Impact Factor
Theranostics 2020; 10(19):8705-8720. doi:10.7150/thno.46854 This issue Cite
Research Paper
Restorative functions of Autologous Stem Leydig Cell transplantation in a Testosterone-deficient non-human primate model
1. Department of Andrology, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China.
2. Center for Stem Cell Biology and Tissue Engineering, Key Laboratory for Stem Cells and Tissue Engineering, Ministry of Education, Sun Yat-sen University, Guangzhou, Guangdong, China.
3. Reproductive Medicine Center, The Key Laboratory for Reproductive Medicine of Guangdong Province, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China.
4. Center for Reproductive Medicine, The Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China.
5. Guangdong Provincial Key Laboratory of Orthopedics and Traumatology, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China.
6. Department of Biochemistry, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou, Guangdong, China.
7. Department of Nuclear Medicine, The Third Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China.
8. Scientific Research Center, The Seventh Affiliated Hospital of Sun Yat-sen University, Shenzhen, Guangdong, China.
*These authors made equal contributions to this work.
Abstract
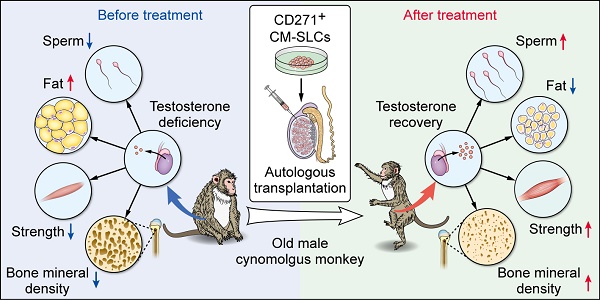
Rationale: Stem Leydig cells (SLCs) transplantation can restore testosterone production in rodent models and is thus a potential solution for treating testosterone deficiency (TD). However, it remains unknown whether these favorable effects will be reproduced in more clinically relevant large-animal models. Therefore, we assessed the feasibility, safety and efficacy of autologous SLCs transplantation in a testosterone-deficient non-human primate (NHP) model.
Methods: Cynomolgus monkey SLCs (CM-SLCs) were isolated from testis biopsies of elderly (> 19 years) cynomolgus monkeys by flow cytometry. Autologous CM-SLCs were injected into the testicular interstitium of 7 monkeys. Another 4 monkeys were injected the same way with cynomolgus monkey dermal fibroblasts (CM-DFs) as controls. The animals were then examined for sex hormones, semen, body composition, grip strength, and exercise activity.
Results: We first isolated CD271+ CM-SLCs which were confirmed to expand continuously and show potential to differentiate into testosterone-producing Leydig cells (LCs) in vitro. Compared with CM-DFs transplantation, engraftment of autologous CM-SLCs into elderly monkeys could significantly increase the serum testosterone level in a physiological pattern for 8 weeks, without any need for immunosuppression. Importantly, CM-SLCs transplantation recovered spermatogenesis and ameliorated TD-related symptoms, such as those related to body fat mass, lean mass, bone mineral density, strength and exercise capacity.
Conclusion: For the first time, our short-term observations demonstrated that autologous SLCs can increase testosterone levels and ameliorate relevant TD symptoms in primate models. A larger cohort with long-term follow-up will be required to assess the translational potential of autologous SLCs for TD therapy.
Keywords: Stem Leydig cell, Autologous, Transplantation, Testosterone deficiency, Non-human primate
 Global reach, higher impact
Global reach, higher impact