13.3
Impact Factor
Theranostics 2020; 10(5):2115-2129. doi:10.7150/thno.37623 This issue Cite
Research Paper
Comprehensive characterization of the phosphoproteome of gastric cancer from endoscopic biopsy specimens
1. Laboratory of Proteome Research, National Institute of Biomedical Innovation, Health and Nutrition, Ibaraki, Osaka 567-0085, Japan
2. Laboratory of Proteomics for Drug Discovery, Center for Drug Design Research, National Institute of Biomedical Innovation, Health and Nutrition, Ibaraki, Osaka 567-0085, Japan
3. Gastrointestinal Medical Oncology Division, National Cancer Center Hospital, Tsukiji, Chuo-ku, Tokyo, 104-0045, Japan
4. Department of Biomarkers for Early Detection of Cancer, National Cancer Center Research Institute, Tokyo 104-0045, Japan
Abstract
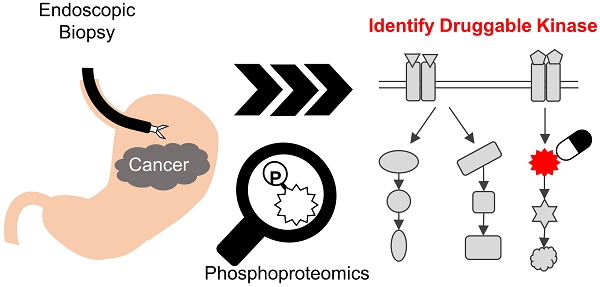
Rationale: Cancer phosphoproteomics can provide insights regarding kinases that can be targeted for therapeutic applications. Monitoring the phosphoproteomics in cancer is expected to play a key role in optimizing treatments with kinase inhibitors. Clinical phosphoproteomics in surgical tissues and patient-derived models has been studied intensively. However, the reported data may not accurately reflect the phosphosignaling status in patients due to the effect of ischemia occurring during surgery or changes in the characteristics of cancer cells when establishing the models. In contrast, endoscopic biopsies have an advantage for clinical phosphoproteomics because they can be rapidly cryo-preserved. We aimed to develop a highly sensitive method for phosphoproteomics in endoscopic biopsies of gastric cancer.
Methods: Three tumor biopsies and three normal gastric biopsies were obtained by endoscopy at one time, and subjected to our optimized phosphoproteomics. Phosphopeptides were enriched with an immobilized metal affinity chromatography, and labeled with Tandem Mass Tag reagent. Quantified phosphosites were compared between the pairs of tumor/normal biopsies within same patient. Cancer-specific activated pathways and kinases were identified by pathway enrichment analysis and kinase-substrate enrichment analysis.
Results: Our protocol enabled the identification of more than 10,000 class 1 phosphosites from endoscopic biopsies. A comparison between samples from cancer tissue and normal mucosa demonstrated differences in the phosphosignaling, including biomarkers of response to DNA damage. Finally, cancer-specific activation of DNA damage response signaling was validated by additional phosphoproteomics of other patients and western blotting of gastric cancer/normal cells.
Conclusion: In summary, our pioneering approach will facilitate more accurate clinical phosphoproteomics in endoscopic biopsies, which can be applied to monitor the activities of therapeutic kinases and, ultimately, can be a useful tool to precision medicine.
Keywords: Endoscopic biopsy, Phosphoproteomics, Precision medicine, Kinome
 Global reach, higher impact
Global reach, higher impact