Impact Factor
Theranostics 2017; 7(2):238-252. doi:10.7150/thno.16086 This issue Cite
Research Paper
Comprehensive Metaproteomic Analyses of Urine in the Presence and Absence of Neutrophil-Associated Inflammation in the Urinary Tract
1. J. Craig Venter Institute, 9714 Medical Center Drive, Rockville, MD 20850.
2. Quest Diagnostics at Shady Grove Adventist Hospital, 9901 Medical Center Drive, Rockville 20850, MD.
3. Advanced Pathology Associates LLC at Shady Grove Adventist Hospital, 9901 Medical Center Drive, Rockville 20850, MD.
# Current address: Laboratory of Parasitic Diseases, NIAID, NIH, 4 Memorial Drive, Bethesda 20892, MD; Department of Microbiology and Immunology, Georgetown University, 3900 Reservoir Road, N.W., 20057, Washington, DC.
Abstract
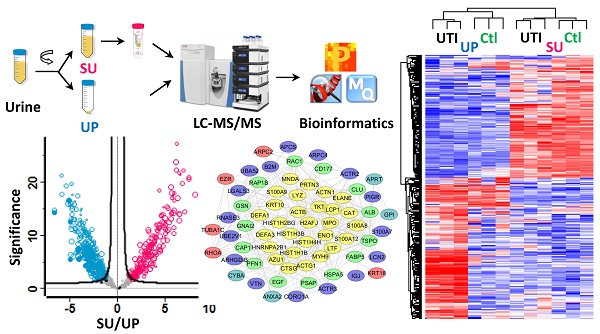
Inflammation in the urinary tract results in a urinary proteome characterized by a high dynamic range of protein concentrations and high variability in protein content. This proteome encompasses plasma proteins not resorbed by renal tubular uptake, renal secretion products, proteins of immune cells and erythrocytes derived from trans-urothelial migration and vascular leakage, respectively, and exfoliating urothelial and squamous epithelial cells. We examined how such proteins partition into soluble urine (SU) and urinary pellet (UP) fractions by analyzing 33 urine specimens 12 of which were associated with a urinary tract infection (UTI). Using mass spectrometry-based metaproteomic approaches, we identified 5,327 non-redundant human proteins, 2,638 and 4,379 of which were associated with SU and UP fractions, respectively, and 1,206 non-redundant protein orthology groups derived from pathogenic and commensal organisms of the urogenital tract. Differences between the SU and UP proteomes were influenced by local inflammation, supported by respective comparisons with 12 healthy control urine proteomes. Clustering analyses showed that SU and UP fractions had proteomic signatures discerning UTIs, vascular injury, and epithelial cell exfoliation from the control group to varying degrees. Cases of UTI revealed clusters of proteins produced by activated neutrophils. Network analysis supported the central role of neutrophil effector proteins in the defense against invading pathogens associated with subsequent coagulation and wound repair processes. Our study expands the existing knowledge of the urinary proteome under perturbed conditions, and should be useful as reference dataset in the search of biomarkers.
Keywords: urine, clinical proteomics, urothelial cells, label-free quantitation, urinary tract infection, neutrophils, inflammation, network analysis, shotgun proteomics, urine sediment
 Global reach, higher impact
Global reach, higher impact