13.3
Impact Factor
Theranostics 2019; 9(26):8277-8293. doi:10.7150/thno.35686 This issue Cite
Research Paper
Conditional knockout of TGF-βRII /Smad2 signals protects against acute renal injury by alleviating cell necroptosis, apoptosis and inflammation
1. The Key Laboratory of Major Autoimmune Diseases, Anhui Institute of Innovative Drugs, School of Pharmacy, Anhui Medical University; The key laboratory of Anti-inflammatory and Immune Medicines, Ministry of Education, Hefei 230032, China.
2. Department of Pharmacy, The 901 Hospital of Chinese People's Liberation Army Joint Service Support Unit, Hefei, China
3. Department of Medicine & Therapeutics, Li Ka Shing Institute of Health Sciences, and Shenzhen Research Institute, The Chinese University of Hong Kong, Hong Kong, China
4. School of Basic Medical Sciences, Anhui Medical University. Anhui, China
5. The Key Laboratory of Major Autoimmune Diseases, Anhui Institute of Innovative Drugs, School of Pharmacy, Anhui Medical University; The key laboratory of Anti-inflammatory and Immune Medicines, Ministry of Education, Hefei 230032, China.
#These authors contribute equally.
Abstract
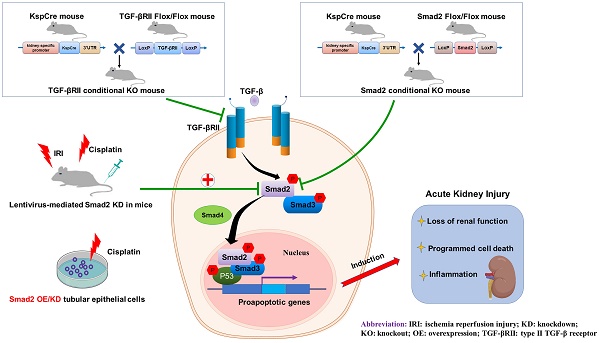
Rationale: TGF-β/Smad signaling is the central mediator for renal fibrosis, however, its functional role in acute kidney injury (AKI) is not fully understood. We previously showed Smad2 protects against renal fibrosis by limiting Smad3 signaling, but details on its role in acute phase are unclear. Recent evidence showed that TGF-β/Smad3 may be involved in the pathogenesis of AKI, so we hypothesized that Smad2 may play certain roles in AKI due to its potential effect on programmed cell death.
Methods: We established a cisplatin-induced AKI mouse model with TGF-β type II receptor or Smad2 specifically deleted from renal tubular epithelial cells (TECs). We also created stable in vitro models with either Smad2 knockdown or overexpression in human HK2 cells. Importantly, we evaluated whether Smad2 could serve as a therapeutic target in both cisplatin- and ischemic/reperfusion (I/R)-induced AKI mouse models by silencing Smad2 in vivo.
Results: Results show that disruption of TGF-β type II receptor suppressed Smad2/3 activation and attenuated renal injury in cisplatin nephropathy. Furthermore, we found that conditional knockout of downstream Smad2 in TECs protected against loss of renal function, and alleviated p53-mediated cell apoptosis, RIPK-mediated necroptosis and p65 NF-κB-driven renal inflammation in cisplatin nephropathy. This was further confirmed in cisplatin-treated Smad2 knockdown and overexpression HK2 cells. Additionally, lentivirus-mediated Smad2 knockdown protected against renal injury and inflammation while restoring renal function in established nephrotoxic and ischemic AKI models.
Conclusions: These findings show that unlike its protective role in renal fibrosis, Smad2 promoted AKI by inducing programmed cell death and inflammation. This may offer a novel therapeutic target for acute kidney injury.
Keywords: Acute kidney injury, Cisplatin, TGF-β receptor, Smad2, Necroptosis, Inflammation, Apoptosis
 Global reach, higher impact
Global reach, higher impact