Impact Factor
Theranostics 2020; 10(21):9674-9685. doi:10.7150/thno.43829 This issue Cite
Research Paper
GSK-3β activation accelerates early-stage consumption of Hippocampal Neurogenesis in senescent mice
1. Department of Pathophysiology, Key Laboratory of Ministry of Education for Neurological Disorders, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
2. Department of Human Anatomy, School of Basic Medicine, Binzhou Medical University, Yantai 264003, China.
3. Department of Histology and Embryology, Key Laboratory of Ministry of Education of China for Neurological Disorders, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
4. Key Laboratory of Ministry of Education for Neurological Disorders, Department of Neurosurgery, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
5. Co-innovation Center of Neuroregeneration, Nantong University, Nantong 226000, China.
Abstract
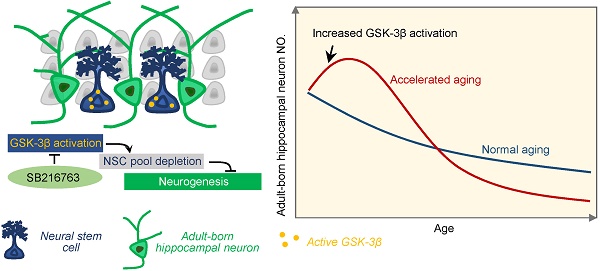
Adult hippocampal neurogenesis (AHN) deficits contribute to the progression of cognitive impairments during accelerated senescence, with the mechanistic causes poorly understood. Glycogen synthase kinase-3β (GSK-3β) is a critical regulator in prenatal neurodevelopment. The present study aims to study whether and how GSK-3β regulates AHN during the accelerated senescence.
Methods: AHN and AHN-dependent cognition and GSK-3β were evaluated in 3- and 6-month senescence-accelerated mice prone 8 (SAM-P8) and senescence resistant 1 (SAM-R1) mice, respectively. GSK-3β was selectively overexpressed in wild-type mice using adeno-associated virus, or knocked-out by crossbreeding with GSK-3β floxed mice in the neural stem cells (NSCs) of Nestin-Cre mice, or pharmacologically inhibited with SB216763 in SAM-P8 mice. AHN was evaluated by BrdU-, DCX-staining and retrovirus-labeling.
Results: AHN transiently increased at 3-month, but dramatically dropped at 6-month of age in SAM-P8 mice with a simultaneous activation of GSK-3β at 3-month. Selective overexpression of GSK-3β in hippocampal NSCs of wildtype mice induced long-term AHN deficits due to an accelerated depletion of NSC pool, although it transiently increased the proliferation and survival of the newborn neurons. Pharmacologically inhibiting GSK-3β by SB216763 efficiently preserved AHN and improved contextual memory in 6-month SAM-P8 mice, while conditional knock-out of GSK-3β in NSCs impaired AHN.
Conclusion: Early-stage activation of GSK-3β in NSCs impairs AHN by accelerating the depletion of NSC pool, and pharmacological inhibition of GSK-3β is efficient to preserve AHN during the accelerated aging. These results reveal novel mechanisms underlying the AHN impairments during accelerated senescence and provide new targets for pro-neurogenic therapies for related diseases.
Keywords: Glycogen synthase kinase-3β, Adult hippocampal neurogenesis, Senescence
 Global reach, higher impact
Global reach, higher impact