Impact Factor
Theranostics 2019; 9(6):1764-1776. doi:10.7150/thno.31233 This issue Cite
Research Paper
Composition-Tunable Ultrasmall Manganese Ferrite Nanoparticles: Insights into their In Vivo T1 Contrast Efficacy
1. Key Laboratory of Synthetic and Natural Functional Molecule Chemistry of the Ministry of Education, College of Chemistry and Materials Science, Northwest University, Xi'an, Shaanxi 710069, China.
2. Division of nephrology, Peking University Third Hospital, Beijing, China.
3. State Key Laboratory of Oncology in South China, Sun Yat-sen University Cancer Center, Collaborative Innovation Center for Cancer Medicine, Guangzhou 510060, China.
4. Department of Nuclear Medicine, The Third Affiliated Hospital of Sun Yat-sen University, 600 Tianhe Road, Guangzhou, Guangdong 510630, China.
5. School of Life Sciences, National Engineering Research Center for Miniaturized Detection Systems, Northwest University, Xi'an, Shaanxi, China.
*Yuqing Miao and Qian Xie have equal contribution.
Abstract
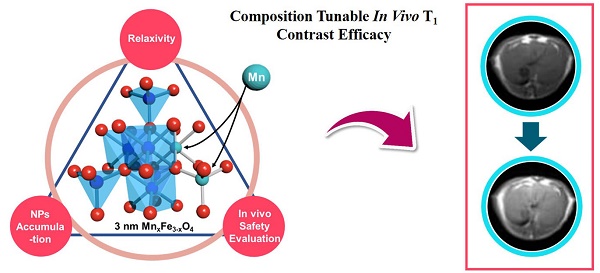
The development of a highly efficient, low-toxicity, ultrasmall ferrite nanoparticle-based T1 contrast agent for high-resolution magnetic resonance imaging (MRI) is highly desirable. However, the correlations between the chemical compositions, in vitro T1 relaxivities, in vivo nano-bio interactions and toxicities remain unclear, which has been a challenge in optimizing the in vivo T1 contrast efficacy.
Methods: Ultrasmall (3 nm) manganese ferrite nanoparticles (MnxFe3-xO4) with different doping concentrations of the manganese ions (x = 0.32, 0.37, 0.75, 1, 1.23 and 1.57) were used as a model system to investigate the composition-dependence of the in vivo T1 contrast efficacy. The efficacy of liver-specific contrast-enhanced MRI was assessed through systematic multiple factor analysis, which included the in vitro T1 relaxivity, in vivo MRI contrast enhancement, pharmacokinetic profiles (blood half-life time, biodistribution) and biosafety evaluations (in vitro cytotoxicity testing, in vivo blood routine examination, in vivo blood biochemistry testing and H&E staining to examine the liver).
Results: With increasing Mn doping, the T1 relaxivities initially increased to their highest value of 10.35 mM-1s-1, which was obtained for Mn0.75Fe2.25O4, and then the values decreased to 7.64 m M-1s-1, which was obtained for the Mn1.57Fe1.43O4 nanoparticles. Nearly linear increases in the in vivo MRI signals (ΔSNR) and biodistributions (accumulation in the liver) of the MnxFe3-xO4 nanoparticles were observed for increasing levels of Mn doping. However, both the in vitro and in vivo biosafety evaluations suggested that MnxFe3-xO4 nanoparticles with high Mn-doping levels (x > 1) can induce significant toxicity.
Conclusion: The systematic multiple factor assessment indicated that the MnxFe3-xO4 (x = 0.75-1) nanoparticles were the optimal T1 contrast agents with higher in vivo efficacies for liver-specific MRI than those of the other compositions of the MnxFe3-xO4 nanoparticles. Our work provides insight into the optimization of ultrasmall ferrite nanoparticle-based T1 contrast agents by tuning their compositions and promotes the translation of these ultrasmall ferrite nanoparticles for clinical use of high-performance contrast-enhanced MRI.
Keywords: ultrasmall ferrite nanoparticles, composition effect, MRI, in vivo T1 contrast efficacy
 Global reach, higher impact
Global reach, higher impact