13.3
Impact Factor
Theranostics 2018; 8(15):4097-4115. doi:10.7150/thno.26195 This issue Cite
Research Paper
Thermo- and pH-dual responsive polymeric micelles with upper critical solution temperature behavior for photoacoustic imaging-guided synergistic chemo-photothermal therapy against subcutaneous and metastatic breast tumors
1. The Key Laboratory of Biomedical Information Engineering of Ministry of Education, School of Life Science and Technology, Xi'an Jiaotong University, Xi'an 710049, China
2. Department of Biomedical Engineering, School of Engineering, Sun Yat-sen University, Guangzhou 510006, China
3. Department of Medical Oncology, Sun Yat-sen University Cancer Center; State Key Laboratory of Oncology in South China, Guangzhou 510060, China
* These authors contribute equally to this work.
Abstract
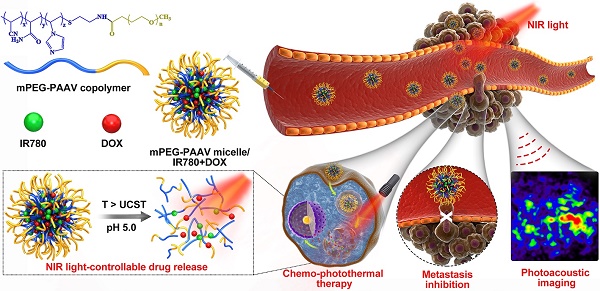
Chemo-photothermal therapy shows great potential for inhibiting tumor growth. However, achieving maximal chemo-photothermal synergistic efficacy is challenging because of the low efficiency of controllable chemo-drug release in response to external or internal triggers. Thus, a nano-delivery system that could effectively achieve photothermal therapy and dual stimuli-responsive (heat and pH) drug release to inhibit both primary breast tumor growth and metastases is required.
Methods: Herein, a thermo- and pH-responsive polymer (mPEG-PAAV) with an upper critical solution temperature (UCST) was synthesized to fabricate a DOX- and IR780-loaded micellar system. After systematic studies of the photothermal performance and controllable drug release of mPEG-PAAV micelles/IR780+DOX under NIR irradiation at different pH values, their chemo-photothermal synergetic therapy efficacies were also estimated both in in vitro and in vivo.
Results: Because of the photothermal conversion of mPEG-PAAV micelle/IR780+DOX (~200 nm, 3.82 mV), high local temperature could be induced at the tumor site under NIR laser irradiation. This hyperthermia not only produced an enhanced tumor necrosis, but also broke down the micelles under the decreased pH environment, resulting in rapid DOX release and enhanced intracellular drug accumulation after NIR laser irradiation. In addition, photoacoustic imaging (PAI) of mPEG-PAAV/IR780+DOX micelle was adopted to monitor the morphology and micro-vascular distribution of the tumor tissue, which could also guide the chemo-photothermal therapy. Most importantly, the systemic administration of mPEG-PAAV micelles/IR780+DOX combined with NIR laser irradiation could simultaneously eliminate the 4T1 breast tumor and thoroughly suppress lung metastasis without any obvious adverse effects.
Conclusion: Herein, a pH- and thermo-dual responsive UCST micelle system was developed for delivering IR780 and DOX, which could achieve NIR laser-controlled drug release and PA imaging guidance for chemo-photothermal synergistic therapy of both primary breast tumors and their metastases.
Keywords: upper critical solution temperature (UCST), chemo-photothermal therapy, controlled release, photoacoustic imaging, breast cancer
 Global reach, higher impact
Global reach, higher impact