Impact Factor
Theranostics 2020; 10(9):4042-4055. doi:10.7150/thno.42218 This issue Cite
Research Paper
Gold clusters prevent breast cancer bone metastasis by suppressing tumor-induced osteoclastogenesis
1. Spine Center, Department of Orthopedics, Changzheng Hospital, Second Military Medical University, Shanghai, China.
2. Department of Chemistry and Chemical Engineering, Beijing University of Technology, Beijing, China.
3. Center of Excellence for Environmental Safety and Biological Effects, Beijing University of Technology, Beijing, China.
4. CAS Key Laboratory for the Biological Effects of Nanomaterials and Nanosafety, Institute of High Energy Physics, Chinese Academy of Sciences, Beijing, China.
#These authors contributed equally to this work.
Abstract
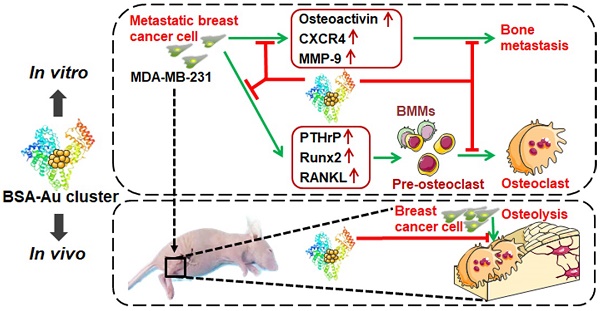
Rationale: Bone is the most frequent site for breast cancer metastasis, which accounts for the leading cause of death in advanced breast cancer patients. Serious skeletal-related events (SREs) caused by bone metastasis have a decisive impact on the life expectancy of breast cancer patients, making breast cancer almost incurable. Metastatic breast cancer cell induced pathological osteoclastogenesis is a key driver of bone metastasis and osteolytic bone lesions. We previously reported that gold clusters can prevent inflammation induced osteoclastogenesis and osteolysis in vivo. In this study, we investigated the effects of a BSA-coated gold cluster on metastatic breast cancer-induced osteoclastogenesis in vitro and tumor-induced osteolysis in vivo, and elucidated its possible mechanism.
Methods: Breast cancer cell line MDA-MB-231 was used to evaluate the regulatory effects of gold clusters on breast cancer metastasis and tumor induced osteoclastogenesis in vitro. Cell counting kit-8, transwell, wound-healing and colony formation assays were performed to evaluate the effect of gold clusters on proliferation and metastasis of MDA-MB-231 cells. Tartrate-resistant acid phosphatase (TRAP) staining and filamentous-actin rings analysis were used to detect the regulatory effects of gold clusters on MDA-MB-231 cell-conditioned medium (MDA-MB-231 CM) triggered and receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclastogenesis in mouse bone marrow-derived mononuclear cells (BMMs). A mouse model of breast cancer bone metastasis was used to evaluate the in vivo activity of the gold cluster on the tumor induced osteolysis.
Results: The gold clusters suppressed the migration, invasion and colony formation of MDA-MB-231 cells in a dose-dependent manner in vitro. The gold clusters strongly inhibited both MDA-MB-231 CM triggered and RANKL-induced osteoclast formation from BMMs in vitro. Cell studies indicated that the gold clusters suppressed the expression of osteolysis-related factors in MDA-MB-231 cells and inhibited the subsequent activation of NF-κB pathway in BMMs. Treatment with the clusters at a dose of 10 mg Au/kg.bw significantly reduces the breast cancer cell induced osteolysis in vivo.
Conclusion: Therefore, the gold clusters may offer new therapeutic agents for preventing breast cancer bone metastasis and secondary osteolysis to improve patient outcomes.
Keywords: Breast cancer bone metastasis, gold clusters, osteoclastogenesis, osteolysis, MDA-MB-231
 Global reach, higher impact
Global reach, higher impact