated carbon nanodots. Biomaterials. 2017;141:29-39
100. Lim JL, Lin CJ, Huang CC, Chang LC. Curcumin-derived carbon quantum dots: Dual actions in mitigating tau hyperphosphorylation and amyloid beta aggregation. Colloids Surf, B. 2024;234:113676
101. Wang Y, Meng Y, Wang S, Li C, Shi W, Chen J. et al. Direct Solvent-Derived Polymer-Coated Nitrogen-Doped Carbon Nanodots with High Water Solubility for Targeted Fluorescence Imaging of Glioma. Small. 2015;11:3575-81
102. Saraiva C, Praca C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: Overcoming blood-brain barrier to treat neurodegenerative diseases. J Control Release. 2016;235:34-47
103. Guo W, Ji M, Li Y, Qian M, Qin Y, Li W. et al. Iron ions-sequestrable and antioxidative carbon dot-based nano-formulation with nitric oxide release for Parkinson's disease treatment. Biomaterials. 2024;309:122622
104. Fang RH, Kroll AV, Gao W, Zhang L. Cell Membrane Coating Nanotechnology. Adv Mater. 2018;30:1706759
105. Ye P, Li L, Qi X, Chi M, Liu J, Xie M. Macrophage membrane-encapsulated nitrogen-doped carbon quantum dot nanosystem for targeted treatment of Alzheimer's disease: Regulating metal ion homeostasis and photothermal removal of beta-amyloid. J Colloid Interface Sci. 2023;650:1749-61
106. Li J, Kong JL, Ma SH, Li JC, Mao MR, Chen K. et al. Exosome-Coated B Carbon Dots for Precise Boron Neutron Capture Therapy in a Mouse Model of Glioma In Situ. Adv Funct Mater. 2021;31:2100969
107. Yang C, Song Y, Luo M, Wang Q, Zhang Y, Cen J. et al. Exosomes-encapsulated biomimetic polydopamine carbon dots with dual-targeting effect alleviate motor and non-motor symptoms of Parkinson's disease via anti-neuroinflammation. Int J Biol Macromol. 2025;296:139724
108. Zheng M, Ruan S, Liu S, Sun T, Qu D, Zhao H. et al. Self-Targeting Fluorescent Carbon Dots for Diagnosis of Brain Cancer Cells. ACS Nano. 2015;9:11455-61
109. Sanadgol N, Abedi M, Hashemzaei M, Kamran Z, Khalseh R, Beyer C. et al. Exosomes as nanocarriers for brain-targeted delivery of therapeutic nucleic acids: advances and challenges. J Nanobiotechnol. 2025;23:453
110. Jia QY, Zhao ZY, Liang K, Nan FC, Li Y, Wang J. et al. Recent advances and prospects of carbon dots in cancer nanotheranostics. Mater Chem Front. 2020;4:449-71
111. Ji Z, Ai P, Shao C, Wang T, Yan C, Ye L. et al. Manganese-Doped Carbon Dots for Magnetic Resonance/Optical Dual-Modal Imaging of Tiny Brain Glioma. ACS Biomater Sci Eng. 2018;4:2089-94
112. Molaei MJ. Gadolinium-doped fluorescent carbon quantum dots as MRI contrast agents and fluorescent probes. Sci Rep. 2022;12:17681
113. Du Y, Qian M, Li C, Jiang H, Yang Y, Huang R. Facile marriage of Gd(3+) to polymer-coated carbon nanodots with enhanced biocompatibility for targeted MR/fluorescence imaging of glioma. Int J Pharm. 2018;552:84-90
114. Molaei MJ. Carbon quantum dots and their biomedical and therapeutic applications: a review. RSC Adv. 2019;9:6460-81
115. Hettiarachchi SD, Graham RM, Mintz KJ, Zhou Y, Vanni S, Peng Z. et al. Triple conjugated carbon dots as a nano-drug delivery model for glioblastoma brain tumors. Nanoscale. 2019;11:6192-205
116. Li S, Amat D, Peng Z, Vanni S, Raskin S, De Angulo G. et al. Transferrin conjugated nontoxic carbon dots for doxorubicin delivery to target pediatric brain tumor cells. Nanoscale. 2016;8:16662-9
117. Zhang X, Yu Q, Zhou P, Yang S, Xia J, Deng T. et al. Blood-brain barrier penetrating carbon dots with intrinsic anti-inflammatory and drug-loading properties. Biomater Adv. 2022;139:212995
118. Zhang W, Kandel N, Zhou Y, Smith N, Ferreira BC, Perez M. et al. Drug delivery of memantine with carbon dots for Alzheimer's disease: blood-brain barrier penetration and inhibition of tau aggregation. J Colloid Interface Sci. 2022;617:20-31
119. Zhang W, Chen J, Gu J, Bartoli M, Domena JB, Zhou Y. et al. Nano-carrier for gene delivery and bioimaging based on pentaetheylenehexamine modified carbon dots. J Colloid Interface Sci. 2023;639:180-92
120. Huang YC, Lai JZ, Luo CL, Chuang CC, Lin TC, Wang PH. et al. A Fluorescent Vector of Carbon Dot to Deliver Rab13 and Rab14 Plasmids for Promoting Neurite Outgrowth. ACS Appl Bio Mater. 2023;6:3739-49
121. Qian M, Du Y, Wang S, Li C, Jiang H, Shi W. et al. Highly Crystalline Multicolor Carbon Nanodots for Dual-Modal Imaging-Guided Photothermal Therapy of Glioma. ACS Appl Mater Interfaces. 2018;10:4031-40
122. Li Y, Zheng X, Zhang X, Liu S, Pei Q, Zheng M. et al. Porphyrin-Based Carbon Dots for Photodynamic Therapy of Hepatoma. Adv Healthc Mater. 2017;6:1600924
123. Shao X, Li M, Yan C, Wang C, Wang X, Guan P. et al. Photocatalytic, photothermal, and blood-brain barrier-permeable carbon nanodots: A potent multifunctional scavenger for beta-amyloid plaque. Colloids Surf, B. 2025;246:114380
124. Yan C, Wang C, Shao X, Teng Y, Chen P, Hu X. et al. Multifunctional Carbon-Dot-Photosensitizer Nanoassemblies for Inhibiting Amyloid Aggregates, Suppressing Microbial Infection, and Overcoming the Blood-Brain Barrier. ACS Appl Mater Interfaces. 2022;14:47432-44
125. Guo Q-L, Dai X-L, Yin M-Y, Cheng H-W, Qian H-S, Wang H. et al. Nanosensitizers for sonodynamic therapy for glioblastoma multiforme: current progress and future perspectives. Mil Med Res. 2022;9:26
126. Cheng M, Liu Y, You Q, Lei Z, Ji J, Zhang F. et al. Metal-Doping Strategy for Carbon-Based Sonosensitizer in Sonodynamic Therapy of Glioblastoma. Adv Sci. 2024;11:2404230
127. Ren X, Shi Y, Yang Y, Liu Z. Integrin αvβ3-targeted engineered carbon dots for efficacious sonodynamic therapy and fluorescence navigation surgery against gliomas. Mater Chem Front. 2024;8:2511-24
128. Nie R, Zhang J, Jia Q, Li Y, Tao W, Qin G. et al. Structurally Oriented Carbon Dots as ROS Nanomodulators for Dynamic Chronic Inflammation and Infection Elimination. ACS Nano. 2024;18:22055-70
129. Fan T, Cao X, Wang C, Shao X, Wang X, Guan P. et al. Vitamin C derived carbon dots: inhibiting amyloid aggregation and scavenging reactive oxygen species. New J Chem. 2025;49:605-14
130. Jia H, Gong J, Hu Z, Wen T, Li C, Chen Y. et al. Antioxidant Carbon Dots Nanozymes Alleviate Stress-induced Depression by Modulating Gut Microbiota. Langmuir. 2024;40:19739-50
131. Yang M, Zhao Z, Deng Z, Sun S, Guo Y, Li Z. et al. <i>Panax notoginseng</i>-Derived Carbon Dots Herbzymes Ameliorate Renal Ischemia-Reperfusion Injury via Anti-Inflammation, Antioxidation and Gut-Kidney Axis Regulation. Theranostics. 2026;16:4019-41
132. Tabish M, Malik I, Alshahrani AM, Afzal M. Detection of an oxidative stress metabolite associated with neurodegenerative diseases: effect of heteroatom doped antioxidant carbon dots. RSC Adv. 2025;15:8354-66
133. Hu Y, Cui J, Sun J, Liu X, Gao S, Mei X. et al. A novel biomimetic nanovesicle containing caffeic acid-coupled carbon quantum dots for the the treatment of Alzheimer's disease via nasal administration. J Nanobiotechnol. 2024;22:642
134. Jin Y, Zhang Q, Qin X, Liu Z, Li Z, Zhong X. et al. Carbon dots derived from folic acid attenuates osteoarthritis by protecting chondrocytes through NF-kappaB/MAPK pathway and reprogramming macrophages. J Nanobiotechnol. 2022;20:469
135. Xiao M, Pan Y, Tang S, Guo T, Yang L, Chen G. Carbon quantum dots amplify beneficial effects of EGCG against neural injuries by NLRP3 inflammasome after intracerebral hemorrhage. Int J Pharm. 2025;671:125281
136. Zhang XL, Gu YY, Liu YC, Cai YT, Sun H, Liu YX. et al. Near-infrared enhanced organic Se-doped carbon nitride quantum dots nanozymes as SOD/CAT mimics for anti-Parkinson via ROS-NF-κB-NLRP3 inflammasome axis. Chem Eng J. 2024;499:156028
137. Yang X, Tang X, Jia G, Wang Y, Yang L, Li Y. et al. Multifunctional Carbon Quantum Dots: Iron Clearance and Antioxidation for Neuroprotection in Intracerebral Hemorrhage Mice. ACS Appl Mater Interfaces. 2023;15:56820-33
138. Wang Y, Yan C, Qi Y, Zhao R, Wan Y, Lyu Z. et al. Harnessing multi-target nanosweepers inhibiting β-amyloid aggregation, scavenging reactive oxygen species and overcoming the blood brain barrier for rescuing Alzheimer's disease. Chem Eng J. 2025;508:161138
139. Boakye-Yiadom KO, Kesse S, Opoku-Damoah Y, Filli MS, Aquib M, Joelle MMB. et al. Carbon dots: Applications in bioimaging and theranostics. Int J Pharm. 2019;564:308-17
140. Xu G, Bao X, Chen J, Zhang B, Li D, Zhou D. et al. In Vivo Tumor Photoacoustic Imaging and Photothermal Therapy Based on Supra-(Carbon Nanodots). Adv Healthc Mater. 2019;8:e1800995
141. Barhum H, McDonnell C, Peltek O, Jain R, Amer M, Kain D. et al. In-Brain Multiphoton Imaging of Vaterite Cargoes Loaded with Carbon Dots. Nano Lett. 2024;24:8232-9
142. Yang S, Chen Z, Zhou P, Xia J, Deng T, Yu C. Carbon dots based on endogenous nutrients with visible and NIR fluorescence to penetrate blood-brain barrier. Carbon. 2023;202:130-40
143. Mauro N, Cillari R, Gagliardo C, Utzeri MA, Marrale M, Cavallaro G. Gadolinium-Doped Carbon Nanodots as Potential Anticancer Tools for Multimodal Image-Guided Photothermal Therapy and Tumor Monitoring. ACS Appl Nano Mater. 2023;6:17206-17
144. Jiang X, Wang J, Gan L, Wu Z, Wu T, Li F. et al. Carbon dot-based treatment for bacterial pneumonia by promoting a PI3K-mediated M1 polarization of macrophages. J Nanobiotechnol. 2025;23:315
145. Tan Q, Zhao S, Xu T, Wang Q, Lan M, Yan L. et al. Getting drugs to the brain: Advances and prospects of organic nanoparticle delivery systems for assisting drugs to cross the blood-brain barrier. J Mater Chem B. 2022;10:9314-33
146. Awad R, Avital A, Sosnik A. Polymeric nanocarriers for nose-to-brain drug delivery in neurodegenerative diseases and neurodevelopmental disorders. Acta Pharm Sin B. 2023;13:1866-86
147. Beach MA, Nayanathara U, Gao Y, Zhang C, Xiong Y, Wang Y. et al. Polymeric Nanoparticles for Drug Delivery. Chem Rev. 2024;124:5505-616
148. Razavi ZS, Razavi FS, Alizadeh SS. Inorganic nanoparticles and blood-brain barrier modulation: Advancing targeted neurological therapies. Eur J Med Chem. 2025;287:117357
149. Li C, Qin S, Wen Y, Zhao W, Huang Y, Liu J. Overcoming the blood-brain barrier: Exosomes as theranostic nanocarriers for precision neuroimaging. J Control Release. 2022;349:902-16
150. Sun M, Qin F, Bu Q, Zhao Y, Yang X, Zhang D. et al. Exosome-Based Therapeutics: A Natural Solution to Overcoming the Blood-Brain Barrier in Neurodegenerative Diseases. MedComm. 2025;6:e70386
151. Ruan S, Qian J, Shen S, Chen J, Zhu J, Jiang X. et al. Fluorescent carbonaceous nanodots for noninvasive glioma imaging after angiopep-2 decoration. Bioconjug Chem. 2014;25:2252-9
152. Tok K, Barlas FB, Bayir E, Senisik AM, Zihnioglu F, Timur S. One step synthesis of tryptophan-isatin carbon nano dots and bio-applications as multifunctional nanoplatforms. Colloids Surf, B. 2025;249:114533
153. Chung YJ, Lee CH, Lim J, Jang J, Kang H, Park CB. Photomodulating Carbon Dots for Spatiotemporal Suppression of Alzheimer's beta-Amyloid Aggregation. ACS Nano. 2020;14:16973-83
154. Yang M, Wei X, Pan K, Zhou Z, Liu Y, Lv X. et al. Brain-targeted ginkgolide B-modified carbonized polymer dots for alleviating cerebral ischemia reperfusion injury. Biomater Sci. 2023;11:3998-4008
155. Zhang J, Yang X, Wang S, Dong J, Zhang M, Zhang M. et al. Metformin carbon dots enhance neurogenesis and neuroprotection in Alzheimer's disease: A potential nanomedicine approach. Mater Today Bio. 2024;29:101347
156. Yan CR, Wang YX, Ma YD, Liu H, Tang S, Li YS. et al. A multifunctional carbon quantum dot overcoming the BBB for modulating amyloid aggregation and scavenging reactive oxygen species. Mater Today Chem. 2024;40:102222
157. Wei Z, Dong X, Sun Y. Quercetin-derived red emission carbon dots: A multifunctional theranostic nano-agent against Alzheimer's beta-amyloid fibrillogenesis. Colloids Surf, B. 2024;238:113907
158. Zhang Y, Wang S, Lu F, Zhang M, Kong H, Cheng J. et al. The neuroprotective effect of pretreatment with carbon dots from Crinis Carbonisatus (carbonized human hair) against cerebral ischemia reperfusion injury. J Nanobiotechnol. 2021;19:257
159. Lewis S. Amyloid clearance. Nat Rev Neurosci. 2017;18:455 -
160. Wang W, Liu Z, Cheng H, Xu M, Du Z, Liu W. et al. Cerium-doped carbon dots as dual-target agents against Alzheimer's β-amyloid fibrillogenesis and reactive oxygen species. Colloids Surf, B. 2025;252:114655
161. Chung YJ, Lee BI, Park CB. Multifunctional carbon dots as a therapeutic nanoagent for modulating Cu(ii)-mediated β-amyloid aggregation. Nanoscale. 2019;11:6297-306
162. Le W-d, Rowe D, Xie W, Ortiz I, He Y, Appel SH. Microglial Activation and Dopaminergic Cell Injury: An <em>In Vitro</em> Model Relevant to Parkinson's Disease. J Neurosci. 2001;21:8447-55
163. Wang H, Zhang M, Chu D, Huang X, Shi Y, Zhao Y. et al. Targeted reversal of iron deposition by highly active manganese-doped carbon dots in MRI-guided treatment of Parkinson's disease. Carbon. 2025;237:120152
164. Uyar R. Glioblastoma microenvironment: The stromal interactions. Pathol Res Pract. 2022;232:153813
165. Yan H, Miao H, Hu J, Pan J, Ge M, Yao J. et al. Oxidative stress induced paclitaxel-derived carbon dots inhibit glioblastoma proliferation and EMT process. J Nanobiotechnol. 2025;23:310
166. Obukohwo OM, Oreoluwa OA, Andrew UO, Williams UE. Microglia-mediated neuroinflammation in traumatic brain injury: a review. Mol Biol Rep. 2024;51:1073
167. Ouyang L, Mu X, Wang J, Li Q, Gao Y, Liu H. et al. Carbon dot targeting to nitrogen signaling molecules for inhibiting neuronal death. J Mater Chem B. 2020;8:2321-30
168. Luo W, Zhang L, Li X, Zheng J, Chen Q, Yang Z. et al. Green functional carbon dots derived from herbal medicine ameliorate blood—brain barrier permeability following traumatic brain injury. Nano Res. 2022;15:9274-85
169. Khan S, Khan MM, Badruddeen, Ahmad U, Akhtar W, Islam A. Exploring NMDAR pathways in ischemic stroke: implications for neurotoxic and neuroprotective mechanisms and therapeutic strategies. Naunyn-Schmiedeberg's Arch Pharmacol. 2025
170. Liu W, Liu L, Li H, Xie Y, Bai J, Guan J. et al. Targeted pathophysiological treatment of ischemic stroke using nanoparticle-based drug delivery system. J Nanobiotechnol. 2024;22:499
171. Lerner RP, Bimpisidis Z, Agorastos S, Scherrer S, Dewey SL, Cenci MA. et al. Dissociation of metabolic and hemodynamic levodopa responses in the 6-hydroxydopamine rat model. Neurobiol Dis. 2016;96:31-7
172. Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S. et al. Requirement of Hippocampal Neurogenesis for the Behavioral Effects of Antidepressants. Science. 2003;301:805-9
173. Qi J, Zhang R, Liu X, Liu Y, Zhang Q, Cheng H. et al. Carbon Dots as Advanced Drug-Delivery Nanoplatforms for Antiinflammatory, Antibacterial, and Anticancer Applications: A Review. ACS Appl Nano Mater. 2023;6:9071-84
174. Soo Choi H, Liu W, Misra P, Tanaka E, Zimmer JP, Itty Ipe B. et al. Renal clearance of quantum dots. Nat Biotechnol. 2007;25:1165-70
175. Shi Y, Xia Y, Zhou M, Wang Y, Bao J, Zhang Y. et al. Facile synthesis of Gd/Ru-doped fluorescent carbon dots for fluorescent/MR bimodal imaging and tumor therapy. J Nanobiotechnol. 2024;22:88
Author contact
Corresponding author: mgpangac.kr (Myung-Geol Pang).
 Global reach, higher impact
Global reach, higher impact