13.3
Impact Factor
Theranostics 2015; 5(9):946-960. doi:10.7150/thno.11974 This issue Cite
Research Paper
A Plasmonic Gold Nanostar Theranostic Probe for In Vivo Tumor Imaging and Photothermal Therapy
1. Fitzpatrick Institute for Photonics, Duke University, Durham, NC, 27708, United States
2. Department of Biomedical Engineering, Duke University, Durham, NC, 27708, United States
3. Department of Chemistry, Duke University, Durham, NC, 27708, United States
4. Department of Pharmacology and Cancer Biology, Duke University Medical Center, Durham, NC, 27710, United States
5. Department of Radiology, Duke University Medical Center, Durham, NC, 27710, United States
6. Center for In Vivo Microscopy, Department of Radiology, Duke University Medical Center, Durham, NC 27710, United States
7. Department of Radiation Oncology, Duke University Medical Center, Durham, NC, 27710, United States
† These authors contributed equally to this work
Abstract
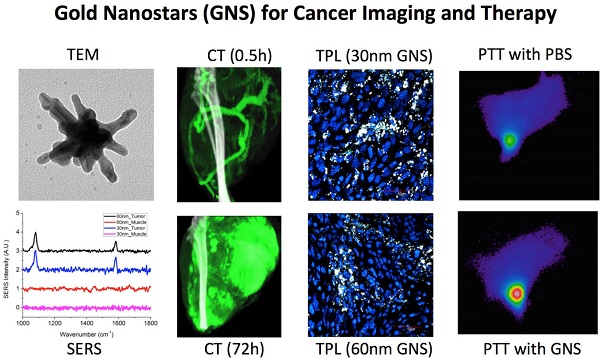
Nanomedicine has attracted increasing attention in recent years, because it offers great promise to provide personalized diagnostics and therapy with improved treatment efficacy and specificity. In this study, we developed a gold nanostar (GNS) probe for multi-modality theranostics including surface-enhanced Raman scattering (SERS) detection, x-ray computed tomography (CT), two-photon luminescence (TPL) imaging, and photothermal therapy (PTT). We performed radiolabeling, as well as CT and optical imaging, to investigate the GNS probe's biodistribution and intratumoral uptake at both macroscopic and microscopic scales. We also characterized the performance of the GNS nanoprobe for in vitro photothermal heating and in vivo photothermal ablation of primary sarcomas in mice. The results showed that 30-nm GNS have higher tumor uptake, as well as deeper penetration into tumor interstitial space compared to 60-nm GNS. In addition, we found that a higher injection dose of GNS can increase the percentage of tumor uptake. We also demonstrated the GNS probe's superior photothermal conversion efficiency with a highly concentrated heating effect due to a tip-enhanced plasmonic effect. In vivo photothermal therapy with a near-infrared (NIR) laser under the maximum permissible exposure (MPE) led to ablation of aggressive tumors containing GNS, but had no effect in the absence of GNS. This multifunctional GNS probe has the potential to be used for in vivo biosensing, preoperative CT imaging, intraoperative detection with optical methods (SERS and TPL), as well as image-guided photothermal therapy.
Keywords: Gold nanostars, photothermal therapy, nanoparticles, tumor imaging, SERS, CT, biodistribution, theranostics
 Global reach, higher impact
Global reach, higher impact