13.3
Impact Factor
Theranostics 2018; 8(11):3164-3175. doi:10.7150/thno.22164 This issue Cite
Research Paper
Pre-existing anti-polyethylene glycol antibody reduces the therapeutic efficacy and pharmacokinetics of PEGylated liposomes
1. Graduate Institute of Medicine, College of Medicine, Kaohsiung Medical University, 100 Shih-Chuan First Road, Kaohsiung 80708, Taiwan
2. Department of Biomedical Imaging and Radiological Sciences and Institute of Microbiology and Immunology, National Yang-Ming University, Taipei, Taiwan
3. Institute of Biomedical Sciences, National Sun Yat-sen University, Kaohsiung, Taiwan
4. Center for Biomarkers and Biotech Drugs, Kaohsiung Medical University, 100 Shih-Chuan First Road, Kaohsiung 80708, Taiwan
5. Institute of Biomedical Sciences, Academia Sinica, 128 Academia Road, Section 2, Taipei 11529, Taiwan
6. Department of Biomedical Science and Environmental Biology, Kaohsiung Medical University, 100 Shih-Chuan First Road, Kaohsiung 80708, Taiwan
7. Department of Surgery, Faculty of Medicine, College of Medicine, Kaohsiung Medical University, 100 Shih-Chuan First Road, Kaohsiung 80708, Taiwan
8. Department of Medical Research, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan
9. Department of Surgery, Kaohsiung Municipal Ta-Tung Hospital, 68 Jhonghua third Road, Kaohsiung 80145, Taiwan
10. Cancer Center, Kaohsiung Municipal Ta-Tung Hospital, 68 Jhonghua third Road, Kaohsiung 80145, Taiwan
11. Division of Breast Surgery, Department of Surgery, Kaohsiung Medical University Hospital, Kaohsiung Medical University Hospital, 100 Tzyou first Road, Kaohsiung 80708, Taiwan
12. Department of Laboratory Medicine, School of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan
* Yuan-Chin Hsieh and Hsin-Ell Wang contributed equally to this work.
Abstract
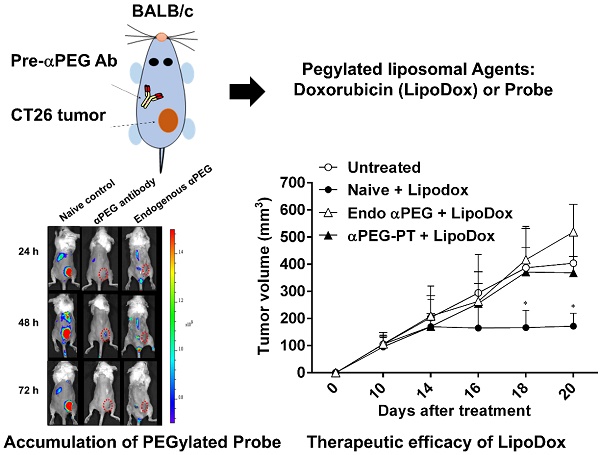
Rationale: Increasing frequency of human exposure to PEG-related products means that healthy people are likely to have pre-existing anti-PEG antibodies (pre-αPEG Ab). However, the influence of pre-αPEG Abs on the pharmacokinetics (PK) and therapeutic efficacy of LipoDox is unknown.
Methods: We generated two pre-αPEG Ab mouse models. First, naïve mice were immunized with PEGylated protein to generate an endogenous αPEG Ab titer (endo αPEG). Second, monoclonal αPEG Abs were passively transferred (αPEG-PT) into naïve mice to establish a αPEG titer. The naïve, endo αPEG and αPEG-PT mice were intravenously injected with 111in-labeled LipoDox to evaluate its PK. Tumor-bearing naïve, endo αPEG and αPEG-PT mice were intravenously injected with 111in-labeled LipoDox to evaluate its biodistribution. The therapeutic efficacy of LipoDox was estimated in the tumor-bearing mice.
Results: The areas under the curve (AUC)last of LipoDox in endo αPEG and αPEG-PT mice were 11.5- and 15.6- fold less, respectively, than that of the naïve group. The biodistribution results suggested that pre-αPEG Ab can significantly reduce tumor accumulation and accelerate blood clearance of 111In-labeled LipoDox from the spleen. The tumor volumes of the tumor-bearing endo αPEG and αPEG-PT mice after treatment with LipoDox were significantly increased as compared with that of the tumor-bearing naïve mice.
Conclusions: Pre-αPEG Abs were found to dramatically alter the PK and reduce the tumor accumulation and therapeutic efficacy of LipoDox. Pre-αPEG may have potential as a marker to aid development of personalized therapy using LipoDox and achieve optimal therapeutic efficacy.
Keywords: PEG, Polyethylene glycol, anti-PEG antibodies, liposome, LipoDox, ELISA
 Global reach, higher impact
Global reach, higher impact