Impact Factor
Theranostics 2019; 9(12):3608-3621. doi:10.7150/thno.32945 This issue Cite
Research Paper
Disease-specific primed human adult stem cells effectively ameliorate experimental atopic dermatitis in mice
1. Adult Stem Cell Research Center and Research Institute for Veterinary Science, College of Veterinary Medicine, Seoul National University, Seoul 08826, Republic of Korea
2. Department of Biochemistry, BK21 PLUS Program for Creative Veterinary Science Research and Research Institute for Veterinary Science, College of Veterinary Medicine, Seoul National University, Seoul 08826, Republic of Korea
3. Department of Life Science in Dentistry, School of Dentistry, Pusan National University, Yangsan 50612, Republic of Korea
4. Institute for Translational Dental Sciences, Pusan National University, Yangsan 50612, Republic of Korea
*Authors share co-corresponding authorship
§Current address: Program in Developmental Endocrinology and Genetics, Eunice Kennedy Shriver National Institute of Child Health and Human Development, NIH, Bethesda, MD 20892, USA
Abstract
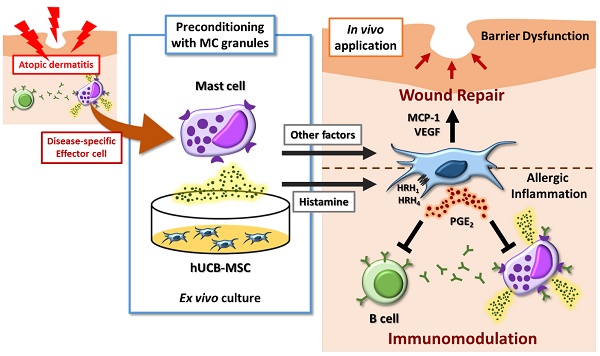
Although human mesenchymal stem cells (hMSCs) hold considerable promise as an alternative therapeutic reagent for allergic disorders including atopic dermatitis (AD), the strategy for enhancing hMSC-based therapy remains challenging. We sought to investigate whether preconditioning with mast cell (MC) granules could enhance the therapeutic efficiency of human umbilical cord blood-derived MSCs (hUCB-MSCs) against AD.
Methods: AD was experimentally induced in NC/Nga mice by repeated applications of 4% sodium dodecyl sulfate (SDS) and dermatophagoides farinae (Df) extract, and preconditioned hUCB-MSCs were subcutaneously injected. The therapeutic effect was determined by gross examination and additional ex vivo experiments performed using blood and skin samples to determine the resolution of allergic inflammation. To explore the underlying mechanisms, several co-culture assays with primary isolated immune cells and wound closure assays were conducted.
Results: Pretreatment of MC granules enhanced the therapeutic effects of hUCB-MSCs by attenuating the symptoms of AD in an experimental animal model. MC granule-primed cells suppressed the activation of major disease-inducing cells, MCs and B lymphocytes more efficiently than naïve cells both in vitro and in vivo. Histamine-mediated upregulation of the COX-2 signaling pathway was shown to play a crucial role in suppression of the allergic immune response by MC-pretreated hUCB-MSCs. Moreover, MC pretreatment improved the wound healing ability of hUCB-MSCs.
Conclusions: Our findings indicate that pre-exposure to MC granules improved the therapeutic effect of hUCB-MSCs on experimental AD by resolving the allergic immune reaction and accelerating the tissue regeneration process more efficiently than naïve cells, suggesting a potential enhancement strategy for stem cell-based therapy.
Keywords: Atopic dermatitis, Mesenchymal stem cells, Immunomodulation, Wound healing, Stem cell-based therapy
 Global reach, higher impact
Global reach, higher impact