13.3
Impact Factor
Theranostics 2018; 8(9):2348-2360. doi:10.7150/thno.22901 This issue Cite
Research Paper
A novel USP9X substrate TTK contributes to tumorigenesis in non-small-cell lung cancer
1. Department of Analytical Chemistry and CAS Key Laboratory of Receptor Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China
2. CAS Key Laboratory of Systems Biology, Innovation Center for Cell Signaling Network, CAS Center for Excellence in Molecular Cell Science, Institute of Biochemistry and Cell Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai 200031, China
3. State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, 555 Zuchongzhi Road, Shanghai 201203, China
4. University of Chinese Academy of Sciences, Number 19A Yuquan Road, Beijing 100049, China
* Xiangling Chen, Chengli Yu and Jing Gao contributed equally to this work.
Abstract
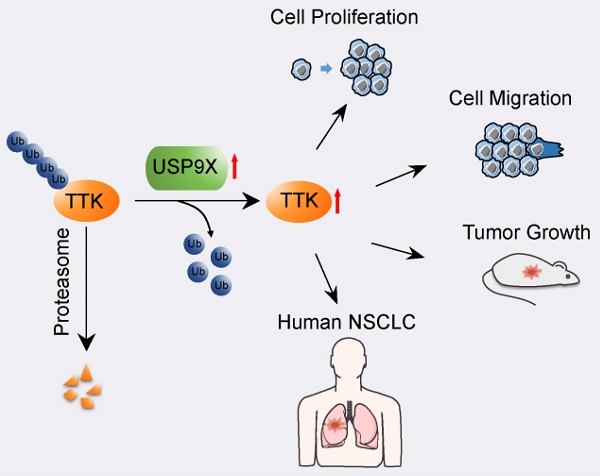
The X-linked deubiquitinase, USP9X, is implicated in multiple cancers by targeting various substrates. Increased expression of USP9X is observed in non-small-cell lung cancer (NSCLC) and is correlated with poor prognosis. However, the molecular mechanism for USP9X regulation of tumor cell survival and tumorigenesis in NSCLC is less defined.
Methods: In this study, chemical labeling, quantitative proteomic screening was applied to analyze A549 cells with or without USP9X RNA interference. Functional in vitro and in vivo experiments were performed to confirm the oncogenic effects of USP9X in NSCLC and to investigate the underlying mechanisms.
Results: The resulting data suggested that dual specificity protein kinase TTK is a potential substrate of USP9X. Further experimental evidences confirmed that USP9X stabilized TTK via direct interaction and efficient deubiquitination of TTK on K48 ubiquitin chain. Moreover, knockdown of USP9X or TTK inhibited cell proliferation, migration and tumorigenesis, and the immunohistochemical analysis of clinical NSCLC samples showed that the protein expression levels of USP9X and TTK were significantly elevated and positively correlated in tumor tissues.
Conclusions: In summary, our data demonstrated that the USP9X-TTK axis may play a critical role in NSCLC, and could be considered as a potential therapeutic target.
Keywords: non-small cell lung cancer (NSCLC), quantitative proteomics, deubiquitinase, USP9X, TTK
 Global reach, higher impact
Global reach, higher impact