13.3
Impact Factor
Theranostics 2020; 10(19):8494-8512. doi:10.7150/thno.44849 This issue Cite
Research Paper
EZH2-mediated Epigenetic Silencing of miR-29/miR-30 targets LOXL4 and contributes to Tumorigenesis, Metastasis, and Immune Microenvironment Remodeling in Breast Cancer
1. Henan Key Laboratory of Immunology and Targeted Therapy, School of Laboratory Medicine, Xinxiang Medical University, Xinxiang, Henan 453003, China.
2. The State Key Laboratory of Cancer Biology, Department of Immunology, Fourth Military Medical University, Xi'an, Shaanxi 710032, China.
3. The State Key Laboratory of Cancer Biology, Department of Biochemistry and Molecular Biology, Fourth Military Medical University, Xi'an, Shaanxi 710032, China.
4. Department of Thyroid, Breast and Vascular Surgery, Xijing Hospital, Fourth Military Medical University, Xi'an, China.
5. Henan Collaborative Innovation Center of Molecular Diagnosis and Laboratory Medicine, School of Laboratory Medicine, Xinxiang Medical University, Xinxiang, Henan 453003, China.
6. Department of Thoracic Surgery, Tangdu hospital, Fourth Military Medical University, Xi'an, China.
*These authors contributed equally to this work.
Abstract
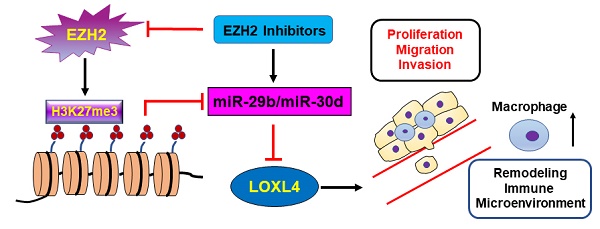
Enhancer of Zeste Homolog 2 (EZH2), a key epigenetic regulator, is involved in breast cancer progression and metastasis. LOXL4 is increasingly recognized as an important player in cancer progression. To date, how EZH2 regulates LOXL4 in the progression of breast cancer remains unclear.
Methods: We evaluated the association between LOX family proteins and EZH2 in invasive breast carcinoma through the starBase v2.0 analysis, and its correlation with breast tumorigenesis using the Oncomine dataset. We then applied miRcode data combined with gene expression omnibus (GEO) data to screen candidate miRNAs mediating the regulation of LOXL4 by EZH2. We explored the regulatory mechanism of EZH2, miR-29b/miR-30d, and LOXL4 in breast cancer cells by qRT-PCR, Western blotting, cell proliferation, colony formation, and wound healing assays, xenograft experiments, dual-luciferase reporter assay, and chromatin immunoprecipitation. All statistical tests were two-sided.
Results: Inhibition of EZH2 or LOXL4, or miR-29b/miR-30d overexpression, decreased breast cancer cell proliferation, migration, and metastasis in vitro and in vivo. LOXL4 was identified as a direct target of miR-29b and miR-30d. EZH2 inhibition enhanced miR-30d and miR-29b transcription via promoter binding activity, leading to the reduced expression of LOXL4. Immunohistochemical analysis of human breast cancer specimens and flow cytometry analysis of tumor-infiltrating macrophages in mice showed a positive association of EZH2 with LOXL4 expression and macrophage infiltration.
Conclusions: Our findings identified EZH2-miR-29b/miR-30d-LOXL4 signaling pathway was involved in breast tumorigenesis, and suggested that the epigenetic modulation represents a potential therapeutic target for breast cancer by controlling macrophage activation.
Keywords: Breast cancer, EZH2, LOXL4, miR-29b/miR-30d, Macrophage
 Global reach, higher impact
Global reach, higher impact