Impact Factor
Theranostics 2026; 16(11):6285-6314. doi:10.7150/thno.133908 This issue Cite
Review
Mechanomedicine in digestive surgery: a theranostic framework integrating mechanical diagnostics and therapeutic modulation across the perioperative continuum
1. Department of Gastrointestinal Surgery, Hainan General Hospital, Hainan Affiliated Hospital of Hainan Medical University, Haikou 570311, Hainan, P.R. China.
2. Department of Gastroenterology, Hainan General Hospital, Hainan Affiliated Hospital of Hainan Medical University, Haikou 570311, Hainan, P.R. China.
3. Key Laboratory of Biomedical Information Engineering of Ministry of Education, School of Life Science and Technology, Xi'an Jiaotong University, Xi'an 710049, Shaanxi, P.R. China.
4. Bioinspired Engineering and Biomechanics Center (BEBC), School of Life Science and Technology, Xi'an Jiaotong University, Xi'an 710049, Shaanxi, P.R. China.
5. Hainan General Hospital, Hainan Affiliated Hospital of Hainan Medical University, Haikou 570311, Hainan, P.R. China.
*These authors contributed equally to this work.
Received 2026-3-4; Accepted 2026-3-28; Published 2026-4-23
Abstract
Digestive surgery remains burdened by anastomotic leakage, postoperative obstruction, and highly variable functional recovery, even when operations are anatomically successful. Accumulating evidence indicates that these complications arise not only from morphology, but also from an unmeasured and unmodulated mechanical microenvironment, including tissue stiffness, anastomotic tension, perfusion-related shear stress, and intra-abdominal pressure. This review outlines a mechanomedicine framework for digestive surgery that treats these mechanical cues as actionable theranostic variables across the perioperative continuum, integrating mechanical diagnostics with targeted mechanical therapeutics. Multiscale biomechanics and mechanotransduction are first considered in relation to healing and fibrosis in the gastrointestinal tract. Preoperative mechanical profiling, using in vivo diagnostic techniques such as magnetic resonance elastography (MRE) and ultrasound shear-wave elastography (SWE), may refine surgical indications and support virtual surgical planning. Intraoperative mechanosensing tools, including functional lumen imaging, quantitative fluorescence perfusion, and portal pressure measurements, may provide real-time diagnostic thresholds to guide anastomotic site selection, reconstruction strategy, and resection extent. In vitro diagnostic platforms, including organ-on-chip models and mechanosensitive biosensors, have further clarified the mechanobiological basis of surgical complications and are informing the design of mechanoresponsive therapeutic systems. Postoperative mechanotherapy, encompassing continuous intra-abdominal pressure monitoring, mechanically tuned scaffolds, early mobilization, and mechanoresponsive drug-delivery systems, may contribute to early warning and personalized rehabilitation. Ethical and equity considerations are also relevant to this framework, including algorithmic bias, data governance, and equitable access across diverse healthcare settings. Together, these advances support a theranostic paradigm in which the same mechanical signals that diagnose risk may also help trigger, guide, or personalize therapy, pointing to a future in which digestive surgery is planned, performed, and followed up with more explicit and integrated control of the mechanical microenvironment to reduce complications and improve long-term function.
Keywords: digestive surgery, anastomotic leakage, mechanical microenvironment, mechanical imaging, theranostics
1. Introduction
Digestive surgery, encompassing a spectrum of procedures from routine colorectal resections to complex pancreaticoduodenectomies, serves as the cornerstone of curative treatment for a myriad of gastrointestinal malignancies and benign disorders [1-3]. Over the past century, the field has witnessed a remarkable evolution in technical sophistication, transitioning from open laparotomy to minimally invasive laparoscopy and, more recently, to robotic-assisted platforms [4-6]. These advancements have undoubtedly improved visualization and reduced surgical trauma. However, despite these technological leaps and the achievement of what is often described as “anatomical perfection” during reconstruction, the clinical community remains burdened by a persistent rate of severe postoperative complications, such as anastomotic leaks, postoperative bowel obstructions (ileus), and unpredictable recovery trajectories [7, 8]. Anastomotic leak remains one of the most feared complications, and is associated with sepsis, reoperations, and even mortality [1-3]. Conventionally, the etiology of these complications has been viewed through a morphological or biological lens, attributing failure to poor blood supply, bacterial contamination, or systemic comorbidities [9, 10]. While valid, this perspective overlooks a fundamental physical reality: tissues are mechanical structures subject to physical forces [11-13]. Accumulating evidence from the intersection of biomechanics and mechanobiology suggests that many of these complications arise not from anatomical errors, but from an unmeasured and unmodulated mechanical microenvironment such as excessive local tension, shear stress at the anastomosis, and tissue compliance mismatches [14, 15].
Historically, surgical planning and intraoperative decision-making have been guided almost entirely by morphology (visual inspection and static anatomy) and such an approach overlooks the dynamic mechanical microenvironment of healing tissues, which can be a decisive determinant of outcomes [16, 17]. In short, surgeons have historically assessed “how things look” but not “how things feel”, and this could predispose to suboptimal results. For example, excessive anastomotic tension or shear forces can directly compromise healing, contributing to leak formation [11]. Importantly, patients exhibit substantial inter-individual differences in tissue mechanical properties and responses, which a one-size-fits-all surgical approach fails to account for [18]. Two patients may show similar anatomy on computed tomography (CT), yet differ markedly in tissue stiffness, fragility, or mechanical adaptability because of fibrosis, aging, or prior treatment. Such differences can explain why outcomes vary widely and why traditional risk models based purely on anatomy or basic vitals remain imperfect [19-21]. By integrating patient-specific mechanical data (e.g., measuring a patient's liver or bowel stiffness before surgery), surgeons can begin to personalize their strategies [22]. Emerging studies suggest that using mechanics-guided planning, such as finite element modeling of anastomotic sites, can help tailor resection technique and tension management to the individual, thereby reducing complications. Mechanomedicine adds a functional, patient-specific dimension to precision surgery that morphology alone cannot provide [22, 23].
From a biophysical perspective, a surgical operation can be understood as a mechanical perturbation of tissues, after which the body enters a phase of force-driven healing and remodeling [24]. Tissues are not static after surgery, as they respond to mechanical loads and gradually evolve toward a new equilibrium state as healing progresses [12, 13]. In essence, wound healing in the gut is a dynamic biomechanical process: the surgical injury triggers a cascade where cells sense and respond to changes in mechanical cues in their microenvironment (e.g., matrix stiffness, stretch, and pressure) over time [25-27]. This process may also be interpreted within a non-equilibrium thermodynamic framework, in which surgery drives tissue away from its baseline state and healing gradually restores mechanical and structural stability [28, 29]. Healing may be conceptualized as a process of progressive structural adaptation that reduces stress concentration and restores tissue continuity [28-30]. In practical terms, this means that mechanical cues (e.g., tension, pressure, and stiffness) continuously influence cellular behaviors during recovery, affecting scar formation and tissue function [11]. When the mechanical microenvironment is favorable, healing is more likely to proceed appropriately; when excessive tension, abnormal shear stress, or inadequate mechanical support persists, the risk of dehiscence, fibrosis, or functional failure increases.
The emerging field of mechanomedicine seeks to bridge this gap. Mechanomedicine is defined here as the clinical discipline that quantifies, interprets, and therapeutically modulates the mechanical microenvironment of living tissues to improve patient outcomes—distinct from mechanobiology (which studies how cells sense and respond to forces at the molecular and cellular level), biomechanics (which characterizes the physical properties and force distributions of biological structures), and bioengineering (which designs devices and materials for biological applications) [31-33]. By treating mechanical cues not as passive bystanders but as actionable theranostic variables, surgical care may be fundamentally refined. This review presents a comprehensive, integrated theranostic framework for mechanomedicine in digestive surgery across the perioperative continuum, in which mechanical diagnostics and mechanical therapeutics are conceived as two inseparable arms of a unified strategy. The discussion begins with fundamental biomechanical principles, including solid stress, fluid shear, tissue stiffness, and mechanotransduction, and their roles in regulating biological responses. Three perioperative phases are then examined: preoperative mechanodiagnostics, intraoperative mechanical monitoring, and postoperative mechanotherapy. Each section summarizes recent advances demonstrating the clinical value of mechanics-based diagnostic and therapeutic approaches. These developments can be integrated into a unified theranostic paradigm and may inform surgical decision-making and patient care. This framework is intended to synthesize current knowledge in surgical mechanomedicine and identify opportunities for future investigation and clinical translation. Figure 1 illustrates this integrated mechanomedicine theranostic framework across the perioperative continuum and highlights digestive surgery as a mechanically driven perturbation-healing process.
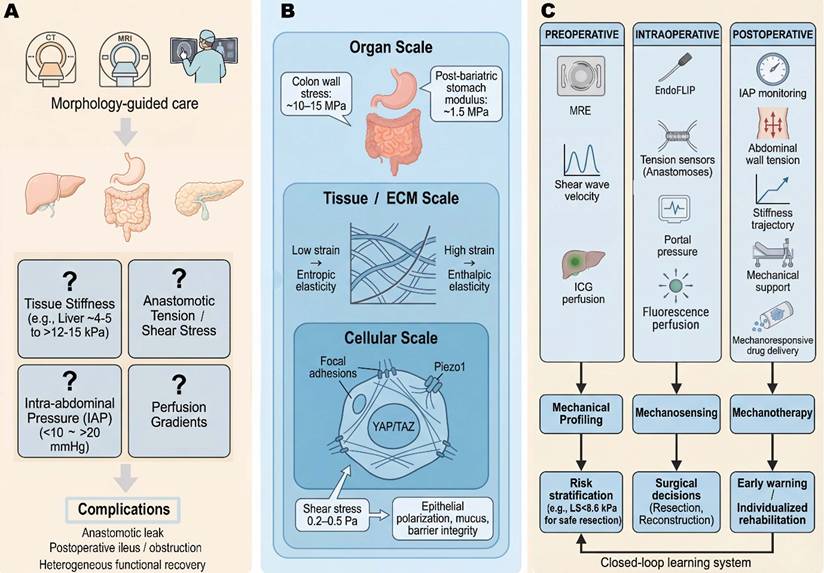
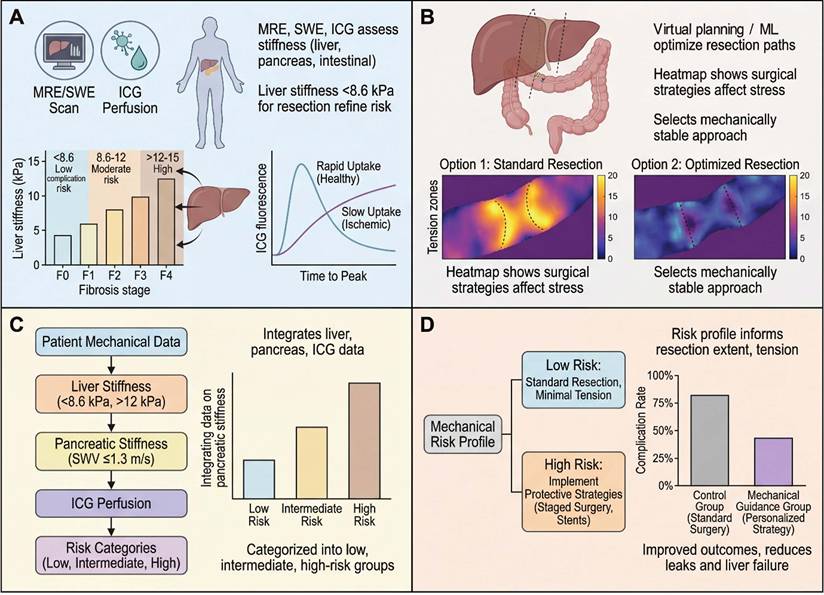
Digestive surgery as a mechanically driven perturbation-healing process and the concept of mechanomedicine across the perioperative continuum. (A) Digestive surgery complications emerge from a mechanical “blind spot” in morphology-guided care. Anastomotic leak, postoperative bowel obstruction/ileus and heterogeneous functional recovery remain common after digestive surgery, despite anatomically “perfect” operations [7, 8]. Conventional planning relies primarily on morphology (e.g., computed tomography (CT), magnetic resonance imaging (MRI) and intraoperative visual inspection), without explicit quantification of key mechanical variables. In reality, these complications reflect unmeasured gradients in tissue stiffness, anastomotic tension, shear stress and intra-abdominal pressure, which vary widely between patients (e.g., liver stiffness ranges from ~ 4-5 kPa in non-fibrotic to > 12-15 kPa in advanced fibrosis [18, 67, 68]; intra-abdominal pressure (IAP) can rise from <10 mmHg to > 20 mmHg in abdominal compartment syndrome) [114]. (B) Multi-scale mechanical microenvironment in gastrointestinal tissues. At the organ scale, digestive organs experience non-uniform stress and strain (e.g., colon wall tensile stress can reach ~ 10 - 15 MPa [11, 12]; post-bariatric stomach modulus can drop to ~ 1.5 MPa as elasticity is altered) [36]. At the tissue and extracellular matrix (ECM) scales, collagen and other fibers exhibit entropic elasticity at low strain and enthalpic elasticity at high strain, producing non-linear strain stiffening. Cells sense these changes via focal adhesion complexes, mechanosensitive ion channels (e.g., piezo type mechanosensitive ion channel component 1 (Piezo1)) and cytoskeletal tension, leading to nuclear translocation of Yes-associated protein/transcriptional coactivator with PDZ-binding motif (YAP (Yes-associated protein) / TAZ (transcriptional co-activator with PDZ-binding motif) and other mechano-effectors that reprogram gene expression [76, 79]. In organ-on-chip systems, physiological shear stress in the range of ~ 0.2-0.5 Pa promotes epithelial polarization, mucus secretion and barrier integrity, illustrating how subtle mechanical cues modulate cell fate [49]. (C) Integrated mechanomedicine framework across the perioperative continuum. In the proposed mechanomedicine paradigm, preoperative mechanical profiling (e.g., liver and pancreas stiffness by elastography, shear wave velocity, quantitative indocyanine green (ICG) perfusion) informs risk stratification and virtual surgical planning [19, 86]; intraoperative mechanosensing (for example, functional lumen imaging for distensibility index, tension sensors on anastomoses, portal pressure measurements, fluorescence perfusion curves) guides on-table decisions about resection extent, anastomotic site and reconstructive strategy [23, 88]; and postoperative mechanotherapy (e.g., intra-abdominal pressure (IAP) monitoring, abdominal wall tension tracking, stiffness trajectory of anastomoses, mechanical support and mechanoresponsive drug delivery) enables early warning and individualized rehabilitation [136]. Longitudinal mechanical data feed back into subsequent preoperative assessment, creating a closed-loop learning system. The figure was created by Figdraw (www.figdraw.com).
2. The biomechanical basis of digestive system health and disease
To effectively modulate the mechanical microenvironment, it is essential to first understand the fundamental biophysical principles that govern the digestive tract [12, 34]. Mechanomedicine is rooted in core biomechanical and mechanobiological concepts, such as solid stress, fluid shear stress, tissue stiffness, and mechanotransduction, that operate from the molecular scale to the whole organ level [11, 24, 35]. The key terms are defined as follows: stress refers to the internal force per unit area within a material (units: Pa or kPa); strain is the fractional deformation of a material in response to stress (dimensionless); stiffness (or elastic modulus, E) is the resistance of a material to deformation under applied force (units: kPa or MPa); compliance is the inverse of stiffness, describing how readily a structure deforms under load; tension is the pulling force transmitted along a structure (units: N or mN); and shear stress (τ) is the tangential force per unit area exerted by a fluid flowing over a surface (units: Pa). These definitions are used consistently throughout the manuscript [11, 12, 24, 34, 35].
2.1 Solid mechanics: stress, strain, and the imperative of tension control
In digestive surgery, solid stress refers to the internal mechanical forces transmitted through the tissue matrix [36]. These forces modulate tissue integrity and cellular behavior during both normal physiology and wound healing [14, 26, 37], and are particularly critical at the anastomotic site, where excessive tension is a primary driver of leakage [10, 23, 38].
The physics of anastomotic tension is governed by Laplace's law, which relates wall tension (T) in a cylindrical vessel to the intraluminal pressure (P) and radius (r) of the lumen:
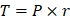 This relationship underscores the mechanical vulnerability of surgical anastomoses [11, 12, 36]. Postoperative bowel distension or obstruction increases r or P, linearly amplifying suture-line tension [39-42]. Healthy bowel typically withstands tensile stresses in the range of tens to hundreds of kPa, although local stress concentrations at suture or staple points may exceed the tissue's tolerance, causing micro-tears, ischemia, and dehiscence [2, 22, 23, 43-45].
This relationship underscores the mechanical vulnerability of surgical anastomoses [11, 12, 36]. Postoperative bowel distension or obstruction increases r or P, linearly amplifying suture-line tension [39-42]. Healthy bowel typically withstands tensile stresses in the range of tens to hundreds of kPa, although local stress concentrations at suture or staple points may exceed the tissue's tolerance, causing micro-tears, ischemia, and dehiscence [2, 22, 23, 43-45].
This principle is particularly relevant in pancreatic surgery, where suture-parenchyma interaction is a primary determinant of postoperative pancreatic fistula (POPF) [1, 3, 46]. Unlike the intestine's tough collagenous submucosa, the pancreas lacks a distinct capsule and is highly susceptible to solid stress [47]. In a soft pancreas, low yield strength predisposes to suture cut-through under tension, leading to anastomotic failure [19, 46, 48].
Stress shielding—complete removal of mechanical stress—leads to tissue atrophy, analogous to Wolff's law in bone [13, 24]. Gut tissues require physiological strain to maintain structural integrity [49-51]; the surgical goal is therefore not to eliminate tension but to maintain it within an optimal range that stimulates healing without causing overstress [26, 40, 52, 53].
2.2 Fluid mechanics: the regulatory role of shear stress
Fluid shear stress (τ) is the frictional force exerted by a fluid flowing over a surface. In the digestive system, shear stress plays a significant role in vascular perfusion and luminal epithelium function, both of which are critical to healing after surgery [42, 49, 50].
Vascular shear stress and perfusion: Blood flow generates shear stress on endothelial cells that line the blood vessels [54, 55]. Surgical interventions such as mesenteric torsion or the creation of stenotic anastomoses can induce complex, oscillatory shear stress patterns that influence endothelial function [54, 56, 57]. Physiological shear stress promotes endothelial health by stimulating the production of vasodilators such as nitric oxide (NO), and it helps maintain an anti-thrombotic surface [37, 49]. However, pathological shear stress—low-magnitude or oscillatory, often caused by low flow or anatomical kinking—triggers endothelial dysfunction, pro-inflammatory signaling (e.g., NF-κB), and a pro-thrombotic state, collectively compromising anastomotic perfusion [25, 58-60].
Luminal shear stress and barrier function: Shear stress from chyme flow in the intestinal lumen also regulates epithelial function [42, 50]. In vitro organ-on-chip studies suggest that physiological shear stress in the range of approximately 0.2-0.5 Pa may promote mucus secretion and epithelial barrier formation, although the direct translation of these values to in vivo conditions remains to be established [37, 50]. These microfluidic systems serve a dual role: they recapitulate the gut mechanobiological environment to diagnose the consequences of abnormal shear stress, and simultaneously serve as platforms for testing mechanoresponsive therapeutic interventions—exemplifying the in vitro theranostic approach.
2.3 Tissue stiffness and viscoelasticity: the material properties of healing
Tissue stiffness refers to a material's resistance to deformation under an applied force, and it is a key determinant of tissue integrity during healing [61]. However, biological tissues, including those of the digestive tract, are not simple linear elastic materials but exhibit viscoelastic behavior, meaning they show both elastic (solid-like) and viscous (fluid-like) properties when subjected to deformation [34, 44, 62].
Non-linear strain stiffening: The extracellular matrix (ECM) of the gut, composed largely of collagen and elastin fibers, exhibits non-linear strain stiffening [34, 63]. At low strain, these fibers are crimped and disordered, offering little resistance to stretching. As strain increases, the fibers straighten and the tissue becomes rapidly stiffer [44, 64]. This is a protective mechanism that prevents over-distension. However, from a surgical perspective, this rapid stiffening can be problematic. As a suture is tightened, the tissue initially exhibits compliant behavior. However, with only a slight additional increase in tension, the tissue transitions into a high-stiffness regime, where stress rises nonlinearly. This abrupt increase in stiffness can elevate the risk of suture cut-through, particularly under certain loading conditions [43, 45].
Viscoelastic hysteresis: Tissues exhibit time-dependent behaviors—stress relaxation (decreasing stress at fixed length) and creep (gradual elongation under constant load)—that directly affect anastomotic stability [30, 38, 64]. A suture line that feels secure at placement may loosen over hours due to these effects, compromising the seal [65, 66].
Pathological stiffness in disease: Fibrosis dramatically alters tissue stiffness. Cirrhotic livers exceed 12-15 kPa, altering handling characteristics and increasing fracture risk during suturing [21, 67, 68]; fibrotic intestinal strictures similarly affect surgical handling and fibroblast behavior [56, 69]. In the pancreas, stiffness is a critical biomarker: a soft pancreas (low stiffness) is the most significant risk factor for pancreatic fistula [19, 46], while chronic pancreatitis and pancreatic ductal adenocarcinoma produce a dense desmoplastic stroma with markedly elevated stiffness [70].
2.4 Mechanotransduction: the cellular interpreters of force
Mechanotransduction refers to the process by which cells convert mechanical forces into biochemical signals, enabling them to adapt to their mechanical environment [14, 58]. This process plays a crucial role in wound healing, fibrosis, and cancer progression in the digestive tract [15, 18, 71].
The integrin-cytoskeleton linkage: Integrins are transmembrane receptors that link the ECM to the cytoskeleton [15, 72]. When cells encounter a stiff matrix, such as in fibrotic liver or scar tissue, integrins cluster and form focal adhesions, which recruit signaling proteins like Focal Adhesion Kinase (FAK) and Rho-associated kinase (ROCK) [72, 73]. This activation increases cytoskeletal contractility, driving fibroblast differentiation into myofibroblasts, which are responsible for wound closure but also for pathological scarring and stricture formation [18, 71, 74]. In the pancreas, this mechanism is driven by pancreatic stellate cells (PSCs). Upon exposure to a stiff mechanical environment or high fluid pressure, quiescent PSCs transform into an activated phenotype, synthesizing excessive ECM and perpetuating pancreatic fibrosis [47]. Thus, a stiff mechanical environment promotes a cellular phenotype that exacerbates stiffness, creating a positive feedback loop of fibrosis [18, 75].
YAP/TAZ signaling: YAP and TAZ translocate to the nucleus on stiff substrates or under high tension, acting as transcriptional co-activators that promote proliferation and cell survival [58, 76-78]. Chronic activation by sustained mechanical stress drives fibrosis and cancer progression; notably, YAP-mediated suppression of the cGAS-STING pathway in macrophages dampens immune responses in both tumor progression and postoperative healing [14, 15].
Mechanosensitive ion channels: Piezo1 and TRPV4 open in response to mechanical deformation, allowing calcium influx that triggers downstream intracellular signaling cascades [52, 79, 80]. In the gut epithelium, activation of Piezo1 by physiological shear stress is essential for maintaining barrier integrity and promoting mucus secretion [49, 52]. Conversely, excessive strain or high stiffness can lead to altered ion channel activity, triggering inflammatory responses in macrophages and potentially driving pathological tissue responses in the form of fibrosis or dysregulated healing [52, 81, 82].
In summary, the biomechanical principles reviewed here—solid stress, fluid shear, tissue stiffness, and mechanotransduction—are not merely theoretical constructs but have the potential to serve as clinically relevant biomarkers and therapeutic targets: the same mechanical signals that indicate pathological states also serve as targets for intervention [35, 83, 84]. Incorporating these principles into routine clinical practice, as both diagnostic biomarkers and therapeutic targets, is the foundation of mechanomedicine across the perioperative continuum [4, 85]. Figure 2 summarizes these multiscale mechanisms and their clinical implications.
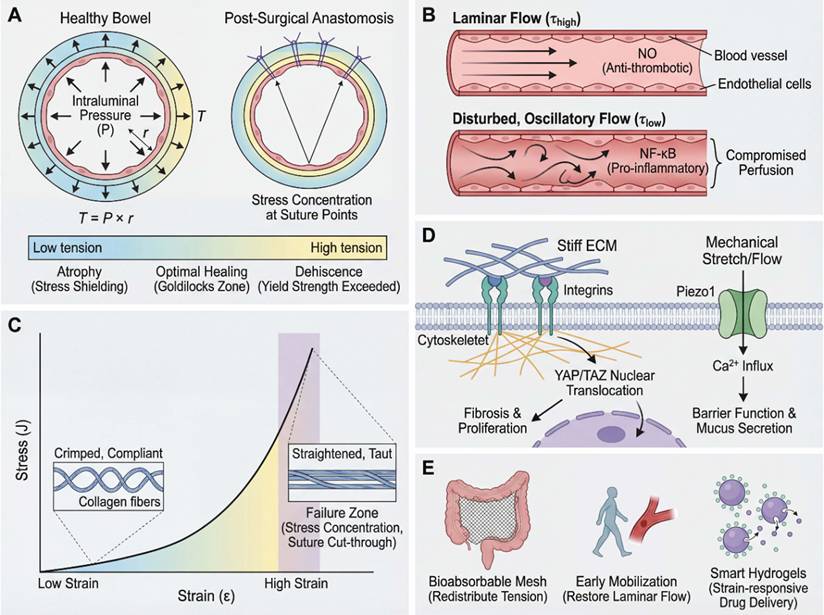
Multiscale biomechanical mechanisms in gastrointestinal health and surgical disease. (A) Solid stress and anastomotic tension (macro-mechanical). The mechanical integrity of the bowel wall is governed by the Law of Laplace (T = P × r), where wall tension (T) is proportional to intraluminal pressure (P) and radius (r) [11, 12]. In a healthy state, tension is distributed evenly. However, at the post-surgical anastomosis, increased pressure or dilation concentrates stress at suture points [22, 23]. Excessive tension exceeding the tissue's yield strength can lead to micro-tears, ischemia, and dehiscence [10, 43], whereas stress shielding may cause atrophy; optimal healing occurs within a mechanical “Goldilocks zone.” (B) Fluid shear stress and vascular perfusion (hemodynamic). Fluid mechanics play a regulatory role through shear stress (τ). Top: Laminar blood flow generates physiological shear stress, stimulating endothelial cells to produce nitric oxide (NO), maintaining a healthy, anti-thrombotic phenotype [49, 50]. Bottom: Pathological conditions (e.g., torsion or stenosis) cause disturbed, oscillatory flow. This low-magnitude shear activates pro-inflammatory pathways (e.g., NF-κB), leading to endothelial dysfunction and compromised perfusion at the healing site [41, 109, 110]. (C) Tissue stiffness and non-linear strain stiffening (material properties). Gastrointestinal tissues exhibit non-linear elasticity. At low strain, collagen fibers are crimped and compliant (bottom inset). As strain increases, fibers straighten and become taut (top inset), causing a rapid, exponential increase in stiffness (J-shaped curve) [34, 102]. Surgical tightening beyond this transition point risks entering the high-stiffness failure zone, where stress concentration leads to suture cut-through. (D) Mechanotransduction and cellular response (cellular interpreter). Cells convert mechanical cues into biochemical signals via two primary pathways. Path 1 (Stiffness Sensing): Integrins link the stiff extracellular matrix (ECM) to the cytoskeleton, triggering the nuclear translocation of YAP/TAZ, which promotes gene expression associated with fibrosis and proliferation [72, 77]. Path 2 (Stretch/Flow Sensing): Mechanical stretch or fluid flow activates mechanosensitive ion channels (e.g., piezo type mechanosensitive ion channel component 1 (Piezo1)), allowing Ca2+ influx to regulate barrier function and mucus secretion [25, 79]. (E) Clinical implications and mechanotherapy (translational solutions). Application of biomechanical principles to “Mechanomedicine.” Vignette 1: Structural modulation using bioabsorbable meshes to redistribute tension and prevent stress concentration [118, 135]. Vignette 2: Physiological modulation via early mobilization to restore laminar flow and perfusion [136, 138]. Vignette 3: Smart materials, such as strain-responsive hydrogels, that adapt to the mechanical environment for targeted drug delivery [129, 171]. The figure was created by Figdraw (www.figdraw.com).
3. Preoperative phase: mechanics-guided precision planning
Before the surgeon ever makes an incision, mechanomedicine can contribute valuable diagnostic information to risk stratification and surgical planning. Two major components are addressed: (1) mechanics-based diagnostics, encompassing both in vivo imaging modalities and in vitro diagnostic platforms, to assess tissue pathology in mechanical terms (e.g., stiffness, compliance), thereby refining surgical indications and strategies; and (2) mechanics-informed optimization of the surgical plan, using computational models or machine learning to simulate surgical scenarios and predict outcomes. This preoperative diagnostic phase is the first arm of the theranostic cycle: mechanical information gathered here directly informs and personalizes the therapeutic strategy.
3.1 Mechanical diagnostics for disease staging and surgical decision-making
MRE and ultrasound shear wave elastography (SWE) are well established in selected clinical contexts, particularly in hepatology, but their application in broader digestive surgical decision-making remains under active investigation [86, 87]. Unlike CT/MRI, which depict anatomical structure, elastography provides a functional map of tissue mechanical properties. This has important implications for disease staging and theranostic decision-making, since many pathological processes—fibrosis, inflammation, tumors—alter tissue stiffness before morphological changes become apparent [20, 21, 61, 67, 68]. These in vivo diagnostic readouts do not merely characterize disease; they directly guide therapeutic decisions, exemplifying the theranostic principle of using the same measurement to both diagnose and direct treatment.
One area with immediate clinical impact is chronic liver disease and surgical candidacy. For patients considered for liver resection (for tumor or other lesions), underlying liver fibrosis significantly affects the risk of postoperative liver failure. MRE and SWE can measure liver stiffness (LS) across the liver noninvasively [21, 68, 71]. Recent studies have defined stiffness thresholds that correlate with fibrosis stage and surgical outcomes. For example, in patients with hepatocellular carcinoma (HCC), using two-dimensional shear wave elastography (2D-SWE), LS below about 8-9 kPa has been identified as a risk-stratifying threshold in selected cohorts (minimal fibrosis), whereas LS above ~ 12-15 kPa suggests advanced fibrosis/cirrhosis where resection plans should be more cautious [21]. In one cohort, an LS < 8.6 kPa predicted low risk of post-hepatectomy complications (sensitivity ~ 89%, specificity ~ 74%), whereas patients with LS > 12 kPa had markedly higher complication rates and often benefited from adjusted surgical strategy (e.g., smaller resection or portal pressure management) [21, 88]. In essence, preoperative liver stiffness can serve as an objective risk indicator, analogous to ejection fraction in cardiology, and may support decisions regarding whether resection is likely to be tolerated safely. This represents a shift towards functional criteria for surgical indications, supplementing anatomical criteria like tumor size or location [21, 68, 71].
In pancreatic surgery, especially pancreatoduodenectomy, the texture of the pancreas is a well-known factor in outcomes. A soft, fatty pancreas is more prone to pancreaticojejunostomy leak (postoperative pancreatic fistula, POPF) than a firm, fibrotic pancreas [1, 19, 47]. Now, tools like endoscopic transient elastography and SWE allow quantification of pancreatic stiffness preoperatively as in vivo diagnostic biomarkers. A lower shear-wave velocity (SWV) or elasticity indicates a softer gland. A recent study confirmed that SWV ~ 1.3 m/s in the pancreas (a very low stiffness) was associated with significantly higher rates of clinically relevant POPF [19, 47]. In practical terms, if elastography reveals a very soft pancreas, the surgical team might need additional safeguards, for example, using a different anastomotic technique, placing a stent, or being prepared for rigorous drain management, to mitigate the anticipated risk of leak [19]. This exemplifies the theranostic use of in vivo mechanical diagnostics: the elastographic diagnosis of a soft pancreas directly prescribes a modified therapeutic strategy. On the other end, elastography can help in tumor characterization: malignant pancreatic tumors tend to be substantially stiffer than benign lesions. MRE studies have shown that pancreatic cancers often exhibit stiffness > 3.5 kPa, whereas healthy pancreas is much softer [70]. Multifrequency MRE (which probes tissue at different vibration frequencies to glean both stiffness and viscosity) has even been used to grade pancreatic neuroendocrine tumors, finding that higher-grade tumors have higher stiffness [70, 89]. This kind of information could assist in borderline cases, for instance, if a pancreatic mass has intermediate imaging features, a high stiffness on elastography might push towards resection, whereas a low stiffness might suggest a benign or less aggressive lesion that could be observed [70, 89]. While not yet standard of care and currently limited to specialized centers, these approaches are under active clinical investigation and hold promise for refining pancreatic surgical decision-making through integrated mechanodiagnostic-therapeutic protocols.
Another promising application is in colorectal liver metastases (CRLM). Beyond simply counting tumors and measuring their size, assessing the mechanical properties of metastases and the surrounding liver may predict how well a patient will respond to chemotherapy and how aggressive surgery should be [62, 90]. A study used SWE to monitor liver metastasis stiffness during chemotherapy and found that a decrease in stiffness by > 13% after treatment was an excellent predictor of tumor response and longer progression-free survival (area under the curve (AUC) ~ 0.84 for response) [90]. Essentially, if the metastasis softens with chemo, indicating tumor kill and less fibrotic stroma, the patient is likely responding well; if it remains stiff, the tumor might be chemoresistant. This could influence the timing of surgery, for instance, operating sooner on non-responders [90]. MRE has also been explored for CRLM characterization, with some evidence that mechanical phenotyping of tumors correlates with their viability and invasive potential. Similarly, in hepatocellular carcinoma, tumor peripheral stiffness has been shown to modulate chemoresistance via mechanotransduction pathways, demonstrating that mechanical properties directly influence therapeutic outcomes [75]. These are paradigmatic examples of theranostics in mechanomedicine: the same in vivo diagnostic measurement, namely tissue stiffness assessed by elastography, simultaneously characterizes disease state, predicts therapeutic response, and guides the selection and timing of treatment.
Even in scenarios like adhesive small-bowel obstruction (ASBO), mechanical diagnostics can help. Typically, when a patient has a partial bowel obstruction from adhesions, the decision to operate vs. continue conservative management can be tricky. One emerging tool is preoperative indocyanine green (ICG) fluorescence perfusion assessment, typically conducted during minimally invasive diagnostic laparoscopy [41, 60]. ICG can be injected and its near-infrared fluorescence tracked to see how well the bowel wall is perfused beyond an obstruction. Quantitative ICG imaging, analyzing fluorescence intensity curves, allows estimation of whether a segment is ischemic or likely to recover [60]. Recent reports indicate that delayed or weak ICG uptake in a distended bowel loop signals compromised perfusion and impending necrosis, meaning that surgical intervention is needed, whereas strong perfusion suggests the bowel might survive once decompressed [41, 60]. Additionally, preoperative SWE of dilated bowel has been piloted to gauge bowel wall stiffness; a very stiff loop may indicate edema and fibrosis from prolonged obstruction, again nudging toward surgery, whereas a more compliant loop might resolve [91]. These applications are still being refined, but they exemplify how mechanical and hemodynamic assessment is adding quantitative data to preoperative planning in emergency general surgery contexts.
Recent clinical studies suggest that mechanical readouts can influence surgical planning across stiffness, distensibility, and perfusion domains. In preoperative hepatobiliary patients, MR elastography-based liver stiffness alone identified severe fibrosis with an AUC of 0.85, whereas combining routine preoperative markers increased the AUC to 0.95, supporting the use of stiffness information to refine assessment of hepatic reserve before major resection [87]. In foregut surgery, EndoFLIP-derived distensibility, which serves as a practical surrogate of luminal tension, showed that Hill fundoplication created a tighter gastroesophageal junction than Toupet fundoplication, with lower distensibility index (0.9 ± 0.4 vs 1.3 ± 0.6 mm²/mmHg) and compliance (25.9 ± 12.8 vs 35.4 ± 13.4 mm3/mmHg), illustrating how quantitative tension-related measurements can help calibrate reconstruction rather than relying on visual judgment alone [92]. Likewise, in rectal cancer surgery, indocyanine green fluorescence angiography changed the initial surgical plan in 10.6% of patients undergoing low anterior resection, and no anastomotic leaks occurred in the subgroup in whom the proximal resection margin was modified, indicating that perfusion imaging can directly alter operative decision-making in real time [93]. Together, these data support the concept that patient-specific mechanical information may complement anatomic imaging when tailoring operative extent, reconstruction, and perioperative strategy.
In summary, the preoperative phase now has tools to measure mechanical parameters (e.g., stiffness, tension, perfusion) both in vivo and in vitro, and leveraging this information can improve surgical decision-making. In vivo diagnostic tools, including MRE, SWE, and quantitative ICG perfusion imaging, provide patient-specific mechanical profiles that directly inform therapeutic planning. Complementarily, in vitro diagnostic platforms, such as organ-on-chip systems and mechanosensitive biosensors, have established the mechanobiological thresholds that underpin clinical decision criteria. Figure 3 illustrates some of these technologies and their impact on planning. For instance, by knowing a priori that a patient's liver is extremely stiff or that their pancreas is soft, the surgical team can stratify risk, perhaps opting for a two-stage liver surgery or preparing pancreatic stents and octreotide to mitigate fistula risk. This preoperative theranostic loop, in which mechanical diagnosis directly prescribes mechanical therapy, is the foundation of precision mechanomedicine. Critically, the mechanical data gathered preoperatively—liver stiffness thresholds, anastomotic tension predictions, and perfusion maps—directly prescribe the intraoperative strategy: which anastomotic site to select, how much tension to tolerate, and which real-time monitoring tools to deploy in the operating room.
Preoperative mechanodiagnostics and virtual stratification of surgical risk in digestive surgery. (A) Liver stiffness thresholds for safe hepatectomy. Box-violin plots illustrate liver stiffness (LS) measured by magnetic resonance elastography (MRE) or shear-wave elastography (SWE) across fibrosis stages (F0-F1 to F4). Horizontal bands indicate risk-stratifying thresholds derived from recent prospective studies [67, 68, 86]: LS < 8.6 kPa is associated with low rates of post-hepatectomy complications and liver failure, LS 8.6-12 kPa indicates intermediate risk, and LS > 12-15 kPa reflects advanced fibrosis/cirrhosis, in which resection extent should be carefully restricted or staged [87]. Inset: complication rates increase stepwise across these strata, underscoring the value of stiffness-based selection. (B) Pancreatic stiffness and risk of postoperative pancreatic fistula (POPF). Preoperative shear-wave velocity (SWV) of the pancreatic parenchyma stratifies the risk of clinically relevant POPF after pancreatoduodenectomy [19]. Patients with SWV ≤ 1.3 m/s (soft pancreas) exhibit the highest POPF rates, whereas risk declines with increasing stiffness [46]. A receiver-operating characteristic (ROC) curve highlights the discriminative performance of a 1.3 m/s threshold (AUC ~ 0.75-0.80 in contemporary cohorts), providing a quantitative criterion for reinforcing reconstruction and drainage strategies [47]. (C) Mechanical phenotyping of colorectal liver metastases to predict chemotherapy response. Elastographic monitoring of liver metastasis stiffness during systemic therapy reveals that a relative reduction in shear-wave velocity of ≥13% after initial cycles is strongly associated with radiologic response and improved progression-free survival, with an ROC area under the curve ~ 0.84 for this threshold [90]. This demonstrates that tumor mechanophenotype is dynamic and prognostically informative [62]. (D) Intestinal perfusion and stiffness guide management of bowel obstruction. Representative indocyanine green (ICG) fluorescence time-intensity curves show rapid, high-amplitude perfusion in viable bowel versus delayed, blunted uptake in strangulated segments [41, 60]. In parallel, shear-wave elastography (SWE) can quantify intestinal stiffness, with values < 5 kPa suggestive of reversible obstruction and > 10 kPa raising concern for ischemia or chronic fibrosis [91]. Together, these preoperative mechanodiagnostic tools support more precise selection of candidates for resection, the extent of resection and the timing of surgery across liver, pancreas and intestine. The figure was created by Figdraw (www.figdraw.com).
3.2 Virtual surgical planning and simulation (mechanics-oriented optimization)
Beyond diagnostics, the preoperative period is increasingly benefiting from computational modeling and emerging artificial intelligence (AI) technologies to simulate surgeries in silico—approaches that remain largely translational but are advancing rapidly toward clinical integration. These methods allow surgeons to perform a virtual test run of different strategies on a patient-specific computational model, optimizing the plan and anticipating potential pitfalls; in the context of mechanomedicine, such simulations further incorporate patient-specific mechanical properties to predict how tissues will behave under surgical manipulation [63, 94-96].
Finite element modeling has become a cornerstone of biomechanics, and now it has been tailored to surgical planning. In the last couple of years, there have been notable successes in creating patient-specific FE models of organs, for example, modeling how a colon anastomosis will stretch and where stress will concentrate once constructed [17, 22, 94]. The typical workflow involves taking the patient's imaging (CT/MRI) to build a 3D geometry of the organ, assigning material properties (e.g., using elastography data for stiffness) [97-99], and then simulating surgical maneuvers like resecting a segment or suturing an anastomosis [63]. Machine learning, especially deep learning, is augmenting this by handling complex data integration and speeding up computations [95, 96]. Machine learning-driven models can analyze patterns from prior surgical cases (e.g., images, mechanical data, outcomes) to assist in predicting complications and guiding intraoperative decisions [84]. One example combined CT scans, elastography maps, and clinical variables to train an AI that outputs risk probabilities for anastomotic leak. These models achieved areas under the receiver operating characteristic (ROC) curve in the range of 0.75 - 0.85 in test cohorts [95, 96]. In other words, by inputting a new patient's CT, stiffness, and lab data, the model might say “this patient has an 80% predicted probability of leak with a standard procedure.” Armed with that, the team can modify the plan, perhaps opting for a diverting stoma or reinforcing the anastomosis before proceeding [95, 96].
One concrete application is optimal anastomosis site selection in colorectal surgery. In Crohn's disease requiring resection of a long inflamed bowel segment, selection of the optimal margins remains challenging. Traditionally, it is by gross appearance [100]. A computational model that maps strain distribution along the bowel under peristaltic load could identify regions of abnormal stiffness, if included in the anastomosis, would create stress concentrations. The surgeon could adjust the resection line to avoid that region [61, 95]. Virtual surgery systems have been tested where different anastomotic configurations are simulated, and the one with the lowest peak stress is recommended [22, 35, 94]. Likewise, in liver surgery, models can estimate the post-resection liver remnant shear stress and venous pressure. If a virtual right hepatectomy shows dangerously high stresses in the remnant, which predicts congestion or failure, the plan can change to a smaller resection or staged approach [57, 101].
Another emerging use is “digital twins” for surgery: creating detailed patient-specific computational models that integrate individual anatomy and tissue mechanical properties [97, 98]. Such models have already been successfully applied in digestive surgery to characterize patient-specific gastric biomechanics and predict outcomes of different bariatric procedures [36, 102]. Recent advances in breast surgery have further proven the clinical feasibility of rapid patient-specific finite element simulation pipelines using preoperative imaging [97]. Extending these validated methodologies to broader gastrointestinal applications is a natural and highly promising next step [94].
Crucially, these simulations are becoming more data-driven and validated. It is not just theoretical: comparative studies have shown that FE-predicted anastomotic tension correlates with intraoperative sensor measurements [38]. As computing power and algorithm sophistication grow, real-time or near-real-time simulation is on the horizon [38, 94]. Research has proposed an “intraoperative digital twin” that acts like a surgical GPS. It is continuously updated by sensor data during an operation, thereby offering real-time guidance to the surgeon [83, 103]. Although intraoperative digital-twin systems are still experimental, simpler data-driven applications have already entered clinical use. Preoperative MRE, for instance, provides a noninvasive assessment of liver stiffness and has shown value in perioperative risk stratification, including predicting postoperative recurrence in hepatocellular carcinoma and liver-related outcomes in chronic liver disease [86, 104].
Finally, on the AI front, deep neural networks—still at the translational research stage—have been trained on large datasets of past surgeries to detect patterns a human might miss. Related studies used a deep learning model to predict anastomotic leak in colorectal surgery patients. The model incorporated variables such as anastomosis level, patient comorbidities, and notably, intraoperative tissue quality assessments, which were based on the surgeon's grading of tissue frailty [35, 105]. The model achieved an AUC ~ 0.80 and was better than traditional risk scores [106]. If such models were augmented with actual mechanical measurements, such as elastography or tension sensor data rather than subjective tissue-quality scores, predictive accuracy might improve further. These findings suggest that AI/machine learning can synthesize multifactorial inputs, including biomechanical variables, to generate clinically useful risk predictions [85].
In summary, the preoperative phase is no longer just about imaging anatomy and optimizing medical comorbidities; it is becoming about mechanical diagnostics and virtual optimization. By identifying mechanical risk factors, such as a stiff liver, a soft pancreas, and abnormal flow—and by rehearsing the operation in silico, surgeons can significantly de-risk the actual surgery. This is a paradigm shift toward mechanics-informed precision surgery: treating each patient's tissues as unique materials and each operation as an engineering challenge to be optimized. When executed well, this should translate into fewer surprises in the operating room and better outcomes after. The next sections will delve into how these plans and predictions are applied and adjusted during the operation and afterwards.
4. Intraoperative phase: real-time mechanosensing and adaptive surgical maneuvers
Once in the operating room, mechanomedicine principles come into play through real-time in vivo diagnostic monitoring of mechanical conditions and targeted interventions to control the mechanical microenvironment of the surgery. This is perhaps the most immediately impactful phase, as decisions made intraoperatively can prevent complications before they start. The evidence base supporting intraoperative mechanosensing tools spans a spectrum: ICG fluorescence perfusion imaging and EndoFLIP distensibility measurement are supported by prospective clinical studies and are increasingly adopted in routine practice; intraoperative shear-wave elastography and implantable suture tension sensors are supported primarily by observational studies and early-phase trials; and closed-loop mechanical control systems remain at the experimental or the proof-of-concept stage. Two key aspects are addressed: (1) real-time mechanical monitoring, measuring tissue tension, distensibility, and perfusion to guide intraoperative decisions; and (2) targeted mechanical interventions, employing specialized materials and techniques to optimize the mechanical microenvironment. The intraoperative phase thus represents the most direct embodiment of theranostics in surgery: diagnostic sensors and therapeutic actuators operate simultaneously and in real time, with each diagnostic readout immediately triggering or adjusting a therapeutic response.
4.1 Real-time mechanical monitoring for surgical decision-making
Surgeons have long made qualitative judgments in the operating room, like “this anastomosis feels a bit tight” or “the tissue is stiff here”. Now, technology is translating those impressions into quantitative in vivo diagnostic readouts that can be objectively acted upon [45, 53, 54]. A suite of intraoperative biosensing and imaging tools has emerged to quantify mechanical parameters, forming the diagnostic arm of the intraoperative theranostic cycle.
Basic mechanical metrics: It is useful to recall the definitions of the mechanical terms (e.g., tension, stress, strain), as they apply to surgical tasks [12, 22, 23, 94]. Tension (T) typically refers to a force applied along a structure (in N, newtons). For a suture line or an anastomosis under pressure, one can estimate tension via Laplace's law: T = P × r, where P is the internal pressure and r is the radius [12]. Stress (σ) is force per unit area, for example, within the intestinal wall, whereas strain (ε) is the fractional change in length (ΔL/L₀) [22]. The elastic modulus (E) is the slope of the stress-strain curve (Δσ/Δε) for small deformations, essentially a measure of stiffness [22]. Finally, for hollow organs intraoperatively, surgeons use the distensibility index (DI), defined as cross-sectional area (of the lumen) divided by pressure, with units mm2/mmHg. DI is directly measured by devices like endoscopic functional lumen imaging probe (EndoFLIP) in real time [40, 53, 107]. These metrics, once abstract, are now being displayed on monitors during surgery to inform decisions.
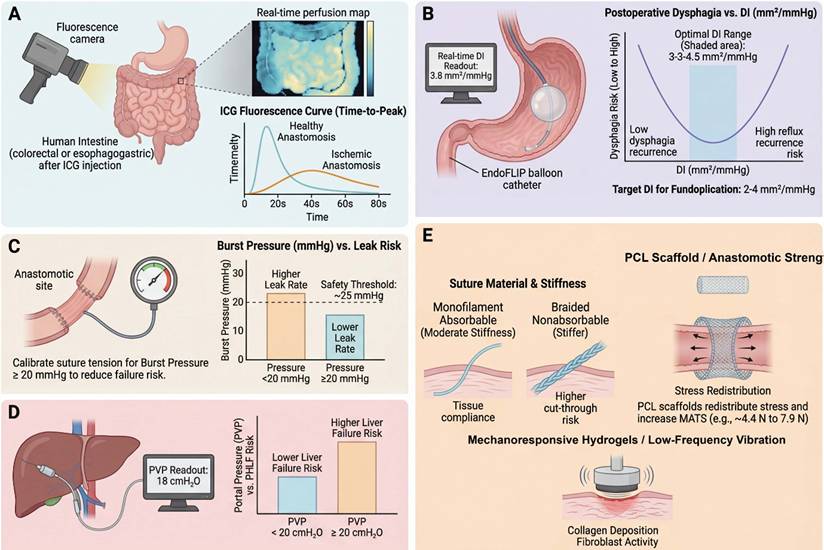
Colorectal anastomoses (preventing leaks): A critical intraoperative decision is whether an anastomosis is secure enough or if it needs revision. Traditionally, surgeons perform a leak test by insufflating air or fluid and seeing if anything leaks out of the suture line [108]. ICG fluorescence perfusion assessment, now widely adopted in colorectal and hepatobiliary surgery, has been evaluated in randomized trials. The AVOID trial (over 1000 patients) found that while routine ICG assessment did not significantly reduce overall leak rates in an unselected population, in certain high-risk cases it helped identify poorly perfused segments and change the surgical plan [109]. Quantitative perfusion thresholds are clinically useful: an ICG delay-to-peak exceeding ~60 seconds, or insufficient fluorescence intensity at the colorectal stump, indicates compromised perfusion and may prompt trimming to healthier tissue or creation of a protective stoma [59]. In fact, trials in esophagogastric surgery illustrate how real-time data can prevent errors: by using ICG to select a well-perfused anastomotic site, thereby avoiding hypoperfused zones that cause leaks, studies have achieved a significant drop in leak rates, for example, from approximately 14% to 6% [110].
Beyond perfusion, mechanical integrity testing is standard. Surgeons perform an intraoperative leak test (air leak test) by pressurizing the segment, not to be confused with destructive burst pressure (BP) testing used in experimental settings [43, 108]. A critical threshold often used is ~ 25 mmHg: a secure anastomosis should hold this pressure without leaking. If leaks occur (e.g., bubbles seen under saline), reinforcing stitches are applied [108]. While experimental studies note that suture material choice (silk vs. polyglactin) may have less impact on ultimate strength than surgical technique [45], standardized intraoperative leak testing is widely advocated to minimize postoperative failure [108]. Novel implantable sensors are being developed to continuously monitor anastomotic tension postoperatively. One prototype is a thin-film sensor embedded in the suture line which wirelessly transmits data and alerts staff to dangerous tension spikes caused by distension or coughing, prompting immediate interventions. This capability blurs the intra- and post-operative phases, exemplifying mechanomedicine's continuity [38, 111, 112].
Antireflux (fundoplication) surgery: Here, achieving the “just right” tightness of the wrap around the esophagus is critical, since too tight causes dysphagia, too loose causes reflux [40]. The EndoFLIP, now in routine clinical use for antireflux and bariatric procedures, has become a game-changer [40, 53]. It is a balloon catheter with impedance sensors that measure the diameter of the gastroesophageal junction in real time at a set volume, giving a readout of DI [40, 53, 107]. Surgeons can now titrate their fundoplication during surgery based on EndoFLIP readings. For instance, studies reported that at a 40 mL balloon fill, a cross-sectional DI > ~ 3.5-3.6 mm2/mmHg correlates with minimal postoperative dysphagia, meaning the wrap is not too tight [53]. However, if DI is much higher (> 6.0 mm2/mmHg at 40 mL), the wrap might be too loose, potentially risking hernia recurrence or persistent reflux. Related research suggests aiming for a final DI between ~ 2 and 4 mm2/mmHg. In practice, if EndoFLIP shows a DI of 1.0 mm2/mmHg at 30 mL, indicating excessive tightness, the surgeon may consider loosening the wrap. Conversely, if it shows 6.5 mm2/mmHg at 40 mL (very loose), they might add an extra stitch [40, 53]. A study provided evidence that using FLIP to guide these adjustments reduced both dysphagia rates and wrap failures [40]. This is a prime example of intraoperative biomechanical guidance; the surgery outcome can be optimized by hitting a numerical target rather than solely relying on feel. EndoFLIP is now being explored in other domains too, like assessing esophageal strictures or even pyloric tightness during gastric surgeries [40, 53, 107].
Liver surgery (hemodynamics): The resection of major liver volumes presents the primary clinical challenge of preventing post-hepatectomy liver failure, specifically small-for-size syndrome, wherein the remnant liver is incapable of sustaining the portal inflow. Consequently, intraoperative measurement of portal venous pressure (PVP) following vascular occlusion or test-clamping has emerged as a critical decision-making guide [21, 68, 88]. A post-resection PVP exceeding 20 mmHg is a known risk factor for liver failure [113]. In such a case, one might abort a major resection or perform an additional procedure to modulate portal pressure, such as a splenectomy or the creation of a shunt [21, 88]. In one recent study, patients with PVP above 20 had a significantly higher incidence of liver failure; keeping PVP < 18 was associated with safer outcomes. Some liver units have adopted an intraoperative algorithm: measure PVP after resection; if ≥ 20 mmHg, consider interventions or stage the resection [68]. Quantifying a parameter like portal pressure, which replaces the subjective assessment of liver congestion with an objective threshold, is a proactive strategy analogous to how cardiothoracic surgeons guide therapy using pulmonary artery pressures [88].
Pancreatic surgery (stiffness monitoring): Preoperative pancreas stiffness has been mentioned, and intraoperatively there is also interest in directly measuring pancreatic texture [19, 46, 47]. Some specialized centers now perform intraoperative ultrasound elastography on the pancreas remnant after resection—a translational practice not yet widely standardized—to objectively quantify stiffness. A shear modulus can be calculated [47]. For a nearly incompressible tissue like pancreas, E ≈ 3μ. If elastography confirms a very low stiffness, consistent with a soft gland, the surgeon might take additional precautions for the pancreatico-enteric reconstruction [19, 46]. These may include using a duct stent, placing more drains, or in a high-risk case, even considering externalizing the pancreatic duct [47]. Conversely, a stiff gland might allow a tighter anastomosis or fewer adjuncts. These elastography devices in the operating room are an extension of the preoperative concept, giving real-time feedback on the organ's mechanical state to tailor reconstruction [19, 46].
Adhesiolysis and small bowel: When operating for adhesive obstruction, one risk is inadvertent enterotomy and another is postoperative re-adhesion. While mechanical monitoring is less established here, some interesting research has looked at intra-abdominal pressure and wall tension during these surgeries. Clinical studies suggest that keeping insufflation pressures as low as feasible during laparoscopy, for example around 8 mmHg in low-pressure protocols, may reduce the mechanical stress on tissues and improve postoperative recovery [114, 115]. Some surgeons now practice “low-pressure” laparoscopy for bowel obstruction for this reason, in combination with careful fluid management to maintain perfusion. Doppler flow probes or ICG angiography can also be used intraoperatively to ensure the released bowel loops regain adequate perfusion. All these measures revolve around monitoring and controlling key mechanical factors, such as pressure, tension, and perfusion, to optimize outcomes during surgery [41, 60].
In summary, the intraoperative diagnostic toolkit is rapidly expanding. Mechanical data acquired during surgery, including anastomotic tension, perfusion indices, and distensibility measurements, may help define baseline parameters for subsequent postoperative monitoring and rehabilitation. Available intraoperative tools include fluorescence imaging for blood flow, probes for pressure and tension, imaging for stiffness and distensibility, and AI-enhanced laparoscopic video analysis that may indicate tissue properties. By quantifying critical mechanical parameters on the spot, surgeons can make informed therapeutic choices, revise the anastomotic site, place additional reinforcing sutures, and adjust insufflation. This real-time coupling of in vivo mechanical diagnosis with immediate therapeutic action is the operational definition of intraoperative theranostics. Table 1 provides an overview of the correspondence between key mechanical parameters, instrumentation, and clinical scenarios across the perioperative continuum, illustrating how each diagnostic modality is paired with a specific therapeutic response.
Representative mechanical parameters, measurement modalities, and clinical scenarios in digestive surgery
| Mechanical dimension | Representative quantity | Typical values or thresholds | Assessment modality | Corresponding clinical scenario | Refs |
|---|---|---|---|---|---|
| Liver stiffness | Elastography-derived stiffness (kPa) | LS < 8.6 kPa: lower-risk hepatectomy candidacy; LS > 12-15 kPa: advanced fibrosis/high-risk liver reserve (C/T) | MRE; 2D-SWE | Preoperative candidacy assessment and planning for liver resection | [12, 67, 68, 86] |
| Pancreatic stiffness | SWV / elastography-derived stiffness | Low stiffness (e.g., SWV < 1.3 m/s) associated with higher POPF risk (T) | SWE; ultrasound elastography | Preoperative risk assessment before pancreatoduodenectomy | [19, 46, 47] |
| Lumen distensibility | DI = cross-sectional area / intraluminal pressure (mm2/mmHg) | Low DI indicates an overly tight wrap; target ranges are procedure-dependent, often about 2-4 mm2/mmHg or > 3.5 mm2/mmHg at 40 mL (C/T) | EndoFLIP | Intraoperative assessment and adjustment of fundoplication | [40, 53, 107] |
| Perfusion / microcirculation | Quantitative fluorescence parameters | Delayed fluorescence or weak intensity suggests impaired perfusion (T) | Quantitative ICG fluorescence imaging | Intraoperative bowel/conduit perfusion assessment and anastomotic site selection | [59, 109, 110, 158] |
| Portal venous pressure | PVP | PVP ≥ 20 mmHg after clamping/resection indicates high risk in selected studies (T) | Direct intraoperative portal pressure measurement | Intraoperative hemodynamic assessment during major hepatectomy | [88] |
| Intra-abdominal pressure | IAP | IAH: IAP ≥ 12 mmHg; ACS: IAP ≥ 20 mmHg (C) | Bladder pressure monitoring; continuous IAP monitoring | Perioperative and postoperative surveillance after major abdominal surgery | [135] |
| Bowel / anastomotic remodeling stiffness | Follow-up stiffness by strain elastography or SWE | No universally accepted cutoff; increasing stiffness may suggest fibrotic remodeling or stricture risk (T) | Ultrasound strain elastography; SWE | Long-term follow-up of Crohn's strictures or postoperative anastomotic remodeling | [20, 56, 91] |
| Fluid shear stress | τ = μ (∂u/∂y) | Physiological-like shear ranges around 0.2-0.5 Pa in intestinal epithelial in vitro systems; not a direct bedside threshold (E) | Organ-on-chip; microfluidics; CFD simulation | Mechanistic modeling of epithelial barrier function and mechanobiology | [49, 50] |
Note: Thresholds are classified as (C) clinically implemented, (T) translational, and (E) experimental. Quantitative values are modality-, organ-, and endpoint-dependent.
Abbreviations: LS, liver stiffness; SWE, shear-wave elastography; MRE, magnetic resonance elastography; SWV, shear-wave velocity; POPF, postoperative pancreatic fistula; DI, distensibility index; EndoFLIP, endoscopic functional lumen imaging probe; ICG, indocyanine green; PVP, portal venous pressure; IAP, intra-abdominal pressure; IAH, intra-abdominal hypertension; ACS, abdominal compartment syndrome; CFD, computational fluid dynamics.
4.2 Targeted mechanical interventions during surgery
Mechanomedicine also encompasses intraoperative interventions, representing the therapeutic arm of the theranostic cycle, that actively shape the mechanical microenvironment on the basis of real-time diagnostic readouts [32].
Mechanical compatibility between biomaterials and native tissue is a key intraoperative principle: sutures, meshes, and scaffolds should be selected for mechanical properties compatible with native tissue [11, 48]. Excessive suture stiffness may promote cut-through under tension, whereas insufficient elasticity may fail to maintain tissue approximation under load [65, 66]. Next-generation sutures—combining natural polymers with graphene, or fabricated from poly(3-hydroxybutyrate-co-4-hydroxybutyrate) copolymers—achieve elasticity closer to soft tissue, allowing the suture to accommodate postoperative swelling while gradually transferring load to the healing tissue [48, 66, 116].
Emerging work indicates that intelligent robotic suturing may be enabled by both real-time visual perception and mechanically encoded force control: one study demonstrated real-time articulated-joint detection for robotic feedback [101], whereas another showed that slipknot-gauged sutures improved knotting-force precision, enhanced tissue perfusion in preclinical models, and allowed vision-based robotic stopping during suturing [117]. Staple-line reinforcement with bovine pericardial strips or bioabsorbable glycolide felts—an established clinical practice in bariatric and thoracic surgery—distributes force more evenly along the staple line, reducing peak stress by over 30% and improving mechanical boundary conditions for healing [22, 94, 118].
Another intraoperative domain under experimental investigation is pharmacomechanical modulation: locally delivering drugs to influence the mechanobiology of healing [44, 81]. When mechanical diagnosis identifies a high-risk anastomosis (e.g., soft pancreas by intraoperative elastography), targeted pharmacological agents are applied locally. For instance, researchers are investigating the use of matrix metalloproteinase (MMP) inhibitors or collagen crosslinkers at anastomosis sites to modulate the rate of extracellular matrix (ECM) degradation [119]. Although a certain degree of MMP activity is required during early postoperative remodeling, excessive activity may weaken the anastomosis [71]. Studies in other tissues, such as spinal cord, suggest MMP inhibition can modulate scarring [120], a concept potentially applicable to anastomosis. Another target is ROCK, part of the pathway that leads to myofibroblast contraction and fibrosis. Using a ROCK inhibitor locally can potentially reduce hyper-contraction of myofibroblasts and limit scar and stricture formation [56, 73]. While these are not yet standard, the key point is that mechanotransduction pathways, such as YAP/TAZ and FAK-RhoA, can be pharmacologically modulated [37, 58, 78]. For instance, anti-fibrotic gels containing agents such as halofuginone or losartan have been explored as local strategies to reduce adhesions and strictures by modulating TGF-β signaling and myofibroblast activation [58, 71], essentially trying to dial down the excessive mechanical signaling that leads to over-scarring [80].
A notable concept is the neuro-immuno-mechanical triad in wound healing [26]: surgical dissection simultaneously induces mechanical injury, nerve disruption, and immune activation. Mechanical stress modulates immune cell behavior, while inflammatory responses in turn reshape tissue mechanics through edema and fibrotic changes [26, 37]. Exemplifying mechanosensitivity, macrophages polarize into a pro-inflammatory M1 state under high stiffness or strain, conversely adopting a pro-healing M2 phenotype in soft environments, thereby guiding the inflammatory response [72, 78, 81]. Thus, control of mechanical conditions may also modulate the immune response in a manner that favors healing [26]. In adhesive small bowel obstruction surgery, for example, simply releasing tension by lysing adhesion bands, followed potentially by maintaining low-pressure peritoneal CO2 insufflation to prevent organs from recoalescing tightly, might reduce the drive for M1 macrophages and intense fibrosis [114, 121]. Additionally, drugs that target mechanotransduction in immune cells are being explored, e.g., modulators of YAP/TAZ in macrophages to encourage a healing phenotype [58, 82, 122]. These approaches remain experimental, but underscore a key insight: the mechanical microenvironment and immune response are intimately linked [123]. Simple intraoperative measures—minimizing unnecessary traction, keeping tissues moist, and employing gentle handling—are low-tech ways to favor a pro-healing milieu [74].
Local pro-healing therapies represent a further intraoperative strategy. Small trials and animal studies show that fibrin sealant mixed with bFGF improves microvessel density and anastomotic burst strength [11, 58, 124, 125]. A key challenge is controlled release: unregulated growth factor delivery risks leaky vessels or exuberant granulation [77, 126]. Biodegradable 3D/4D-printed scaffolds—currently at prototype and the proof-of-concept stage—address this by embedding growth factors or anti-fibrotics for timed release tuned to the healing timeline, with prototype smart anastomotic patches providing initial mechanical support before dissolving as tissue regains strength [118, 127, 128].
Direct mechanical stimulation represents an experimental intraoperative strategy, currently supported only by animal model data [129]. In animal models, low-frequency vibration (~50 Hz, 5 minutes) applied to a completed intestinal anastomosis improved collagen alignment and smooth muscle actin expression without tissue damage [129]. Periodic tension cycling during surgery may similarly condition tissue to handle postoperative strains [25, 79, 130]. These approaches remain preclinical but are grounded in mechanotransduction: controlled mechanical stress activates pro-healing pathways, whereas either excessive or absent stress is detrimental [52, 72, 74, 78].
Figure 4 illustrates these intraoperative strategies in tandem. The overarching goal is to ensure the anastomosis or resection bed has adequate perfusion, controlled tension, well-distributed stresses, and optimized biomaterial support—collectively reducing the risk of leaks, strictures, and other complications arising from suboptimal mechanical conditions.
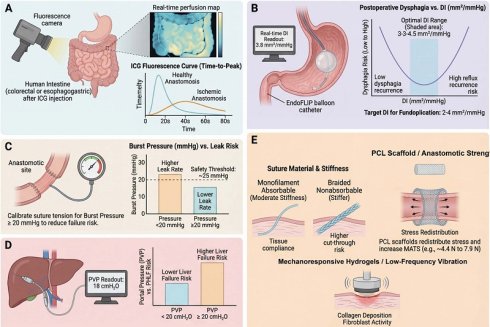
Intraoperative mechanosensing and targeted mechanical interventions to optimize anastomotic safety and organ function. (A) Quantitative perfusion assessment of anastomotic sites. Indocyanine green (ICG) near-infrared fluorescence is used to compare candidate transection margins in colorectal and esophagogastric surgery. Perfusion maps and time-intensity curves allow surgeons to select segments with rapid, high-amplitude uptake (for example, time-to-peak < 40 s, relative fluorescence ≥70% of reference) and avoid hypoperfused zones [59, 110]. Randomized and cohort studies indicate that fluorescence-guided site selection reduces anastomotic leakage in selected or high-risk subgroups, even though overall leak rates in unselected populations may not significantly decrease [109]. (B) Functional lumen imaging and distensibility index (DI)-based titration of antireflux surgery. Endoscopic functional lumen imaging probe (EndoFLIP) measurements during fundoplication reveal a U-shaped relationship between DI (mm2/mmHg) and postoperative symptoms: low DI (<1.5 at 30 mL) correlates with dysphagia, whereas very high DI (> 6.0 at 40 mL) correlates with reflux recurrence [40]. Intermediate DI values (~ 3-4 at 40 mL) define an optimal window with minimal dysphagia and acceptable reflux, providing a mechanistically grounded target to guide wrap tightness and bougie size intraoperatively [53]. (C) Anastomotic tension and burst pressure thresholds. Intraoperative pressure testing of colorectal anastomoses demonstrates that constructs failing at low pressures (< 20-25 mmHg) are more likely to leak clinically [43]. Standardized air or dye leak testing at predefined pressures, combined with reinforcement of mechanically weak sites, shifts the distribution of burst pressures upward and lowers leak incidence in clinical series [108]. Emerging implantable tension sensors further allow continuous quantification of loading across anastomoses as bowel distends postoperatively [38]. (D) Portal pressure-guided liver resection. In patients undergoing major hepatectomy, intraoperatively measured portal venous pressure (PVP) serves as a physiologic readout of remnant capacity. PVP ≥ ~ 19.5 cmH2O after inflow control is associated with higher rates of post-hepatectomy liver failure, prompting consideration of reduced resection extent, staged approaches or adjunctive portal pressure-lowering strategies [88]. (E) Targeted intraoperative mechanical interventions. Mechanocompatible sutures and buttressing materials are selected to match tissue stiffness and redistribute stress along anastomotic lines, reducing cutting and micro-ischemia [65, 75]. Local delivery of bioactive molecules (for example, matrix metalloproteinase inhibitors or Rho-associated kinase (ROCK) inhibitors) modulates mechanotransduction and fibrosis in the early healing niche [73, 120], while low-amplitude vibration or controlled loading may beneficially prime fibroblasts and endothelial cells [129]. The figure was created by Figdraw (www.figdraw.com).
5. Postoperative phase: mechanotherapy and personalized rehabilitation
The influence of mechanomedicine does not end when the surgery is over; the postoperative period is crucial for solidifying the success of the operation. The evidence supporting postoperative mechanotherapy also varies by maturity: early mobilization and enteral nutrition are supported by randomized controlled trials and meta-analyses; continuous IAP monitoring is supported by prospective observational studies; and mechanoresponsive drug delivery systems and implantable anastomotic biosensors remain largely experimental, with evidence from animal models and early-phase feasibility studies. In this phase, the focus shifts to promoting tissue repair, monitoring for early signs of trouble, and tailoring rehabilitation based on mechanical insights. This postoperative phase completes the theranostic cycle: continuous in vivo mechanical diagnostics, including intra-abdominal pressure monitoring, anastomotic stiffness tracking, and wearable biosensors, provide real-time feedback that drives personalized therapeutic adjustments. In this section, three aspects are discussed: (1) Mechanical modulation to promote healing, how controlling mechanical loads and environment after surgery (e.g., through mobilization, supports, and mechanoresponsive scaffolds) can enhance tissue repair; (2) Early warning systems for complications, novel biosensors and monitoring strategies to catch issues like leaks or compartment syndrome before they fully manifest; and (3) Long-term functional outcomes through mechanics-based rehabilitation, focusing on how the mechanical remodeling of tissues, such as anastomotic maturation, correlates with patient function, and how targeted interventions may be used to support optimal recovery.
5.1 Mechanical modulation to promote tissue repair
Right after surgery, an anastomosis or surgical repair is at its weakest [11]. The body then initiates a healing sequence typically described as inflammation (days 1-3), proliferation (days 4-7), and remodeling (beyond the first postoperative week) [26, 30]. Mechanical factors at each stage can speed up or hinder progress [37].
One immediate consideration is stress distribution in the fresh repair [11]. This issue has already been introduced in the intraoperative context, but it remains relevant postoperatively [25, 131]. If one spot bears too much stress, it may tear. Research has pinpointed the mesenteric border of intestinal anastomoses as a common weak link, likely due to stress risers there [23]. To address this, engineers have designed bioresorbable scaffolds or patches that can be placed over the anastomosis, particularly reinforcing the mesenteric side [118, 132]. For example, a patch made of poly-ε-caprolactone (PCL) nanofibers has been tested in pig models: it acted like a temporary reinforcing support that distributed tension more evenly around the anastomosis. In ischemic conditions, such a scaffold increased the maximum tensile strength of the anastomosis almost two-fold at 1 week post-op from approximately 4.4 N to 7.9 N [118]. The scaffold dissolves over a few months, by which time the tissue has strengthened [133, 134]. This concept of force redistribution is beginning to translate clinically in other contexts too, e.g., internal adhesion barriers that also bear load so that healing tissues aren't directly strained. Figure 5 will highlight such strategies.
Postoperative intelligent mechanical monitoring, force redistribution and personalized rehabilitation in digestive surgery. (A) Continuous intra-abdominal pressure (IAP) and abdominal wall tension monitoring for early detection of decompensation. Representative IAP trajectories over the first 5 postoperative days are shown for two patients. Patient 1 maintains IAP values within the normal range (<12 mmHg), whereas Patient 2 exhibits a progressive rise into the intra-abdominal hypertension (IAH) range (≥12 mmHg) with excursions above 20 mmHg, consistent with abdominal compartment syndrome (ACS) risk [114]. Parallel abdominal wall tension traces, recorded by transcutaneous stretch sensors, mirror these changes. Continuous or high-frequency monitoring via novel bladder-based or implantable pressure sensors enables threshold-based alerts and timely interventions (for example, fluid removal, decompression, loosening of external supports) before irreversible organ dysfunction occurs [143, 149]. (B) Mechanical reinforcement of anastomoses and stress redistribution. Finite-element-style stress maps of an intestinal anastomosis highlight the mesenteric border as a biomechanical weak point with high peak tensile stress and low maximal tensile strength (MATS ≈ 4.4 ± 2.5 N under induced ischaemia). Incorporation of a poly-ε-caprolactone (PCL) scaffold redistributes stress across the circumference, lowers peak stress and increases MATS to ≈ 7.9 ± 4.2 N, thereby providing a mechanical safety margin during the vulnerable early healing period [118]. (C) Early mobilization as a systemic mechanotherapy. In a randomized controlled trial, mobilization initiated within 2 h after major abdominal surgery improved peripheral oxygen saturation by ~ 2.5% and arterial partial pressure of oxygen by ~ 1.4 kPa compared with standard care [136]. Early mobilization likely improves diaphragm excursion, lowers sustained IAP, promotes mesenteric perfusion and accelerates gastrointestinal motility, providing a low-cost, system-level mechanical intervention integrated into enhanced recovery pathways [138]. (D) Closed-loop, mechanically aware rehabilitation and drug delivery. Postoperative care can be conceptualized as a closed-loop system in which multichannel mechanical inputs (IAP, abdominal wall tension, elastography-derived stiffness of the anastomosis) feed into a control layer that prescribes individualized adjustments in mobilization intensity, external mechanical support (for example, abdominal binders), enteral feeding regimens and local or systemic therapies [137, 145]. Mechanoresponsive drug delivery systems, such as hydrogels engineered to release matrix metalloproteinase inhibitors or growth factors in response to local strain or stress, further align biochemical cues with mechanical needs [81, 151]. The figure was created by Figdraw (www.figdraw.com).
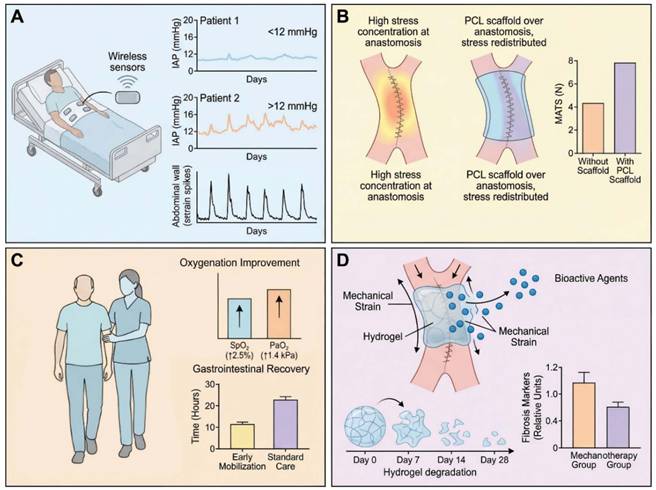
Another key aspect is the viscoelastic nature of healing tissues [12, 62, 81]. Immediately after surgery, tissues are often edematous and soft, but then gain stiffness over the following week as collagen is deposited [30, 71]. It has been observed in GI anastomoses that mechanical strength rises nonlinearly over the first week, slow at first, then rapidly after day ~ 3-4 as collagen fibrils mature [11]. This means that days 0-3 are a critical window where support is needed. Some surgeons use devices like vacuum-assisted closure (VAC) for difficult abdominal wounds to help approximate tissues under controlled tension, recognizing that the initial days are precarious [74]. Meanwhile, tissue engineers examine the time-dependent stress relaxation of tissues. For instance, when the stomach wall is stretched, it exhibits viscoelastic relaxation, which is characterized by an initial high stress that gradually decreases under constant strain; using models like the Ogden hyperelastic model fitted to stress-relaxation tests, researchers can quantify the tissue's parameters and see how a surgical procedure such as a sleeve gastrectomy alters them [34, 36, 102]. Weight loss surgery significantly reduced the elastic modulus (E) and yield stress of the stomach, with reports showing that elastic modulus (E) decreased to about 1.5 MPa after insertion, far below natural levels [34, 36, 102]. This implies that the stomach wall is floppier right after such surgery, potentially delaying the return of normal motility and strength. Knowing this, one might adjust feeding regimens or add external support (like a binder) until the tissue re-stiffens with healing [11]. Similarly, in colon surgery, measuring the compliance of the abdominal cavity is important [131]. Low abdominal compliance, characterized by a stiff abdomen often due to obesity or prior surgeries, means any increase in intra-abdominal volume translates to a significant pressure spike, which can strain repairs [131]. This underlies why obese patients with thick abdominal walls have higher risk of wound dehiscence, their abdomens don't expand easily, so internal pressure can blow out incisions [34]. FE modeling studies have confirmed that adding a mesh reinforcement in the abdominal midline increases wall stiffness but also can increase overall abdominal compliance if overlapped properly, distributing forces better [135]. Clinically, this suggests that prophylactic mesh in high-risk laparotomy closures may reduce hernias not just by brute force but by modulating wall mechanics [11, 135].
Early mobilization is one of the simplest and most effective mechanical interventions post-op [136]. It improves oxygenation, but also influences gut motility and intra-abdominal pressure patterns [136, 137]. Early mobilization prevents the persistently elevated intra-abdominal pressure as observed in bedridden patients, a phenomenon attributable to factors such as diaphragm splinting and ileus [114]. Enhanced Recovery After Surgery (ERAS) protocols universally include early mobilization for these reasons, which is supported by findings that even sitting in a chair for a few hours on the day of surgery leads to improved pulmonary function and potentially faster return of bowel function [136]. Another study in high-risk abdominal surgery patients found that more than 80% of patients were able to complete four mobilization sessions per day by postoperative day 3, which correlated with shorter length of stay [138]. Mechanistically, early mobilization likely stimulates mesenteric circulation and the release of growth factors or hormones that facilitate healing, with supporting data showing that exercise can increase VEGF levels and improve collagen deposition quality [54, 58]. It also conditions the abdominal wall, preventing stiffening from inactivity [131].
Nutritional support has a mechanical angle too [139]. Early enteral feeding, in contrast to maintaining a nil-by-mouth (NPO) status or total parenteral nutrition, promotes gastrointestinal motility; the resulting peristaltic activity imposes cyclical mechanical stresses on the anastomosis, delivering a stimulus that is both gentle and physiologically normative [79, 140]. This may actually strengthen the repair by encouraging normal tissue remodeling as opposed to stasis [25]. Additionally, luminal nutrition ensures that the mucosa, which is one of the fastest-healing layers, recovers quickly to seal the inside [139]. Studies in critical care have shown benefits of enteral over parenteral nutrition in outcomes, partly attributed to maintenance of gut integrity [140, 141]. Conversely, excessively aggressive feeding may increase luminal pressure or overdistension, and therefore a balanced approach is required [137]. Fluid dynamics of chyme are considered: a high-volume, low-viscosity feed might cause more turbulent flow and shear stress in the gut; therefore, starting with small, frequent, nutrient-dense feeds (which have higher viscosity) could impose less shear stress on the anastomosis initially [42]. Using Caco-2 cells as an intestinal epithelial model, experiments established a beneficial shear stress range of 0.2-0.5 Pa for promoting mucus secretion and barrier function, with deviations to either extreme being detrimental [49, 50]. A moderate flow (as occurs with trickle feeds) may actually encourage the epithelium to strengthen, whereas no flow (ileus) or extremely high flow (diarrhea) could be harmful to healing [49]. So, practically, starting enteral feeds early at a slow rate aligns with this principle, providing that gentle mechanical stimulation along with nutrition [137, 140].
All these strategies, reinforcement devices, mobilization, careful feeding, are forms of mechanotherapy: treating the patient by modulating mechanical conditions [37]. They can be thought of as “prescriptions” just like medications. In some places, physical therapists now work with surgical patients on the first day after surgery to mobilize them, essentially prescribing a specific mechanical activity as therapy [136, 138]. Abdominal binders are another simple tool: by supporting the abdominal wall, they can reduce the force on incisions with coughing or movement [135]. However, binders also increase external constraint, so one must not overtighten which would raise baseline IAP [137, 142]. It is a fine example of how subtle mechanical nuances matter, a binder snug enough to support but not so tight as to elevate pressure.
5.2 Early warning and proactive mechanical intervention for complications
Despite these efforts, complications can still occur; early detection and prompt intervention are therefore crucial [2, 4]. Mechanomedicine contributes here by providing continuous in vivo diagnostic monitoring systems and criteria for early intervention based on mechanical signals [37]. The postoperative biosensor ecosystem, encompassing implantable pressure capsules, wearable abdominal strain sensors, and wireless motility trackers, constitutes a distributed theranostic network in which each diagnostic signal is coupled to a predefined therapeutic response protocol.
One of the clearest cases is intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS) after major surgery [114, 137]. If fluid accumulates or organs swell significantly, IAP can rise to dangerous levels, impairing organ perfusion and threatening anastomoses [131]. Traditionally, IAP is measured intermittently via the bladder manometry technique [142]. Continuous or high-frequency IAP monitoring devices are emerging [143]. For example, a wireless bladder pressure biosensor has been developed that sits in the Foley catheter and transmits real-time bladder pressure (which reflects IAP) [143]. There are also implantable pressure capsules, conceptually similar to the “SmartPill” but designed for the peritoneal cavity, which can provide constant telemetric pressure readings [144]. These devices represent miniaturized in vivo diagnostic biosensors that embody the theranostic principle and could potentially support alert-based intervention protocols once validated.
For anastomotic leak detection, mechanical and biochemical in vivo diagnostic monitoring holds promise but remains largely experimental [95, 96]. One approach is measuring transanastomotic impedance or pressure changes [38]. A healing anastomosis, if intact, often exhibits a certain pattern: slight increase in luminal pressure when peristalsis hits it, and no free air leak. If a dehiscence begins, abnormal pressure fluctuations or a loss of expected resistance may be observed [25, 79]. In fact, one device currently in trials is a nanoparticle-based biosensor film that changes electrical resistance in response to a local pH change, a change that occurs upon exposure to leaking bowel contents [145-147]. This nanobiosensor exemplifies the theranostic potential of implantable diagnostic devices: it provides continuous in vivo monitoring of anastomotic integrity and, in future iterations, could be coupled to a drug-releasing layer that responds to the same pH signal by locally delivering antimicrobials or sealants, thereby integrating diagnosis and therapy in a single implantable platform. Another simpler measure is abdominal wall tension: if an anastomotic leak begins, often ileus and fluid collections develop, subtly increasing IAP and causing abdominal wall strain [131]. A noninvasive device fitted with abdominal stretch sensors can infer elevations in intra-abdominal pressure by monitoring changes in abdominal wall strain [142]. A pilot study reported a strong correlation (r ~ 0.85) between abdominal surface strain and bladder pressure in postoperative patients. In principle, such a device could be used on the ward to flag rapid increases in abdominal wall strain or girth that may warrant further evaluation for ileus, fluid accumulation, hemorrhage, or leak [142]. These wearable and implantable sensing approaches are still at an early experimental stage, but the direction is clear: mechanical vital signs, such as intra-abdominal pressure, abdominal wall tension, and organ movement, will become part of the standard monitoring parameters for postoperative patients, alongside heart rate and temperature [148-150].
Another technology is the “SmartPill” intra-abdominal sensor capsule mentioned above [144]. A version of this can measure pressure from 0 to 30 mmHg with 0.1 mmHg resolution and respond within < 1 second [144]. If placed near an anastomosis, such a device could theoretically detect pressure spikes or vibrations indicative of a leak, which are caused by the distinct pressure pattern generated as air bubbles pass through a defect [25]. Integration of such data into AI-assisted monitoring systems could improve early detection of anastomotic leaks. At present, clinical recognition still relies mainly on delayed signs such as fever, tachycardia, peritonitis, or intermittent imaging assessment [84, 95]. These implantable biosensor platforms, combining point-of-care in vivo diagnostics with AI-driven interpretation, represent a new generation of theranostic devices for postoperative care.
Another area where mechanical early warning is useful is ileus vs. obstruction differentiation in the days after surgery [39]. Everyone expects some ileus post-op, but differentiating a prolonged ileus from an early adhesive obstruction is hard [25]. The use of mechanical monitoring, for instance via a wireless motility capsule to measure intraluminal pressures and motility, could enable the differentiation of coordinated high-pressure patterns characteristic of obstructive pathologies from the patterns indicative of an atonic ileus [79, 144]. Hypothetically, future systems may define quantitative motility thresholds for distinguishing prolonged ileus from early mechanical obstruction.
Lastly, consider intraoperatively applied anti-adhesion materials that provide preventive mechanical benefits during the postoperative period, such as PuraStat, a hemostatic peptide gel [121]. These not only help hemostasis but can also serve a mechanical function: they create a protective layer that reduces friction and shear between organs as they slide postoperatively [121, 151]. PuraStat, for instance, has a measured elastic modulus tuned to mimic peritoneal tissue, allowing it to move with adjacent tissues rather than generating additional shear at the interface [121]. Related studies have shown that it reduced adhesion formation in a rat model, presumably because it absorbed mechanical stresses that would otherwise stimulate adhesion-forming cells [152, 153]. While this is more preventive than an “early warning”, it shows another proactive mechanical strategy: physically intervening to alter how tissues interface in the healing period [37, 154].
In summary, the postoperative period benefits from a proactive, surveillance mindset, using mechanical cues as early indicators and intervening before a problem becomes full-blown [37]. This strategy mirrors the evolution in cardiology from treating infarctions to preempting them by monitoring ischemia. In surgery, it entails detecting abnormal mechanical signatures, namely pathological pressure or strain, that foreshadow a leak, rather than waiting for clinical decline [95]. Implementing these requires interdisciplinary collaboration, surgeons, engineers, critical care physicians, to validate that these new monitors and devices truly correlate with meaningful endpoints [83, 84]. However, recent trends, including evolving regulatory pathways for such devices are evolving and a growing volume of clinical studies, highlight the potential of this technology [155, 156]. With the emergence of accurate continuous IAP monitoring systems, there is potential for this mechanical parameter to become a key diagnostic tool in the perioperative management of high-risk surgical patients, addressing the current limitations in diagnosing abdominal compartment syndrome.
5.3 Long-term functional outcomes and mechanics-based rehabilitation
Even after the immediate recovery phase, the mechanical microenvironment continues to influence long-term outcomes [37, 71]. Patients often care most about functional recovery, e.g., regaining normal bowel function, avoiding strictures or hernias, and returning to daily activities [56]. Long-term outcomes are closely related to how tissues remodel mechanically in the weeks and months after surgery [30, 72]. Long-term mechanical monitoring, using outpatient elastography and wearable biosensors, thus extends the theranostic cycle beyond the hospital, enabling personalized, data-driven rehabilitation that adjusts therapeutic interventions based on ongoing diagnostic measurements of tissue remodeling.
Consider an intestinal anastomosis months later, it should ideally exhibit compliance similar to that of the adjacent bowel so that peristalsis propels contents smoothly [25]. If it becomes a rigid scar ring (high stiffness), the patient might develop a stricture and obstructive symptoms [56, 69]. Thus, monitoring the mechanical properties of healing tissues in the longer term can identify who's at risk of such issues [20]. Techniques like ultrasound strain elastography or SWE can be used even after a patient has left the hospital, to measure anastomosis stiffness during follow-ups [20, 91]. In the context of Crohn's disease, SWE measures stiffness in strictures, with higher values signifying a fibrotic phenotype suggestive of a need for surgery, thereby distinguishing it from an inflammatory stricture amenable to medical treatment [20]. Similarly, a healing colorectal anastomosis that shows increasing stiffness over two months might indicate excessive fibrosis, prompting a potential intervention such as endoscopic dilation or the use of anti-fibrotic medications to prevent full narrowing [56]. Emerging evidence suggests that fibrotic remodeling is driven by reciprocal interactions between mechanical cues and profibrotic signaling pathways, including TGF-β/Smads, Hippo-YAP/TAZ, and mechanosensitive channels such as TRPV4, highlighting these pathways as potential targets for future personalized anti-fibrotic interventions in patients with mechanically high-risk healing phenotypes [58, 71, 80].
Another concept introduced is the Mechanical Compatibility Index (MCI) of an anastomosis. This index could combine measures of stiffness uniformity and strength [11]. A perfectly integrated anastomosis would have a MCI approaching 1, denoting that it is mechanically indistinguishable from the native surrounding tissue [12]. If the anastomosis is too stiff or too weak relative to adjacent tissue, the MCI deviates from the ideal, a deviation which one study proposed to quantify with a formula based on the ratios of its elastic modulus and tensile strength to those of normal tissue [148]. They found that an MCI within a specific range predicted the return of normal bowel function, whereas a low MCI predicted dysfunction due to the anastomosis being either excessively compliant or overly stiff [64, 148, 157]. This kind of quantitative metric is reminiscent of how cardiologists quantify valve function or graft patency; surgeons might do the same for repairs.
Long-term, patients worry about things like incisional hernias after abdominal surgery [135]. Mechanically, an incisional hernia is a failure of the abdominal wall to withstand internal pressure [131, 158]. From a mechanistic perspective, this may reflect reduced collagen strength or uneven force distribution, or maybe the distribution of forces across their closure was uneven [135]. Some surgeons proactively reinforce high-risk incisions with mesh to offload forces, while follow-up imaging or even dynamic MRI can assess how the abdominal wall moves and strains under Valsalva, enabling differentiation between patients with abnormal bulge and high strain at the scar who may benefit from earlier hernia repair versus those whose wall remains stable [131, 135].
Adhesions are another long-term outcome [121]. Mechanically, adhesions form when scar bridges between surfaces that move relative to each other [74]. Reduction of friction, for example through adhesion barriers, or short-term reduction of relative motion between surfaces may lessen adhesion formation. Conversely, too much motion early might cause micro-bleeds and inflammation leading to adhesions [26]. An appropriate mechanical balance is required, in which complete immobilization is avoided while unnecessary abrasion is minimized through microsurgical gentle technique and copious irrigation to prevent dry surfaces from scraping [121].
It is worth noting the interplay of mechanics and ongoing biology: radiation therapy after surgery can stiffen tissues through radiation fibrosis [71, 80]. Patients who get pelvic radiation post resection might have stricter, less compliant anastomoses and higher risk of bowel dysfunction [157]. Recognizing this risk, clinicians might employ more aggressive dilation or medical therapy to mitigate radiation fibrosis, such as pentoxifylline and vitamin E, which some oncologists use for radiation proctitis to soften tissues [71]. Again, mechanomedicine provides the rationale: radiation injures microvasculature and causes collagen crosslinking, a mechanical issue [37].
Finally, patient-specific factors like age and activity level matter [74]. Older patients heal more slowly and often develop stiffer scars with less elastic collagen [26]. Encouraging appropriate exercise, such as light core exercises a few weeks after surgery, could stimulate better remodeling of the abdominal wall scar, analogous to Wolff's law for fascia [136]. Younger patients might exhibit excessive healing responses, including keloids and exuberant scars; modulating the mechanical microenvironment by avoiding premature scar overloading could potentially prevent such outcomes [74].
In essence, just as physical therapy is essential after orthopedic surgery to ensure optimal function, mechanical rehabilitation after abdominal surgery can be viewed as a continual process of monitoring surgical repair integration and adjusting patient activities or applying therapies to guide that integration. Advanced imaging and sensors might be employed to ensure that by three months postoperatively, the stiffness of the repair approximates native tissue, or if discrepancies exist, to implement targeted interventions such as endoscopic stricturotomy for excessive stiffness or supportive abdominal binders for insufficient tension.
Table 2 summarizes the modulation strategies across the preoperative, intraoperative, and postoperative stages.
Modulation of the mechanical microenvironment in digestive surgery across the perioperative continuum
| Stage | Key mechanical parameters | Assessment / monitoring tools | Representative thresholds or examples | Typical clinical decisions / interventions | Maturity | References |
|---|---|---|---|---|---|---|
| Preoperative | Liver stiffness; fibrosis burden | MRE; 2D-SWE | LS < 8.6 kPa suggests lower-risk hepatectomy candidacy; LS > 12-15 kPa suggests advanced fibrosis/high-risk liver reserve | Adjust resection extent; consider staged surgery or intensified liver protection | C/T | [21, 67, 68, 86] |
| Preoperative | Pancreatic stiffness / gland texture | SWE; ultrasound elastography | Low pancreatic stiffness (e.g., SWV < 1.3 m/s) associated with higher POPF risk | Modify anastomotic strategy; consider stent placement, drain planning, or intensified fistula prevention | T | [19, 46, 47] |
| Preoperative | Tumor or stricture mechanical phenotype | SWE; MRE | Decrease in CRLM stiffness during chemotherapy may indicate response; higher bowel stiffness may suggest fibrotic rather than inflammatory phenotype | Refine timing of surgery; help distinguish surgery from medical therapy in fibrostenotic disease | T | [20, 62, 90] |
| Preoperative | Patient-specific tissue mechanics in simulation models | CT/MRI-based geometry; elastography-informed FE modeling | Outputs include predicted stress concentration, deformation, or remnant load | Optimize resection plane, anastomotic site, or reconstruction strategy in silico | T/E | [22, 57, 94, 97, 101] |
| Intraoperative | Lumen distensibility | EndoFLIP | Low DI indicates an overly tight wrap; selected studies suggest target DI ranges of about 2-4 mm2/mmHg or > 3.5 mm2/mmHg at 40 mL | Adjust wrap tightness or reconstruction before completion | C/T | [40, 53, 107] |
| Intraoperative | Anastomotic perfusion / conduit perfusion | Quantitative ICG fluorescence imaging | Delayed fluorescence or weak intensity suggests impaired perfusion | Revise transection line; select a better-perfused anastomotic site; consider diversion or reinforcement | C/T | [59, 109, 110, 158] |
| Intraoperative | Portal inflow and remnant liver hemodynamic load | Intraoperative portal pressure measurement | PVP ≥ 20 mmHg after resection or clamping suggests high risk in selected studies | Modify or stage hepatectomy; consider portal pressure modulation | T | [88] |
| Intraoperative | Tissue force transmission during suturing | Mechanical sensors; force-sensing concepts; slipknot-gauged systems | No universally accepted clinical threshold; device-assisted force control aims to reduce excessive traction | Improve suture force consistency; reduce cut-through risk; support robotic/assisted force control | T/E | [38, 116, 117] |
| Postoperative | Intra-abdominal pressure | Continuous bladder pressure monitoring; sensor-based IAP monitoring | IAP ≥ 12 mmHg = IAH; IAP ≥ 20 mmHg = ACS | Decompression, fluid optimization, abdominal pressure management, escalation of surveillance | C | [135, 140, 141, 146, 147] |
| Postoperative | Early warning of leak, distension, or impaired recovery | Implantable pH/impedance sensors; wearable abdominal strain sensors; wireless motility monitoring | No standardized threshold; abnormal dynamic trends may indicate leak, distension, or ileus/obstruction | Trigger imaging, drainage, antibiotic treatment, nutritional adjustment, or reoperation assessment | T/E | [140, 143-145, 147] |
| Postoperative | Mechanical support and load redistribution | Bioresorbable scaffolds; reinforcement patches; abdominal support | Benefit inferred from improved tensile strength, load sharing, or reduced local stress in translational studies | Reinforce vulnerable anastomoses or closures; reduce peak stress during early healing | T/E | [117, 130-133] |
| Postoperative / rehabilitation | Functional loading, motility, and tissue remodeling | Early mobilization; enteral nutrition; follow-up elastography | Early mobilization and early enteral feeding support recovery; increasing follow-up stiffness may suggest fibrotic remodeling or stricture risk | Promote perfusion and motility; tailor rehabilitation; intensify surveillance or intervene for stricture-prone healing | C/T | [20, 56, 91, 134, 135, 138, 139] |
Note: Technologies are classified by clinical maturity as (C) clinically implemented, (T) translational, and (E) experimental.
Abbreviations: LS, liver stiffness; SWE, shear-wave elastography; MRE, magnetic resonance elastography; CRLM, colorectal liver metastases; FE, finite element; DI, distensibility index; EndoFLIP, endoscopic functional lumen imaging probe; ICG, indocyanine green; PVP, portal venous pressure; POPF, postoperative pancreatic fistula; IAP, intra-abdominal pressure; IAH, intra-abdominal hypertension; ACS, abdominal compartment syndrome.
By continuously linking mechanical parameters to clinical decisions, from start to finish, the patient's journey becomes one of mechanically-informed care [37]. The long-term vision is that surgeons will not only operate but also guide tissue healing toward optimal integration, using data and devices to ensure that at 6 months postoperatively, the patient's tissues are as functional as possible [136]. This approach should reduce late complications like strictures, hernias, or adhesions that often necessitate reoperations or cause chronic issues [56, 135].
6. Future directions and challenges in mechanomedicine
As mechanomedicine in digestive surgery is an emerging field, significant innovations and challenges lie ahead. Several key future directions are outlined below, focusing on the integration of emerging technologies, the need for standardizing mechanical measures in clinical practice, and addressing foundational scientific questions that remain unresolved.
A central theme across all future directions is the progressive realization of a fully integrated theranostic system, in which in vitro diagnostic platforms, in vivo biosensors, and mechanoresponsive therapeutic devices are seamlessly connected through AI-driven decision layers.
6.1 Integrating emerging technologies for precision surgery
The coming years will likely see an even deeper convergence of mechanomedicine with advanced technologies such as AI, robotics, and biofabrication [37, 83]. On the intraoperative front, the rise of surgical robotics and intelligent assistance systems provides an ideal platform to incorporate mechanical diagnostic-therapeutic feedback loops. For instance, next-generation robotic surgery systems are being developed with built-in force biosensors and AI-driven controllers that can maintain optimal tension on tissues automatically. Related reports demonstrated a robot that could adjust its grasping force on bowel tissue in real time to avoid excessive stress, using reinforcement learning to learn the delicate balance [38, 103]. This hints at a future where the surgeon sets a target tension range, and the robot ensures that is not exceeded as it sutures, effectively embedding theranostic mechanomedicine algorithms into the robotic workflow. Such robotic theranostic systems, in which force biosensors continuously monitor the mechanical state of tissue and actuators simultaneously deliver the appropriate therapeutic response, could represent an advanced form of diagnostic-therapeutic integration.
AI-driven planning and guidance will also advance substantially. Mechanics-aware AI systems may eventually provide recommendations that extend beyond anatomical image analysis. For example, such platforms may help predict where high-stress regions are likely to arise under a given surgical approach. This capability could be integrated into augmented reality displays for surgeons, perhaps presenting a color overlay on organs indicating stiffness variations or risk zones, such as a red band showing areas where tension would be excessive if transected. Preliminary versions of this concept have been reported: one system analyzed liver elastography data and projected a stiffness map onto the surgeon's augmented reality glasses during liver resection to identify and avoid highly stiff, cirrhotic areas that might not heal well [21, 159].
Miniaturized, wireless biosensors—currently at the proof-of-concept stage—represent a major component of future theranostic integration. Building on implantable and wearable technologies discussed previously, future iterations will be increasingly sophisticated, possibly powered by the body through thermoelectric or piezoelectric conversion of movement-derived energy and networked via Internet of Things infrastructure. A patient recovering at home could have a tiny implanted intra-abdominal pressure biosensor and a wearable gut motility tracker transmitting data to a smartphone application that uses artificial intelligence to interpret signals and provide simplified guidance indicating either stable recovery or the need for clinical evaluation. Recent advances in implantable flexible and soft mechanical sensors suggest that wireless, mechanically conformable, and potentially bioresorbable platforms for in vivo monitoring are becoming increasingly feasible, laying the groundwork for transient postoperative biosensors that could integrate sensing, communication, and eventual resorption [160, 161]. Imagine a theranostic implant placed at an anastomosis that monitors mechanical integrity for 2 weeks, triggers local drug release if abnormal signals are detected, and then dissolves harmlessly once healing is confirmed. This vision of a bioresorbable, self-reporting, self-treating theranostic implant represents the convergence of nanobiosensor technology, bioelectronics, and mechanoresponsive drug delivery that defines the frontier of the field.
3D and 4D printing for patient-specific implants and supports is poised to expand significantly. Currently, three-dimensional-printed ostomy supports, custom hernia meshes precisely shaped to a patient's anatomical defect, and bioresorbable three-dimensional printed stents for gastrointestinal anastomoses are already being used to maintain luminal patency before they resorb [127, 128]. The next step is the development of 4D-printed devices, which are designed to change their properties over time. For example, a 4D-printed anastomotic ring that is initially stiff to provide support but gradually softens after 2 weeks to avoid obstruction, and then fully resorbs by 6 weeks when healing is solid. Such smart implants could adapt to the healing stage, something not possible with static materials. In oncology, patient-tailored 3D-printed scaffolds might be used after tumor resection to both replace tissue and deliver localized therapy, for example, a printed patch for the abdominal wall that releases chemotherapy to prevent peritoneal metastases, while mechanically reinforcing the wall.
All these integrations are directed towards a single fundamental goal: precision and personalization. Mechanomedicine provides the data framework and therapeutic targets, such as maintaining tension below a specified threshold and stiffness above a minimum value, while technologies including artificial intelligence and robotics provide the means to achieve these targets with superhuman consistency. As these tools mature, surgeries will likely become safer, with fewer human errors, greater adaptability, and more predictable outcomes. Human oversight and clinical judgment remain essential; however, the substantial work of simultaneously monitoring numerous parameters and making micro-adjustments can be delegated to machines that excel at such tasks.
In summary, the operating room of the future might incorporate robotic arms that sense and respond to tissue properties, augmented reality for visualizing occult mechanical properties, artificial intelligence-driven copilots predicting complication risks in real time, and patient-specific implants fabricated on-demand to ensure each patient's tissue receives precisely calibrated mechanical support. Early versions of these technologies already exist, some in research phases and others in early clinical trials [155, 156]. The integration of mechanomedicine with such technologies will drive a new era of “digital mechanosurgery”, where data-driven and automated systems work alongside surgeons for optimal outcomes.
6.2 Standardizing mechanical metrics and guidelines
One major challenge for bringing mechanomedicine fully into routine practice is the lack of standardized reference values and guidelines. Unlike blood pressure or blood glucose, where well-defined normal ranges and treatment thresholds are available, mechanical parameters in surgery, such as normal liver stiffness for a 60-year-old patient or safe anastomotic tension ranges, are not yet universally established [12, 159]. This hampers comparability and broad adoption.
For example, different elastography machines might report slightly different absolute stiffness values for the same liver. A stiffness of 10 kPa on one device might correspond to 8 kPa on another due to calibration differences. Thus, a key step is creating cross-platform calibration standards. Phantom materials mimicking tissue stiffness need to be standardized so that MRE at Hospital A and SWE at Hospital B can both calibrate to the same reference. Organizations could then publish reference stiffness ranges for various organs stratified by factors such as age and body mass index, since even normal stiffness might vary with these parameters. Indeed, a global consortium on elastography has been called for, analogous to an internationally harmonized stiffness reference framework.
Similarly, for intraoperative measures, one center might aim for a DI of 3-4 in fundoplication while another uses a different device and thus different numerical targets. If standardized protocols are universally adopted, such as a standard EndoFLIP protocol involving 40 mL fill volume and measurement of DI at fundoplication closure, then guidelines can recommend ensuring fundoplication DI between 3-4 mm2/mmHg may eventually support formal guideline recommendations, allowing all surgeons to train to that standard. This requires professional societies to endorse these metrics and provide appropriate training.
Another area is developing clinical guidelines that incorporate mechanical metrics. For instance, an “Enhanced Recovery Mechanomedicine Protocol” could be developed to specify the following: measure IAP every 8 hours for first 3 days, if > 12 mmHg initiate such-and-such [137, 143]; measure liver stiffness preoperative in major hepatectomy, if > 15 kPa do these additional steps [21, 67]; measure colorectal anastomotic perfusion with ICG, if time-to-peak exceeds 30 s, consideration may be given to creation of a stoma [59, 162]. In the future, specialty guidelines may incorporate elastography into hepatobiliary surgical assessment and include ICG-based perfusion evaluation in colorectal surgical guidance, provided that stronger multicenter evidence becomes available [90, 109].
To build that evidence, large databases need to collect mechanical data alongside outcomes. A proposal has been made for a multilayer mechanical parameter registry collecting data at the organ level, such as stiffness of liver and intestine in various conditions; at the pathology level, tracking how stiffness changes from normal to mild fibrosis to severe fibrosis; and at the healing phase level, tracking anastomotic tension over time and correlating with leak occurrence [71, 104]. Such datasets could support the development of quantitative risk models relating anastomotic tension to leak probability.
Another need is defining a “mechanical dose” in surgery, analogous to radiation dose in radiotherapy [12, 22]. At present, anastomotic tension is often described qualitatively rather than quantified using standardized mechanical dose metrics [23, 43]. Some have proposed quantifying the energy density at the anastomosis, potentially as the integral of stress-strain over time, as a dose metric [22, 45, 64]. Definition of a threshold mechanical dose would allow clinical anastomoses to be maintained below that level. This remains challenging because tissues exhibit nonlinear, patient-specific behavior, but this concept might see development, especially as sensors provide more real-time data to calculate such values.
Finally, training and culture is a part of standardization. Surgeons will need to become as comfortable with reading an elastogram or interpreting a sensor readout as they are with reading an electrocardiogram [159, 163]. That means integrating this education into surgical training curricula. The more data and guidelines emphasize these, the more surgeons will adopt them. It's analogous to the adoption of laparoscopy, at first some resisted, but once guidelines and training incorporated it, it became second nature.
In summary, to fully leverage mechanomedicine, a common language and standardized benchmarks are needed: reference ranges for mechanical measures, agreed protocols for measurement, and guidelines for acting on those measures. Given the importance of this issue, it is anticipated that, over the next 5 years, surgical societies and regulatory bodies such as the FDA will increasingly mandate reporting of certain mechanical metrics in device trials or surgical audits, which will naturally promote standardization. Early movements include the FDA's recent guidance on brain compliance monitors and the European consensus on SWE for liver disease, which defined cutoff values for fibrosis stages [164, 165]. As these accumulate, mechanomedicine will move from interesting research to everyday practice.
6.3 Addressing foundational scientific questions
While applying current knowledge is crucial, there remain fundamental scientific questions in mechanomedicine that require further research. Addressing these questions will reduce uncertainty and facilitate further progress in the field [12, 37].
Inverse problems in biomechanics represent a significant challenge in determining how accurately true tissue properties can be inferred from measurements such as elastography waves [44, 166]. For example, MRE inversion algorithms attempt to compute the shear modulus from observed wave fields [167, 168]. But under conditions of limited field of view or noise, there are identifiability limits, meaning that there are thresholds beyond which a given measurement becomes unreliable in distinguishing a change in stiffness vs. an artifact [169, 170]. Research here overlaps with applied math and physics: establishing error bounds and robust inversion methods [166]. Such uncertainty limits reproducibility across platforms and remains a major barrier to clinical standardization. Machine learning-based inversion also introduces challenges related to interpretability, generalization, and training-data bias. Recent work has improved inversion algorithms by incorporating machine learning, essentially training on simulated wave data to better decode overlapping waves in small organs such as the pancreas. However, more work is needed, and multi-modality approaches combining elastography with imaging constraints will likely prove beneficial.
Another fundamental area is multi-scale mapping: linking micro-scale properties, such as ECM fiber organization, to macro-scale properties, such as organ compliance. It is well established that collagen crosslinking increases stiffness qualitatively, but predictive models are desirable: for instance, given a biopsy with specific collagen percentage and crosslink density, one could predict liver stiffness and portal pressure for that patient. Some progress has been made using micro-computed tomography of resected tissues to model ECM networks; recent studies have shown a direct correlation between collagen fiber alignment in intestinal strictures and the degree of nonlinearity in their stress-strain response [51, 56]. Eventually, an “ECM fingerprint” may become available to indicate which helps in creating personalized models without having to individually measure everything in vivo.
A highly intriguing concept involves defining an “anastomotic mechanical dose”, analogous to radiation dose definition in radiation oncology. Surgeons might quantify how much mechanical stress an anastomosis endures over time as its dose, which correlates with healing or complication rates. For instance, one could integrate the tension over each day for the first postoperative week using some function of magnitude and duration. One possibility is that leakage risk increases once a threshold dose is exceeded. Early porcine studies have attempted to relate time under tension to leak occurrence, although the concept remains incompletely defined. This likely requires a time-dependent model of tissue strength gain versus applied stress, essentially a dynamic fracture mechanics model for living tissue. Answering this question requires both computational modeling and animal studies in which the mechanical microenvironment is tightly controlled to observe when failure occurs. Solving this would represent a paradigm shift: surgeons could then aim to keep mechanical dose below a certain level through rest, dietary management, supportive measures, and other interventions until tissue strength adequately increases, representing a highly quantitative approach to the principle of “don't stress the anastomosis too much too soon.”
Another challenge involves maintaining stability and robustness in closed-loop control during surgery. If such a system is implemented, care must be taken to avoid over-correction or oscillation, which are classic control theory issues. Additionally, considering the human element, surgeons must trust and effectively interact with these systems. Research analogous to autopilot systems in aviation is needed: developing fail-safes, alarm hierarchies determining when the machine versus the human takes control, and proving that these closed-loop systems actually improve outcomes. Early closed-loop trials exist in anesthesia, such as closed-loop propofol infusion controlling bispectral index brain monitoring. Closed-loop fluid management may emerge to maintain optimal abdominal compliance or closed-loop insufflator systems adjusting pressure to maintain target indocyanine green perfusion metrics. However, rigorous trials are needed to demonstrate safety and benefit, and those are on the horizon.
Finally, broad adoption will also depend on demonstrating cost-effectiveness and training surgeons in these new paradigms. This represents more health-services research but constitutes a necessary component of answering the question: is mechanomedicine worth implementing? If it reduces complication rates, which are highly costly, the balance may ultimately prove favorable, although this will require confirmation in multicenter studies.
6.4 Data Ethics, Equity, and Responsible Implementation
The integration of AI and the Internet of Things into mechanomedicine raises important ethical and equity considerations that must be addressed alongside technological development. First, data privacy and governance: continuous biosensors, wearable devices, and AI-driven monitoring systems generate large volumes of sensitive patient data. Robust, transparent data governance frameworks—including informed consent protocols, data anonymization standards, and secure storage architectures—are essential to protect patient privacy and maintain public trust. Second, algorithmic bias and generalizability: AI models trained predominantly on data from high-resource settings or specific demographic groups may perform poorly when applied to diverse populations. Future research must rigorously evaluate the generalizability and fairness of algorithmic models across different ethnicities, ages, body habitus, and healthcare settings, and actively work to mitigate bias in training datasets. Third, healthcare equity: advanced mechanomedicine technologies, including high-field MRE systems, robotic surgical platforms, and implantable biosensors, are currently concentrated in well-resourced academic centers. Without deliberate efforts to reduce cost, simplify deployment, and expand access, these innovations risk exacerbating existing healthcare inequalities between high- and low-income settings. Responsible implementation of mechanomedicine therefore requires not only technical validation and clinical translation, but also a commitment to equitable access, transparent algorithmic accountability, and inclusive research design.
In summary, future progress in mechanomedicine will rely on sustained interdisciplinary collaboration among surgeons, engineers, biologists, data scientists, and other relevant specialists. Addressing current challenges will require both technical innovation and effective integration across disciplines. Over the coming decade, several emerging concepts may move toward routine clinical application, supported by growing evidence and increasing clinical interest. The long-term goal is a model of digestive surgery in which clinical decisions are guided by a deeper understanding of tissue mechanics, perioperative care is personalized through mechanical profiling, and adverse outcomes are reduced through more precise intervention. Figure 6 summarizes the major scientific challenges and outlines a translational roadmap for mechanomedicine in digestive surgery, including sensing-modeling integration, standardization, and closed-loop control strategies.
Future scientific challenges and translational roadmap for mechanomedicine in digestive surgery. This figure outlines a forward-looking framework linking sensing technologies, biomechanical modeling, standardization efforts and control strategies to enable precision mechanomedicine in digestive surgery. (A) Integrating sensing, modeling and artificial intelligence for precision mechanosurgery. The technological trajectory of mechanomedicine involves three tightly coupled layers: (1) miniaturized, biocompatible sensing and imaging modalities, including elastography, functional lumen imaging, implantable tension and pressure sensors, and wearable abdominal patches, which provide high-frequency, quantitative measurements of tissue stiffness, tension and pressure in vivo [144, 146]; (2) data-fusion and modeling engines that integrate physics-based finite-element (FE) simulations with machine learning algorithms and multimodal data streams to infer latent mechanical states, predict patient-specific risk and support surgical decision-making [85, 105]; (3) clinical interfaces, such as robotic surgical consoles, augmented reality overlays and bedside dashboards, that deliver mechanorelevant information to surgeons and clinicians in real time and enable adaptive, personalized intervention strategies [55]. (B) Standardizing mechanical metrics and clinical practice across centers. For mechanomedicine to transition from research to routine care, mechanical parameters must be harmonized across institutions, devices and vendors. This requires: the establishment of multicentre registries of organ and lesion stiffness, stratified by age, sex, body-mass index and disease stage [168]; phantom-based calibration frameworks for elastography, EndoFLIP and related technologies to ensure cross-platform and cross-vendor comparability [165]; and the incorporation of validated mechanical thresholds—such as liver stiffness cut-offs for hepatectomy, distensibility index windows for fundoplication, or intra-abdominal pressure (IAP) triggers for decompression—into international clinical guidelines and consensus statements [159]. (C) Open scientific questions in mechanomedicine. Key unresolved challenges include: defining the identifiability limits of shear modulus (μ) and elastic modulus (E) from noisy and incomplete wave-field data in organs with heterogeneous composition and complex geometry [166]; establishing a quantitative, multiscale mapping from extracellular-matrix architecture and cellular organization to organ-level compliance and distensibility indices, enabling mechanistic interpretation of clinical cut-offs [63]; and formulating a clinically meaningful concept of an anastomotic “mechanical dose”, for example as time-integrated stress, strain or strain-energy density, and linking it to outcomes such as leak, fibrosis and stricture formation [69]. (D) Closed-loop control systems for real-time surgical optimization. Building on standardized metrics and mechanistic models, closed-loop control architectures can be developed in which intraoperative mechanical measurements (such as tissue tension, distensibility index, IAP and perfusion metrics derived from indocyanine green imaging) are continuously fed into a controller that adjusts surgical parameters—including tension, perfusion optimization and decompression strategies—in real time. AI-driven predictive guidance layers can further enhance robustness and personalization, enabling stable, adaptive control across heterogeneous patient populations and clinical settings. Together, addressing these scientific, technological and translational challenges—alongside parallel regulatory, ethical and educational initiatives—will be essential to evolve mechanomedicine from promising prototypes into a mature, globally adopted pillar of digestive surgery [155, 164]. The figure was created by Figdraw (www.figdraw.com).
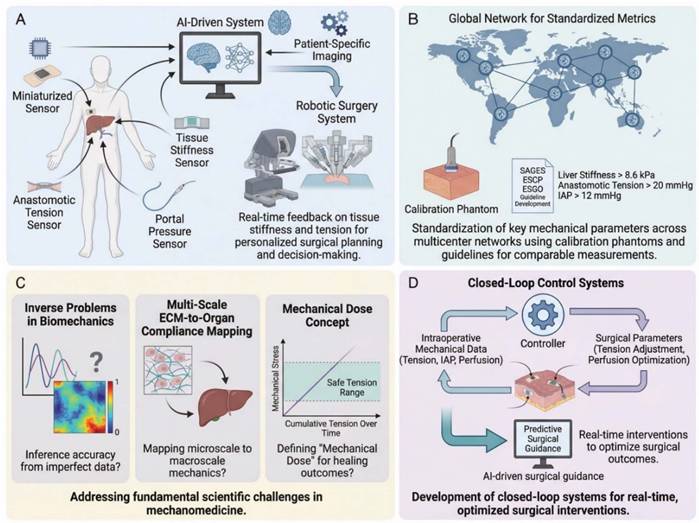
7. Conclusions
Mechanomedicine is reshaping digestive surgery by bringing the mechanical microenvironment into the center of perioperative care. Mechanical parameters such as tissue stiffness, anastomotic tension, perfusion-related forces, and intra-abdominal pressure are no longer merely descriptive features but increasingly actionable variables for risk stratification, surgical guidance, and postoperative monitoring.
This review highlights a unifying concept: across the perioperative continuum, mechanical information can link diagnosis with intervention and thereby support a more quantitative and individualized surgical strategy. Preoperative profiling, intraoperative mechanosensing, and postoperative mechanotherapy together outline a framework in which surgical outcomes may be improved not only by anatomical precision, but also by better control of tissue mechanics during healing and remodeling.
At present, mechanomedicine remains an evolving field. Some tools have already shown clinical utility, whereas others still require further validation, standardization, and integration into routine workflows. Nevertheless, the overall direction is clear: incorporating biomechanics into digestive surgery offers a promising path toward more predictive, precise, and personalized care.
Abbreviations
2D-SWE: two-dimensional shear wave elastography; ACS: abdominal compartment syndrome; AI: artificial intelligence; ASBO: adhesive small-bowel obstruction; AUC: area under the curve; BP: burst pressure; bFGF: basic fibroblast growth factor; CFD: computational fluid dynamics; CRLM: colorectal liver metastases; CT: computed tomography; DI: distensibility index; ECM: extracellular matrix; EndoFLIP: endoscopic functional lumen imaging probe; ERAS: enhanced recovery after surgery; FAK: focal adhesion kinase; FE: finite element; FDA: U.S. Food and Drug Administration; HCC: hepatocellular carcinoma; IAH: intra-abdominal hypertension; IAP: intra-abdominal pressure; ICG: indocyanine green; LS: liver stiffness; MCI: mechanical compatibility index; MMP: matrix metalloproteinase; MRE: magnetic resonance elastography; MRI: magnetic resonance imaging; NO: nitric oxide; NPO: nil-by-mouth; PCL: poly-ε-caprolactone; POPF: postoperative pancreatic fistula; PSCs: pancreatic stellate cells; PVP: portal venous pressure; ROC: receiver operating characteristic; ROCK: Rho-associated kinase; SWE: shear-wave elastography; SWV: shear-wave velocity; TGF-β: transforming growth factor-beta; TRPV4: transient receptor potential vanilloid 4; VAC: vacuum-assisted closure; VEGF: vascular endothelial growth factor; YAP: Yes-associated protein; TAZ: transcriptional co-activator with PDZ-binding motif; Piezo1: piezo type mechanosensitive ion channel component 1.
Acknowledgements
AI Tool Disclosure: During the preparation of this work, the authors used ChatGPT-4 (OpenAI) and DeepSeek-V3 (DeepSeek) for English language polishing and preliminary organization of literature information. After using these tools, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication. No AI tools were used for data collection, data analysis, image generation, or decision-making regarding the study design or conclusions.
Scientific Illustrations and Copyright: All schematic and mechanism diagrams in this manuscript were created using the professional scientific illustration platform Figdraw (www.figdraw.com). The authors have obtained legitimate copyright authorization for all illustrations. The corresponding authorization IDs are as follows: Figure 1: ITUSUda674; Figure 2: TPOTP8c88d; Figure 3: YTTIYdddfd; Figure 4: ISWTY88888; Figure 5: OSIYT117d1; Figure 6: OTAOY304f3.
Funding
This study was supported by the National Natural Science Foundation of China (12225208, 82560470), Academic Enhancement Support Program of Hainan Medical University (XSTS2025063), and the Innovation Platform for Academicians of Hainan Province (YSPTZX202005).
Authorship contributions
Ning Liu: Conceptualization, Writing - Original Draft, Writing - Review & Editing, Project administration, Supervision. Xiang Wang: Writing - Original Draft, Writing - Review & Editing, Visualization. Na Liu: Investigation, Resources, Data Curation. Feng Xu: Writing - Review & Editing, Supervision, Funding acquisition. Ting Wen: Conceptualization, Writing - Review & Editing, Supervision, Funding acquisition.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bruna CL, Emmen A, Wei K, Sutcliffe RP, Shen B, Fusai GK. et al. Effects of Pancreatic Fistula After Minimally Invasive and Open Pancreatoduodenectomy. JAMA Surg. 2025;160:190-8
2. Weber MC, Berlet M, Stoess C, Reischl S, Wilhelm D, Friess H. et al. A nationwide population-based study on the clinical and economic burden of anastomotic leakage in colorectal surgery. Langenbecks Arch Surg. 2023;408:55
3. Voldby AW, Boolsen AW, Aaen AA, Burcharth J, Ekeløf S, Loprete R. et al. Complications and Their Association with Mortality Following Emergency Gastrointestinal Surgery-an Observational Study. J Gastrointest Surg. 2022;26:1930-41
4. Walshaw J, Huo B, McClean A, Gajos S, Kwan JY, Tomlinson J. et al. Innovation in gastrointestinal surgery: the evolution of minimally invasive surgery-a narrative review. Front Surg. 2023;10:1193486
5. Ye SP, Zhu WQ, Huang ZX, Liu DN, Wen XQ, Li TY. Role of minimally invasive techniques in gastrointestinal surgery: Current status and future perspectives. World J Gastrointest Surg. 2021;13:941-52
6. Rudiman R. Minimally invasive gastrointestinal surgery: From past to the future. Ann Med Surg (Lond). 2021;71:102922
7. Gielen AHC, Heuvelings DJI, Sylla P, van Loon YT, Melenhorst J, Bouvy ND. et al. Impact of Anastomotic Leakage After Colorectal Cancer Surgery on Quality of Life: A Systematic Review. Dis Colon Rectum. 2025;68:154-70
8. de Wit A, Bootsma BT, Huisman DE, van Wely B, van Hoogstraten J, Sonneveld DJA. et al. Risk Factor Targeted Perioperative Care Reduces Anastomotic Leakage after Colorectal Surgery: The DoubleCheck study. Ann Surg. 2024;283:154-61
9. Liu Y, Li B, Wei Y. New understanding of gut microbiota and colorectal anastomosis leak: A collaborative review of the current concepts. Front Cell Infect Microbiol. 2022;12:1022603
10. Alverdy JC, Schardey HM. Anastomotic Leak: Toward an Understanding of Its Root Causes. J Gastrointest Surg. 2021;25:2966-75
11. Shi N, Ma Y, Liu N, Lv Y, Xu F. Mechanomedicine in anastomosis: Bridging biomechanics and clinical outcomes for improved surgical healing. Phys Life Rev. 2025;55:123-41
12. Durcan C, Hossain M, Chagnon G, Perić D, Girard E. Mechanical experimentation of the gastrointestinal tract: a systematic review. Biomech Model Mechanobiol. 2024;23:23-59
13. Almet AA, Byrne HM, Maini PK, Moulton DE. The role of mechanics in the growth and homeostasis of the intestinal crypt. Biomech Model Mechanobiol. 2021;20:585-608
14. Wang Z, Xie N, Liang X, Shu Q, Hong Y, Shi H. et al. Gut mechanoimmunology: Shaping immune response through physical cues. Phys Life Rev. 2024;50:13-26
15. Xiao C, Xie N, Shu Q, Liang X, Wang Z, Wu J. et al. Synergistic Effects of Matrix Biophysical Properties on Gastric Cancer Cell Behavior via Integrin-Mediated Cell-ECM Interactions. Small. 2024;20:e2309907
16. Donmazov S, Saruhan EN, Pekkan K, Piskin S. Review of Machine Learning Techniques in Soft Tissue Biomechanics and Biomaterials. Cardiovasc Eng Technol. 2024;15:522-49
17. Yang G, Shen H, Jang Y, Cheng X. Finite element analysis-assisted surgical planning and evaluation of flap design in hand surgery. Front Bioeng Biotechnol. 2025;13:1611993
18. Li N, Zhang X, Zhou J, Li W, Shu X, Wu Y. et al. Multiscale biomechanics and mechanotransduction from liver fibrosis to cancer. Adv Drug Deliv Rev. 2022;188:114448
19. von Ehrlich-Treuenstätt VH, Guenther M, Ilmer M, Knoblauch MM, Koch D, Clevert D-A. et al. Preoperative ultrasound elastography for postoperative pancreatic fistula prediction after pancreatoduodenectomy: A prospective study. Surgery. 2024;175:491-7
20. Dal Buono A, Faita F, Peyrin-Biroulet L, Danese S, Allocca M. Ultrasound Elastography in Inflammatory Bowel Diseases: A Systematic Review of Accuracy Compared with Histopathological Assessment. J Crohns Colitis. 2022;16:1637-46
21. Lee DH, Lee ES, Bae JS, Lee JY, Han JK, Yi NJ. et al. 2D shear wave elastography is better than transient elastography in predicting post-hepatectomy complication after resection. Eur Radiol. 2021;31:5802-11
22. Zhou Y, Tan Z, Jiang P, Sun Y, Wu D. Evaluation of intestinal tissue safety during the compression process of circular end-to-end anastomosis stapler based on finite element simulation. Front Bioeng Biotechnol. 2025;13:1594969
23. Cira K, Janett SN, Micheler C, Heller S, Obermeier A, Friess H. et al. The mesenteric entry site as a potential weak point in gastrointestinal anastomoses - findings from an ex-vivo biomechanical analysis. Langenbecks Arch Surg. 2024;409:124
24. Cheng B, Li M, Lin M, Guo H, Xu F. Mechanobiology across timescales. Nature Reviews Physics. 2025;7:621-44
25. Kola JB, Docsa T, Uray K. Mechanosensing in the Physiology and Pathology of the Gastrointestinal Tract. Int J Mol Sci. 2022;24:177
26. Kimura S, Tsuji T. Mechanical and Immunological Regulation in Wound Healing and Skin Reconstruction. Int J Mol Sci. 2021;22:5474
27. Qiu J, Yu B, Ren C, Wang T, Zhang G, Jian Z. et al. Deep-supercooling preservation of stem cell spheroids for chondral defect repairment. Stem Cell Reports. 2024;19:1665-76
28. Wilson DF, Matschinsky FM. Metabolic Homeostasis in Life as We Know It: Its Origin and Thermodynamic Basis. Frontiers in Physiology. 2021;12:658997
29. Dutta A, Chattopadhyay H. A Brief On Biological Thermodynamics for Human Physiology. Journal of Biomechanical Engineering. 2021;143:070802
30. Zuo D, He Y, Avril S, Yang H, Hackl K. A thermodynamic framework for unified continuum models for the healing of damaged soft biological tissue. Journal of the Mechanics and Physics of Solids. 2022;158:104662
31. Kalukula Y, Ciccone G, Mohammed D, Procès A, Versaevel M, Deridoux A. et al. Unlocking the therapeutic potential of cellular mechanobiology. Sci Adv. 2025;11:eaea6817
32. O'Neill GM, Zhu C, Kim DH, Shin J. Mechanomedicine: Translating mechanical forces into therapeutic strategies. APL Bioeng. 2025;9:040401
33. Xin Y, Li K, Huang M, Liang C, Siemann D, Wu L. et al. Biophysics in tumor growth and progression: from single mechano-sensitive molecules to mechanomedicine. Oncogene. 2023;42:3457-90
34. Carniel EL, Albanese A, Fontanella CG, Pavan PG, Prevedello L, Salmaso C. et al. Biomechanics of stomach tissues and structure in patients with obesity. J Mech Behav Biomed Mater. 2020;110:103883
35. Xie N, Tian J, Li Z, Shi N, Li B, Cheng B. et al. Invited Review for 20th Anniversary Special Issue of PLRev "AI for Mechanomedicine". Phys Life Rev. 2024;51:328-42
36. Toniolo I, Fontanella CG, Foletto M, Carniel EL. Biomechanical Investigation of the Stomach Following Different Bariatric Surgery Approaches. Bioengineering (Basel). 2020;7:159
37. Di X, Gao X, Peng L, Ai J, Jin X, Qi S. et al. Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal Transduction and Targeted Therapy. 2023;8:282
38. Hartog F, Yurtkap Y, Vlot J, Lange JF, Tanis PJ, Kleinrensink GJ. Developing and validating an implantable suture tension sensor. Heliyon. 2024;10:e28907
39. Xu WX, Zhong QH, Cai Y, Zhan CH, Chen S, Wang H. et al. Comprehensively evaluate the short outcome of small bowel obstruction: A novel medical-economic score system. World J Gastroenterol. 2023;29:1509-22
40. Wu H, Attaar M, Wong HJ, Campbell M, Kuchta K, Denham EW 3rd. et al. Impedance Planimetry (Endoflip) and Ideal Distensibility Ranges for Optimal Outcomes after Nissen and Toupet Fundoplication. J Am Coll Surg. 2022;235:420-9
41. Ryu S, Hara K, Goto K, Okamoto A, Kitagawa T, Marukuchi R. et al. Fluorescence angiography vs. direct palpation for bowel viability evaluation with strangulated bowel obstruction. Langenbecks Arch Surg. 2022;407:797-803
42. Zhang X, Gao Y, Huo B. Fluid-Solid Coupling Simulation of Wall Fluid Shear Stress on Cells under Gradient Fluid Flow. Appl Bionics Biomech. 2021;2021:8340201
43. McKinley TMR, Chow D, Livezey JB, Anklowitz A, Williams TM, Kippenberger T. et al. Impact of Surgical Experience on Burst Pressure of Hand-Sewn and Stapled Anastomoses. J Surg Res. 2025;310:122-7
44. Ayyalasomayajula V, Ervik Ø, Sorger H, Skallerud B. Macro-indentation testing of soft biological materials and assessment of hyper-elastic material models from inverse finite element analysis. J Mech Behav Biomed Mater. 2024;151:106389
45. Uzunlu O, Aydin E, Çomut E, Avcı E, Şenol H. The comparison of the suture materials on intestinal anastomotic healing: an experimental study. Ulus Travma Acil Cerrahi Derg. 2023;29:956-62
46. Tian XF, Zhang L, Lou WH, Qiu YJ, Zuo D, Wang WP. et al. Application of ultrasound shear wave elastography in pre-operative and quantitative prediction of clinically relevant post-operative pancreatic fistula after pancreatectomy: a prospective study for the investigation of risk evaluation model. Eur Radiol. 2023;33:7866-76
47. Wada Y, Aoki T, Fujimori A, Ohike N, Koizumi T, Kusano T. et al. Intraoperative Shear Wave Elastography as a Quantitative Predictor of Pancreatic Fibrosis and Exocrine Function. Anticancer Res. 2021;41:1013-9
48. Wu D, Huang Y, Lei M, Zhao Z, Guo X, Fang D. Stiffness and toughness of soft/stiff suture joints in biological composites. Applied Mathematics and Mechanics. 2022;43:1469-84
49. Lindner M, Laporte A, Block S, Elomaa L, Weinhart M. Physiological Shear Stress Enhances Differentiation, Mucus-Formation and Structural 3D Organization of Intestinal Epithelial Cells In Vitro. Cells. 2021;10:2062
50. Fois CAM, Schindeler A, Valtchev P, Dehghani F. Dynamic flow and shear stress as key parameters for intestinal cells morphology and polarization in an organ-on-a-chip model. Biomed Microdevices. 2021;23:55
51. He S, Lei P, Kang W, Cheung P, Xu T, Mana M. et al. Stiffness Restricts the Stemness of the Intestinal Stem Cells and Skews Their Differentiation Toward Goblet Cells. Gastroenterology. 2023;164:1137-51.e15
52. Hu J, Jia Y, Feng F, Zhao Q, Peng B, Wang X. et al. Transforming destructive mechanical cues into therapeutic power: Activation of PIEZO1 and TRPV4 counteracts mechano-induced damage of cellular junctions in Hailey-Hailey disease. J Adv Res. 2025;1232:00951-8
53. Amundson JR, Kuchta K, Zimmermann CJ, VanDruff VN, Joseph S, Che S. et al. Target distensibility index on impedance planimetry during fundoplication by choice of wrap and choice of bougie. Surg Endosc. 2023;37:8670-81
54. Montalvo S, Gomez M, Lozano A, Arias S, Rodriguez L, Morales-Acuna F. et al. Differences in Blood Flow Patterns and Endothelial Shear Stress at the Carotid Artery Using Different Exercise Modalities and Intensities. Front Physiol. 2022;13:857816
55. Gates PE, Gurung A, Mazzaro L, Aizawa K, Elyas S, Strain WD. et al. Measurement of Wall Shear Stress Exerted by Flowing Blood in the Human Carotid Artery: Ultrasound Doppler Velocimetry and Echo Particle Image Velocimetry. Ultrasound Med Biol. 2018;44:1392-401
56. Yavuz A, Pehlevan-Özel H, Tez M. Pathophysiology of anastomotic stricture following rectal anastomosis: Insights into mechanisms, risk factors, and preventive strategies. World J Gastrointest Pathophysiol. 2025;16:107492
57. Tithof J, Pruett TL, Rao JS. Lumped parameter liver simulation to predict acute haemodynamic alterations following partial resections. J R Soc Interface. 2023;20:20230444
58. Liu H, Sun M, Wu N, Liu B, Liu Q, Fan X. TGF-β/Smads signaling pathway, Hippo-YAP/TAZ signaling pathway, and VEGF: Their mechanisms and roles in vascular remodeling related diseases. Immun Inflamm Dis. 2023;11:e1060
59. Depalma N, D'Ugo S, Manoochehri F, Libia A, Sergi W, Marchese TRL. et al. NIR ICG-Enhanced Fluorescence: A Quantitative Evaluation of Bowel Microperfusion and Its Relation to Central Perfusion in Colorectal Surgery. Cancers (Basel). 2023;15:5528
60. Nakashima K, Ryu S, Okamoto A, Hara K, Ishida K, Ito R. et al. Usefulness of blood flow evaluation with indocyanine green fluorescence imaging during laparoscopic surgery for strangulated bowel obstruction: A cohort study. Asian J Surg. 2022;45:867-73
61. Plekhanov AA, Kozlov DS, Shepeleva AA, Kiseleva EB, Shimolina LE, Druzhkova IN. et al. Tissue Elasticity as a Diagnostic Marker of Molecular Mutations in Morphologically Heterogeneous Colorectal Cancer. Int J Mol Sci. 2024;25:5337
62. Skrip L-M, Moosburner S, Tang P, Guo J, Görner S, Tzschätzsch H. et al. Viscoelastic properties of colorectal liver metastases reflect tumour cell viability. Journal of Translational Medicine. 2024;22:774
63. Wu W, Daneker M, Turner KT, Jolley MA, Lu L. Identifying heterogeneous micromechanical properties of biological tissues via physics-informed neural networks. ArXiv. 2024;9:e2400620
64. Godo ZA, Fazekas LA, Fritsch G, Szabo B, Nemeth N. A Custom-Developed Device for Testing Tensile Strength and Elasticity of Vascular and Intestinal Tissue Samples for Anastomosis Regeneration Research. Sensors (Basel). 2024;24:5984
65. Elgohary DH, Saad MA, Salem MM, Sherazy EH, Khalifa TF. Assessment the properties of various surgical sutures. Sci Rep. 2025;15:33330
66. Murayama A, Yoneda H, Maehara A, Shiomi N, Hirata H. A highly elastic absorbable monofilament suture fabricated from poly(3-hydroxybutyrate-co-4-hydroxybutyrate). Sci Rep. 2023;13:3275
67. Long H, Zhong X, Su L, Huang T, Duan Y, Ke W. et al. Liver Stiffness Measured by Two-Dimensional Shear Wave Elastography for Predicting Symptomatic Post-hepatectomy Liver Failure in Patients with Hepatocellular Carcinoma. Ann Surg Oncol. 2022;29:327-36
68. Long H, Xu W, Zhong X, Chen Z, Su L, Duan Y. et al. Feasibility of liver stiffness measured using two-dimensional shear wave elastography in assessing preoperative liver function for patients with hepatocellular carcinoma. Abdom Radiol (NY). 2022;47:664-71
69. Cong J, Zhang H, Chen C. Definition and grading of anastomotic stricture/stenosis following low anastomosis after total mesorectal excision: A single-center study. Asian J Surg. 2023;46:3722-6
70. Liu D, Chen J, Zhang Y, Dai Y, Yao X. Magnetic resonance elastography-derived stiffness: potential imaging biomarker for differentiation of benign and malignant pancreatic masses. Abdom Radiol (NY). 2023;48:2604-14
71. Long Y, Niu Y, Liang K, Du Y. Mechanical communication in fibrosis progression. Trends Cell Biol. 2022;32:70-90
72. Saraswathibhatla A, Indana D, Chaudhuri O. Cell-extracellular matrix mechanotransduction in 3D. Nat Rev Mol Cell Biol. 2023;24:495-516
73. Hadzipasic M, Sten MS, Massaad E, Kiapour A, Connolly ID, Esposito E. et al. ROCK-dependent mechanotransduction of macroscale forces drives fibrosis in degenerative spinal disease. Nat Biomed Eng. 2025;9:1677-90
74. Berry CE, Downer M Jr, Morgan AG, Griffin M, Liang NE, Kameni L. et al. The effects of mechanical force on fibroblast behavior in cutaneous injury. Front Surg. 2023;10:1167067
75. Wu K, Li J, Fu Y, Yang K, Lin T, Wang Y. et al. Tumor peripheral stiffness modulates lenvatinib resistance in HCC preclinical models by regulating FIS1-dependent mitophagy. JHEP Rep. 2025;7:101588
76. Kim OH, Choi YW, Park JH, Hong SA, Hong M, Chang IH. et al. Fluid shear stress facilitates prostate cancer metastasis through Piezo1-Src-YAP axis. Life Sci. 2022;308:120936
77. Shi H, Zou Y, Zhong W, Li Z, Wang X, Yin Y. et al. Complex roles of Hippo-YAP/TAZ signaling in hepatocellular carcinoma. J Cancer Res Clin Oncol. 2023;149:15311-22
78. De Belly H, Paluch EK, Chalut KJ. Interplay between mechanics and signalling in regulating cell fate. Nat Rev Mol Cell Biol. 2022;23:465-80
79. Joshi V, Strege PR, Farrugia G, Beyder A. Mechanotransduction in gastrointestinal smooth muscle cells: role of mechanosensitive ion channels. Am J Physiol Gastrointest Liver Physiol. 2021;320:G897-g906
80. Adapala RK, Katari V, Teegala LR, Thodeti S, Paruchuri S, Thodeti CK. TRPV4 Mechanotransduction in Fibrosis. Cells. 2021;10:3053
81. Tao D, Wang H, Chang S, Cheng J, Da N, Zhang L. et al. Matrix Viscoelasticity Orchestrates Osteogenesis via Mechanotransduction Mediated Metabolic Switch in Macrophages. Adv Healthc Mater. 2025;14:e2405097
82. Liu Y, Yao X, Zhao Y, Fang D, Shi L, Yang L. et al. Mechanotransduction in response to ECM stiffening impairs cGAS immune signaling in tumor cells. Cell Rep. 2023;42:113213
83. Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial Intelligence in Surgery: Promises and Perils. Ann Surg. 2018;268:70-6
84. Shin TH, Ashley SW, Tsai TC. Defining the Role of Machine Learning in Optimizing Surgical Outcomes. JAMA Surg. 2024;159:1432
85. Celotto F, Bao QR, Capelli G, Spolverato G, Gumbs AA. Machine learning and deep learning to improve prevention of anastomotic leak after rectal cancer surgery. World J Gastrointest Surg. 2025;17:101772
86. Chiba H, Sato N, Ozeki A, Sugawara R, Azuma T, Tsukida S. et al. Prognostic significance of the liver stiffness value on magnetic resonance imaging elastography in patients undergoing hepatectomy for hepatocellular carcinoma. Surg Today. 2025;55:1361-72
87. Nakazawa Y, Okada M, Tago K, Kuwabara N, Mizuno M, Abe H. et al. MR elastography vs a combination of common non-invasive tests for estimation of severe liver fibrosis in patients with hepatobiliary tumors. Eur Radiol. 2025;35:1464-72
88. Nakajima T, Ikuta S, Aihara T, Ikuta L, Matsuki G, Fujikawa M. et al. Intraoperatively measured prehepatectomy portal vein pressure as a useful predictor of posthepatectomy liver failure. Langenbecks Arch Surg. 2024;409:314
89. Yuan J, Liu J, Wen T, Wang L, Peng Z, Zhang N. et al. Added value of multifrequency magnetic resonance elastography in predicting pathological grading of pancreatic neuroendocrine neoplasms. Insights into Imaging. 2025;16:119
90. Bae JS, Lee JY, Lee DH, Han SW, Lim Y, Kim TY. Predicting the chemotherapeutic response of colorectal cancer liver metastasis using shear-wave elastography. Ultrasonography. 2023;42:457-65
91. Wang S, Li S, Chen S, Li M, Xie X, Ren M. et al. Real-time shear wave elastography in measuring normal ileocolon intestinal wall stiffness using colonoscopy as reference: A single-center research. Eur J Radiol Open. 2025;14:100632
92. Al Asadi H, Najah H, Edelmuth R, Greenberg JA, Marshall T, Salehi N. et al. Impedance planimetry (EndoFLIPTM) and surgical outcomes after Hill compared to Toupet fundoplication. Surg Endosc. 2024;38:1020-8
93. Charbonneau J, Papillon-Dion É, Brière R, Singbo N, Legault-Dupuis A, Drolet S. et al. Fluorescence angiography with indocyanine green for low anterior resection in patients with rectal cancer: a prospective before and after study. Tech Coloproctol. 2025;29:45
94. Hu C, Zhang J. Simulation and Experimental Study of Staple Line Reinforcement Surgery. Journal of Biomedical Science and Engineering. 2024;17:83-95
95. Taha-Mehlitz S, Wentzler L, Angehrn F, Hendie A, Ochs V, Wolleb J. et al. Machine learning-based preoperative analytics for the prediction of anastomotic leakage in colorectal surgery: a swiss pilot study. Surg Endosc. 2024;38:3672-83
96. Yang X, Dou F, Tang G, Xiu R, Zhao X. Interpretable machine learning model for predicting anastomotic leak after esophageal cancer surgery via LightGBM. BMC Cancer. 2025;25:976
97. Mazier A, Bordas SPA. Breast simulation pipeline: From medical imaging to patient-specific simulations. Clin Biomech (Bristol). 2024;111:106153
98. Peng L, Yu L, Jia J, Gan Y, Ding A, Xiong P. et al. The effect of thickness and elastic modulus of the anterior talofibular ligament on anterior ankle joint stiffness: A subject-specific finite element study. Front Bioeng Biotechnol. 2023;11:1175347
99. Huang ZH, Wang LK, Cai SY, Chen HX, Zhou Y, Cheng LK. et al. Palm-Sized Wireless Transient Elastography System with Real-Time B-Mode Ultrasound Imaging Guidance: Toward Point-of-Care Liver Fibrosis Assessment. Diagnostics (Basel). 2024;14:0
100. Pinton P. Computational models in inflammatory bowel disease. Clin Transl Sci. 2022;15:824-30
101. Zhang X, Zhang W, Sun W, Song A, Xu T. A high-fidelity virtual liver model incorporating biological characteristics. Heliyon. 2023;9:e22978
102. Toniolo I, Fontanella CG, Foletto M, Carniel EL. Coupled experimental and computational approach to stomach biomechanics: Towards a validated characterization of gastric tissues mechanical properties. J Mech Behav Biomed Mater. 2022;125:104914
103. Sun Y, Pan B, Fu Y. Lightweight Deep Neural Network for Articulated Joint Detection of Surgical Instrument in Minimally Invasive Surgical Robot. J Digit Imaging. 2022;35:923-37
104. Kobayashi T, Iwaki M, Nogami A, Kawamura N, Honda Y, Ogawa Y. et al. Prediction of outcomes in patients with metabolic dysfunction-associated steatotic liver disease based on initial measurements and subsequent changes in magnetic resonance elastography. J Gastroenterol. 2024;59:56-65
105. Mascarenhas M, Mendes F, Fonseca F, Carvalho E, Santos A, Cavadas D. et al. A Novel Deep Learning Model for Predicting Colorectal Anastomotic Leakage: A Pioneer Multicenter Transatlantic Study. J Clin Med. 2025;14:5462
106. Litchinko A, Buchs N, Balaphas A, Toso C, Liot E, Meurette G. et al. Score prediction of anastomotic leak in colorectal surgery: a systematic review. Surg Endosc. 2024;38:1723-30
107. Knowles TB, Jackson AS, Chang SC, Schembre DB, Farivar AS, Aye RW. et al. Changes in Distensibility Index During an Incremental POEM Myotomy. J Gastrointest Surg. 2022;26:1140-6
108. Rudnicki Y, White I, Tiomkin V, Lahav L, Raguan B, Avital S. Intraoperative evaluation of colorectal anastomotic integrity: a comparison of air leak and dye leak tests. Tech Coloproctol. 2021;25:841-7
109. Faber RA, Meijer RPJ, Droogh DHM, Jongbloed JJ, Bijlstra OD, Boersma F. et al. Indocyanine green near-infrared fluorescence bowel perfusion assessment to prevent anastomotic leakage in minimally invasive colorectal surgery (AVOID): a multicentre, randomised, controlled, phase 3 trial. The Lancet Gastroenterology & Hepatology. 2024;9:924-34
110. He W, Li Z, Deng X, Zheng K, Wang C, Peng L. et al. Fluorescence quantitative assessment of blood perfusion in the gastric conduit to reduce anastomotic leakage after esophagectomy: a randomized controlled trial. Surg Endosc. 2025;39:6644-54
111. Lu D, Hao Y, Wang Z, He J, Huang X, Shi Y. et al. Zn-PAA-C hydrogel for integrated energy storage and self-diagnostic health monitoring in wearable biomedical devices. Energy Storage Materials. 2025;80:12
112. Zheng B, Zhou H, Zhao G, Wang K, Wu P, Liu H. et al. Bioinspired electrically conductive hydrogels: Rational engineering for next-generation flexible mechanosensors. Materials Science and Engineering: R: Reports. 2025 166
113. Orue-Echebarria MI, Lozano P, Olmedilla L, García Sabrido JL, Asencio JM. "Small-for-Flow" Syndrome: Concept Evolution. J Gastrointest Surg. 2020;24:1386-91
114. Reijnders-Boerboom G, Albers KI, Jacobs LMC, Helden EV, Rosman C, Díaz-Cambronero O. et al. Low intra-abdominal pressure in laparoscopic surgery: a systematic review and meta-analysis. Int J Surg. 2023;109:1400-11
115. Brusasco C, Germinale F, Dotta F, Benelli A, Guano G, Campodonico F. et al. Low Intra-Abdominal Pressure with Complete Neuromuscular Blockage Reduces Post-Operative Complications in Major Laparoscopic Urologic Surgery: A before-after Study. J Clin Med. 2022;11:7210
116. Gayathri R, ArulJothi KN, Hari Raj K, S G. Next-generation sutures: Combining natural polymers and graphene for optimal wound healing. Results in Surfaces and Interfaces. 2024;17:100287
117. Xue Y, Cao J, Feng T, Zhang K, Li S, Hu J. et al. Slipknot-gauged mechanical transmission and robotic operation. Nature. 2025;647:889-96
118. Rimereit JE, Lindgren CGW, Nerup N, Madsen GI, Le DQS, Möller S. et al. Incorporating a poly-ε-caprolactone scaffold in a stapled small intestinal anastomosis with induced ischemia significantly increased anastomotic tensile strength. An experimental study in pigs. Scand J Gastroenterol. 2025;60:54-61
119. Reischl S, Wilhelm D, Friess H, Neumann PA. Innovative approaches for induction of gastrointestinal anastomotic healing: an update on experimental and clinical aspects. Langenbecks Arch Surg. 2021;406:971-80
120. Mishra RR, Nielsen BE, Trudrung MA, Lee S, Bolstad LJ, Hellenbrand DJ. et al. The Effect of Tissue Inhibitor of Metalloproteinases on Scar Formation after Spinal Cord Injury. Cells. 2024;13:1547
121. Gil ES, Aleksi E, Spirio L. PuraStat RADA16 Self-Assembling Peptide Reduces Postoperative Abdominal Adhesion Formation in a Rabbit Cecal Sidewall Injury Model. Front Bioeng Biotechnol. 2021;9:782224
122. Zhao L, Zhao G, Fu J, Liu Y, Feng J, Zhang G. et al. Programmed Cell Death Protein 1 Engagement Impairs Cytoskeletal Forces and Nuclear Mechanotransduction in T Cells. ACS Nano. 2025;19:39667-81
123. Goulas S, Gelfand MV, Carmi I. Cellular Mechanobiology. Dev Cell. 2021;56:155
124. Feldman DS, Osborne S. Fibrin as a Tissue Adhesive and Scaffold with an Angiogenic Agent (FGF-1) to Enhance Burn Graft Healing In Vivo and Clinically. J Funct Biomater. 2018 9
125. Lin YB, Fu JH, Huang Y, Hu YH, Luo KJ, Wang KX. et al. Fibrin sealant for esophageal anastomosis: A phase II study. World J Gastrointest Oncol. 2020;12:651-62
126. Mohsin MEA, Siddiqa AJ, Mousa S, Shrivastava NK. Design, Characterization, and Release Kinetics of a Hybrid Hydrogel Drug Delivery System for Sustained Hormone Therapy. Polymers: Polymers (Basel). 2025 p. 999
127. Maintz M, Tourbier C, de Wild M, Cattin PC, Beyer M, Seiler D. et al. Patient-specific implants made of 3D printed bioresorbable polymers at the point-of-care: material, technology, and scope of surgical application. 3D Printing in Medicine. 2024 10: 13
128. Shen C, Shen A. 4D printing: innovative solutions and technological advances in orthopedic repair and reconstruction, personalized treatment and drug delivery. Biomed Eng Online. 2025;24:5
129. Guo X, Gao Y, Yu J, Qiu S, Wang X, Wang S. et al. Dynamic Mechanical Stimulation of Thermoresponsive Nanofibers for Activation of Fibroblasts in Skin Repair. Adv Healthc Mater. 2025;14:e2500277
130. Zhang T, Song F, Li BQ, Xu F, Yang Q. Electrohydrodynamic deformation of liquid film underneath a topographical template. Journal of Fluid Mechanics. 2025;1011:A46
131. Joppin V, Jourdan A, Bendahan D, Soucasse A, Guye M, Masson C. et al. Towards a better understanding of abdominal wall biomechanics: In vivo relationship between dynamic intra-abdominal pressure and magnetic resonance imaging measurements. Clin Biomech (Bristol). 2025;121:106396
132. Xie X, Chen X, Zhou J, Wang T, Yang G, Han F. et al. Dynamic Hydrogels with Tunable Mechanics for 3D Organoid Derivation. Small. 2025;21:e2501862
133. Smith AN, Ulsh JB, Gupta R, Tang MM, Peredo AP, Teinturier TD. et al. Characterization of degradation kinetics of additively manufactured PLGA under variable mechanical loading paradigms. Journal of the Mechanical Behavior of Biomedical Materials. 2024;153:106457
134. Maadani AM, Salahinejad E. Performance comparison of PLA- and PLGA-coated porous bioceramic scaffolds: Mechanical, biodegradability, bioactivity, delivery and biocompatibility assessments. J Control Release. 2022;351:1-7
135. Le Ruyet A, Yurtkap Y, Hartog F, Vegleur A, Turquier F, Lange JF. et al. Differences in biomechanics of abdominal wall closure with and without mesh reinforcement: A study in post mortem human specimens. J Mech Behav Biomed Mater. 2020;105:103683
136. Svensson-Raskh A, Schandl AR, Ståhle A, Nygren-Bonnier M, Fagevik Olsén M. Mobilization Started Within 2 Hours After Abdominal Surgery Improves Peripheral and Arterial Oxygenation: A Single-Center Randomized Controlled Trial. Phys Ther. 2021;101:pzab094
137. Du L, Zhao Y, Yin C, Liu S, Cui Z, Zhang M. Application of intra-abdominal pressure monitoring in early enteral nutrition after abdominal surgery. Am J Transl Res. 2021;13:7140-7
138. Jønsson LR, Foss NB, Orbæk J, Lauritsen ML, Sejrsen HN, Kristensen MT. Early intensive mobilization after acute high-risk abdominal surgery: a nonrandomized prospective feasibility trial. Can J Surg. 2023;66:E236-e45
139. Wang J, Yan J, Shi L, Wang Y, Tian X, Qi Y. et al. Intestinal barrier function as a key determinant of inflammation and nutritional status in digestive surgery patients: a real-world study. Front Nutr. 2025;12:1637877
140. Gao X, Zhang Y, Qi X, Xiao Y, Gao T, Jin G. et al. Early enteral nutrition versus early supplemental parenteral nutrition in patients undergoing major abdominal surgery: a secondary analysis of 2 randomized clinical trials. Am J Clin Nutr. 2024;119:1036-43
141. Wu JY, Liu MY, Liu TH, Kuo CY, Hung KC, Tsai YW. et al. Clinical efficacy of enteral nutrition feeding modalities in critically ill patients: a systematic review and meta-analysis of randomized controlled trials. Eur J Clin Nutr. 2023;77:1026-33
142. Kušar M, Djokić M, Djordjević S, Hribernik M, Krašna S, Trotovšek B. Preliminary study of reliability of transcutaneous sensors in measuring intraabdominal pressure. Scientific Reports. 2022;12:8268
143. Iacubovici L, Karol D, Baar Y, Beri A, Herzberg H, Zarour S. et al. Assessment of Intra-Abdominal Pressure with a Novel Continuous Bladder Pressure Monitor-A Clinical Validation Study. Life (Basel). 2023;13:384
144. Soucasse A, Jourdan A, Edin L, Meunier E, Bege T, Masson C. Assessment of the Smartpill, a Wireless Sensor, as a Measurement Tool for Intra-Abdominal Pressure (IAP). Sensors (Basel). 2023;24:54
145. Huynh M, Tjandra R, Helwa N, Okasha M, El-Falou A, Helwa Y. Continuous pH monitoring using a sensor for the early detection of anastomotic leaks. Front Med Technol. 2023;5:1128460
146. Li S, Lu D, Li S, Liu J, Xu Y, Yan Y. et al. Bioresorbable, wireless, passive sensors for continuous pH measurements and early detection of gastric leakage. Sci Adv. 2024;10:eadj0268
147. Ben-David M, Carmeli I, Orgad R, Nathansohn-Levi B, Yered T, Shor E. et al. Implantation of an Impedance Sensor for Early Detection of Gastrointestinal Anastomotic Leaks. J Surg Res. 2022;278:49-56
148. Liao CH, Cheng CT, Chen CC, Wang YH, Chiu HT, Peng CC. et al. Systematic Review of Diagnostic Sensors for Intra-Abdominal Pressure Monitoring. Sensors (Basel). 2021 21
149. Plešnik B, Djokić M, Djordjević S, Krašna S, Žumer J, Trotovšek B. Non-invasive and continuous intra-abdominal pressure assessment using MC sensors. Sci Rep. 2025;15:10775
150. Kong Y, Chang T, Cui Y, Ding X, Liu W. Behaviour and cognition of adult critical care nurses regarding intra-abdominal pressure monitoring: a cross-sectional study. BMC Nurs. 2025;24:1108
151. Ye W, Yang Z, Cao F, Li H, Zhao T, Zhang H. et al. Articular cartilage reconstruction with TGF-β1-simulating self-assembling peptide hydrogel-based composite scaffold. Acta Biomater. 2022;146:94-106
152. Balbona J, Chen L, Malafa MP, Hodul PJ, Dineen SP, Mehta R. et al. Outcomes of Gastric Resection in the Establishment of a Comprehensive Oncologic Robotic Program. J Surg Res. 2020;252:30-6
153. Gao R, Zhang Y, Deng B, Zhang J, Liang Z, Wei Z. et al. GM@mTG-V microspheres promote NP regeneration by reconstructing IVD biomechanics and inflammatory microenvironment. Mater Today Bio. 2025;32:101897
154. Chen S, Ma T, Wang J, Liang S, Liao H, Tan W. et al. Macrophage-derived biomimetic nanoparticles enhanced SDT combined with immunotherapy inhibited tumor growth and metastasis. Biomaterials. 2024;305:122456
155. Fink M, Akra B. Comparison of the international regulations for medical devices-USA versus Europe. Injury. 2023;54:110908
156. Gupte T, Nitave T, Gobburu J. Regulatory landscape of accelerated approval pathways for medical devices in the United States and the European Union. Front Med Technol. 2025;7:1586070
157. Liu H, Xiong M, Zeng Y, Shi Y, Pei Z, Liao C. Comparison of complications and bowel function among different reconstruction techniques after low anterior resection for rectal cancer: a systematic review and network meta-analysis. World J Surg Oncol. 2023;21:87
158. El Bojairami I, Jacobson N, Driscoll M. Development and evaluation of a numerical spine model comprising intra-abdominal pressure for use in assessing physiological changes on abdominal compliance and spinal stability. Clin Biomech (Bristol). 2022;97:105689
159. Suffredini G, Gao WD, Dodd OJ. Ultrasound Shear Wave Elastography Evaluation of the Liver and Implications for Perioperative Medicine. J Clin Med. 2024;13:11
160. Yu A, Zhu M, Chen C, Li Y, Cui H, Liu S. et al. Implantable Flexible Sensors for Health Monitoring. Adv Healthc Mater. 2024;13:e2302460
161. Papani R, Li Y, Wang S. Soft mechanical sensors for wearable and implantable applications. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2024;16:e1961
162. Iguchi K, Watanabe J, Suwa Y, Chida K, Atsumi Y, Numata M. et al. The usefulness of indocyanine green fluorescence imaging for intestinal perfusion assessment of intracorporeal anastomosis in laparoscopic colon cancer surgery. Int J Colorectal Dis. 2023;38:7
163. Cepeda S, García-García S, Arrese I, Velasco-Casares M, Sarabia R. Advantages and Limitations of Intraoperative Ultrasound Strain Elastography Applied in Brain Tumor Surgery: A Single-Center Experience. Oper Neurosurg. 2022;22:305-14
164. Song X, Hu M, Li B, Zhang K, Zhang X, Wang L. Advancing medical device regulatory reforms for innovation, translation and industry development in China. J Orthop Translat. 2022;37:89-93
165. Charoenchue P, Khorana J, Chitapanarux T, Inmutto N, Na Chiangmai W, Amantakul A. et al. Two-Dimensional Shear-Wave Elastography: Accuracy in Liver Fibrosis Staging Using Magnetic Resonance Elastography as the Reference Standard. Diagnostics (Basel). 2024;15:62
166. Ashikuzzaman M, Hall TJ, Rivaz H. Incorporating Gradient Similarity for Robust Time Delay Estimation in Ultrasound Elastography. IEEE Trans Ultrason Ferroelectr Freq Control. 2022;69:1738-50
167. Anders M, Meyer T, Warmuth C, Pfeuffer J, Tzschaetzsch H, Herthum H. et al. Rapid MR elastography of the liver for subsecond stiffness sampling. Magn Reson Med. 2024;91:312-24
168. Chen J, Chen J, Heilman JA, Glaser KJ, Grimm RC, Owusu N. et al. Abdominal MR elastography with multiple driver arrays: performance and repeatability. Abdom Radiol (NY). 2023;48:1945-54
169. Nacheva-Georgieva EL, Doykov DI, Hristov BK, Doykova KA, Doykov MI. Comparison of Point Shear Wave Elastography and 2-Dimensional Shear Wave Elastography Values of Liver Metastases from Colorectal Cancer. Gastroenterology Insights: Gastroenterology Insights. 2023 p. 271-81
170. Kohli DR, Mettman D, Andraws N, Haer E, Porter J, Ulusurac O. et al. Comparative accuracy of endosonographic shear wave elastography and transcutaneous liver stiffness measurement: a pilot study. Gastrointest Endosc. 2023;97:35-41.e1
171. Xie X, Wang Y, Deng B, Blatchley MR, Lan D, Xie Y. et al. Matrix metalloproteinase-responsive hydrogels with tunable retention for on-demand therapy of inflammatory bowel disease. Acta Biomater. 2024;186:354-68
Author contact
Corresponding authors: liuningedu.cn (Ning Liu); fengxuxjtu.edu.cn (Feng Xu); tingwenedu.cn (Ting Wen).
 Global reach, higher impact
Global reach, higher impact