13.3
Impact Factor
Theranostics 2020; 10(19):8468-8478. doi:10.7150/thno.46241 This issue Cite
Research Paper
Serum piRNA-54265 is a New Biomarker for early detection and clinical surveillance of Human Colorectal Cancer
1. Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China and Collaborative Innovation Center for Cancer Medicine, Guangzhou, China.
2. Department of Occupational & Environmental Health, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
3. Department of Colorectal Surgery, Sun Yat-sen University Cancer Center, Guangzhou, China.
4. Department of Pathology, Sun Yat-sen University Cancer Center, Guangzhou, China.
5. Department of Etiology and Carcinogenesis, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China.
6. Key Laboratory of Environment and Health, Ministry of Education & Ministry of Environmental Protection, State Key Laboratory of Environmental Health (Incubating), School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, China.
#These authors contributed equally to this work.
Abstract
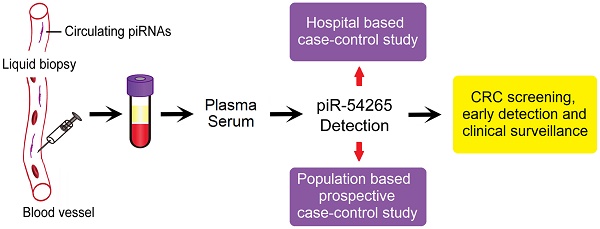
Background: Our previous study has demonstrated an oncogenic role of PIWI-interacting RNA-54265 (piR-54265) in colorectal cancer (CRC). Here, we investigate whether it can be a blood biomarker for population screening and clinical applications.
Methods: Serum piR-54265 levels were determined by a digital PCR method in 209 cancer-free healthy controls, 725 patients with CRC, 1303 patients with other types of digestive cancer and 192 patients with benign colorectal tumors. A prospective case-control analysis was conducted to assess the predictive value of serum piR-54265 for future CRC diagnosis. Receiver operating characteristic (ROC) curve was constructed to quantify the diagnostic performance of serum piR-54265 levels by assessing its sensitivity, specificity and respective areas under curve (AUC). The odds ratios (ORs) were computed using multivariate logistic regression models.
Results: Serum piR-54265 levels were significantly elevated only in patients with CRC compared with controls and patients with other cancer types. The AUC for recognizing CRC was 0.896 (95% CI, 0.874-0.914), with a sensitivity and specificity being 85.7% and 65.1% at 1500 copies/µL as a cut-off value. The serum piR-54265 levels in patients declined substantially after surgery but increased significantly again when tumor relapses. The prediagnostic serum piR-54265 levels were significantly associated with future CRC diagnosis, with the ORs of 7.23, 2.80, 2.45, and 1.24 for those whose CRC was diagnosed within 1, 2, 3 and >3 years. Serum piR-54265 test is more sensitive than other blood CRC markers.
Conclusion: Serum piR-54265 may serve as a valuable biomarker for CRC screening, early detection and clinical surveillance.
Keywords: colorectal cancer, serum piRNA-54265, biomarker
 Global reach, higher impact
Global reach, higher impact