Impact Factor
Theranostics 2020; 10(12):5259-5275. doi:10.7150/thno.42705 This issue Cite
Research Paper
Marizomib suppresses triple-negative breast cancer via proteasome and oxidative phosphorylation inhibition
1. QIMR Berghofer Medical Research Institute, 300 Herston Road, Herston, Brisbane QLD 4006, Australia
2. Radiation Biology Research Center, Institute for Radiological Research, Chang Gung Memorial Hospital/Chang Gung University, Taoyuan 333, Taiwan
3. School of Medical Science, Griffith University, Southport, QLD 4222, Australia
4. UQ Centre for Clinical Research, Faculty of Medicine, The University of Queensland, Herston, QLD 4029, Australia
5. Olivia Newton-John Cancer Research Institute, 145 Studley Road, Heidelberg Vic 3084 Australia
6. Institute of Biotechnology, Czech Academy of Sciences, Prague-West, 252 50 Czech Republic
Abstract
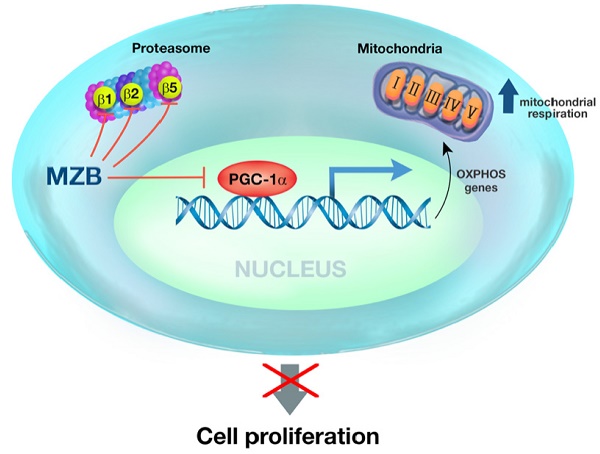
Purpose: Lacking effective targeted therapies, triple-negative breast cancer (TNBCs) is highly aggressive and metastatic disease, and remains clinically challenging breast cancer subtype to treat. Despite the survival dependency on the proteasome pathway genes, FDA-approved proteasome inhibitors induced minimal clinical response in breast cancer patients due to weak proteasome inhibition. Hence, developing effective targeted therapy using potent proteasome inhibitor is required.
Methods: We evaluated anti-cancer activity of a potent proteasome inhibitor, marizomib, in vitro using breast cancer lines and in vivo using 4T1.2 murine syngeneic model, MDA-MB-231 xenografts, and patient-derived tumor xenografts. Global proteome profiling, western blots, and RT-qPCR were used to investigate the mechanism of action for marizomib. Effect of marizomib on lung and brain metastasis was evaluated using syngeneic 4T1BR4 murine TNBC model in vivo.
Results: We show that marizomib inhibits multiple proteasome catalytic activities and induces a better anti-tumor response in TNBC cell lines and patient-derived xenografts alone and in combination with the standard-of-care chemotherapy. Mechanistically, we show that marizomib is a dual inhibitor of proteasome and oxidative phosphorylation (OXPHOS) in TNBCs. Marizomib reduces lung and brain metastases by reducing the number of circulating tumor cells and the expression of genes involved in the epithelial-to-mesenchymal transition. We demonstrate that marizomib-induced OXPHOS inhibition upregulates glycolysis to meet the energetic demands of TNBC cells and combined inhibition of glycolysis with marizomib leads to a synergistic anti-cancer activity.
Conclusions: Our data provide a strong rationale for a clinical evaluation of marizomib in primary and metastatic TNBC patients.
Keywords: Marizomib, triple-negative breast cancer, metastasis, oxidative phosphorylation, glycolysis
 Global reach, higher impact
Global reach, higher impact