13.3
Impact Factor
Theranostics 2020; 10(20):9395-9406. doi:10.7150/thno.48392 This issue Cite
Research Paper
Hypoxia dynamics on FMISO-PET in combination with PD-1/PD-L1 expression has an impact on the clinical outcome of patients with Head-and-neck Squamous Cell Carcinoma undergoing Chemoradiation
1. Department of Radiation Oncology, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
2. German Cancer Consortium (DKTK), Partner Site Freiburg and German Cancer Research Center (DKFZ), Heidelberg, Germany.
3. Department of Nuclear Medicine, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
4. Department of Nuclear Medicine, Technical University of Munich, Munich, Germany.
5. Institute of Surgical Pathology, Department of Pathology, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
#Shared senior authorship.
Abstract
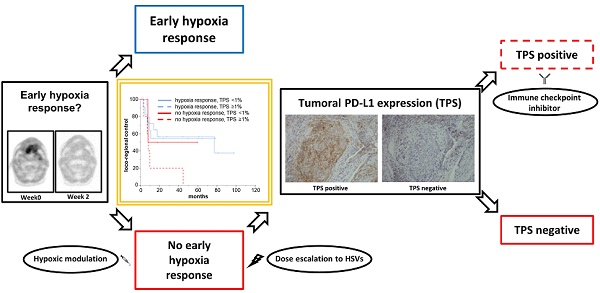
Tumor-associated hypoxia influences the radiation response of head-and-neck cancer (HNSCC) patients, and a lack of early hypoxia resolution during treatment considerably deteriorates outcomes. As the detrimental effects of hypoxia are partly related to the induction of an immunosuppressive microenvironment, we investigated the interaction between tumor hypoxia dynamics and the PD-1/PD-L1 axis in HNSCC patients undergoing chemoradiation and its relevance for patient outcomes in a prospective trial.
Methods: 49 patients treated with definitive chemoradiation for locally advanced HNSCC were enrolled in this trial and received longitudinal hypoxia PET imaging using fluorine-18 misonidazole ([18F]FMISO) at weeks 0, 2 and 5 during treatment. Pre-therapeutic tumor biopsies were immunohistochemically analyzed regarding the PD-1/PD-L1 expression both on immune cells and on tumor cells, and potential correlations between the PD-1/PD-L1 axis and tumor hypoxia dynamics during chemoradiation were assessed using Spearman's rank correlations. Hypoxia dynamics during treatment were quantified by subtracting the standardized uptake value (SUV) index at baseline from the SUV values at weeks 2 or 5, whereby SUV index was defined as ratio of maximum tumor [18F]FMISO SUV to mean SUV in the contralateral sternocleidomastoid muscle (i.e. tumor-to-muscle ratio). The impact of the PD-1/PD-L1 expression alone and in combination with persistent tumor hypoxia on locoregional control (LRC), progression-free survival (PFS) and overall survival (OS) was examined using log-rank tests and Cox proportional hazards models.
Results: Neither PD-L1 nor PD-1 expression levels on tumor-infiltrating immune cells influenced LRC (HR = 0.734; p = 0.480 for PD-L1, HR = 0.991; p = 0.989 for PD-1), PFS (HR = 0.813; p = 0.597 for PD-L1, HR = 0.796; p = 0.713 for PD-1) or OS (HR = 0.698; p = 0.405 for PD-L1, HR = 0.315; p = 0.265 for PD-1). However, patients with no hypoxia resolution between weeks 0 and 2 and PD-L1 expression on tumor cells, quantified by a tumor proportional score (TPS) of at least 1%, showed significantly worse LRC (HR = 3.374, p = 0.022) and a trend towards reduced PFS (HR = 2.752, p = 0.052). In the multivariate Cox regression analysis, the combination of absent tumor hypoxia resolution and high tumoral PD-L1 expression remained a significant prognosticator for impaired LRC (HR = 3.374, p = 0.022). On the other side, tumoral PD-L1 expression did not compromise the outcomes of patients whose tumor-associated hypoxia declined between week 0 and 2 during chemoradiation (LRC: HR = 1.186, p = 0.772, PFS: HR = 0.846, p = 0.766).
Conclusion: In this exploratory analysis, we showed for the first time that patients with both persistent tumor-associated hypoxia during treatment and PD-L1 expression on tumor cells exhibited a worse outcome, while the tumor cells' PD-L1 expression did not influence the outcomes of patients with early tumor hypoxia resolution. While the results have to be validated in an independent cohort, these findings form a foundation to investigate the combination of hypoxic modification and immune checkpoint inhibitors for the unfavorable subgroup, moving forward towards personalized radiation oncology treatment.
Keywords: head-and-neck cancer, PD-L1, hypoxia, FMISO PET, radiotherapy
 Global reach, higher impact
Global reach, higher impact