Impact Factor
Theranostics 2021; 11(1):292-303. doi:10.7150/thno.51872 This issue Cite
Research Paper
Cytogenetic and molecular aberrations and worse outcome for male patients in systemic mastocytosis
1. Department of Haematology University Medical Center Groningen, University of Groningen, Groningen, The Netherlands.
2. III. Medizinische Klinik, Universitätsmedizin Mannheim, Universität Heidelberg, Mannheim, Germany.
3. Internal Medicine, section Allergology, University Medical Center Groningen, University of Groningen, Groningen, The Netherlands.
4. Division of Hematology, Department of Medicine, Stanford University School of Medicine, Stanford, USA.
5. Division of Allergy, Department of Dermatology, University of Basel, Basel, Switzerland (KH); *Department of Biomedicine, University of Basel, Basel, Switzerland.
6. Department of Dermatology and Venerology, Uniklinik Köln, Köln, Germany.
7. Department of Allergology, Medical University of Gdansk, Gdańsk, Poland.
8. Department of Dermatology, Venereology and Allergology, Medical University of Gdansk, Gdańsk, Poland.
9. Section of Hematology, Department of Medicine, Verona University Hospital, Verona, Italy.
10. Allergy Unit, Verona University Hospital, Verona, Italy.
11. Department of Molecular Medicine and Department of Hematology Oncology, University of Pavia and Fondazione IRCCS Policlinico San Matteo, Pavia, Italy.
12. Department of Hematology, Oncology and Stem Cell Transplantation, Medical Center, Faculty of Medicine, University of Freiburg, Freiburg, Germany; German Cancer Consortium (DKTK) Partner Site Freiburg, Freiburg, Germany.
13. Department of Hematology and Oncology, Medical Center, University of Schleswig-Holstein, Campus Lübeck, Lübeck, Germany.
14. Department of Dermatology, Medical Center University of Freiburg, Faculty of Medicine, University of Freiburg, Germany; German Cancer Consortium (DKTK) Partner Site Freiburg, Freiburg, Germany.
15. Division of Allergy and Clinical Immunology, University of Salerno, Salerno, Italy.
16. Institute of Environmental Health, Medical University of Vienna, Vienna, Austria.
17. Pediatric Dermatology Unit, Department of Medicine, University of Padova, Padova, Italy.
18. Department of Dermatology and Allergy Biederstein, School of Medicine, Technical University of Munich, Munich, Germany.
19. University Clinic for Hematology and Oncology, Kepler University Hospital, Johannes Kepler University, Linz, Austria.
20. Department of Dermatology and Venereology, Allergy Centrr, Kepler University Hospital, Linz, Austria.
21. Division of Hematology, Istanbul Medical School, University of Istanbul, Istanbul, Turkey.
22. University Hospital and CEITEC Masaryk University, Brno, Czech Republic.
23. Dept Immunol, Genetics and Pathology (MM) and Dept Hematol (MM, HH), Uppsala University Hospital, Uppsala University, Uppsala, Sweden.
24. Department of Oncology, Hematology, Hemostaseology and Stem Cell Transplantation, University Hospital RWTH Aachen, Aachen, Germany.
25. Faculty of Medicine and Health Sciences, Department of Immunology-Allergology-Rheumatology, University of Antwerp and Antwerp University Hospital, Antwerpen, Belgium.
26. Department of Dermatology and Venereology, Medical University of Graz, Graz, Austria.
27. Medical Clinic and Policlinic 1, Hematology and Cellular Therapy,Leipzig University Hospital,Germany.
28. Department of Dermatology, University Hospitals Leuven, Leuven, Belgium.
29. Department of Hematology, Semmelweis University, Budapest, Hungary.
30. Division of Hematology/Oncology, Department of Medicine, University of California San Francisco, San Francisco, USA.
31. French Reference Center for Mastocytosis (CEREMAST), Hôpital Necker, Assistance Publique Hôpitauxde Paris, Imagine Institute, University Paris Descartes, Paris, France.
32. Department of Hematology, Gustave Roussy Cancer Center, Villejuif, France.
33. Laboratory of Hematology, Pitié-Salpêtrière Hospital, Paris, France.
34. Department of Internal Medicine I, Division of Hematology and Hemostaseology, Medical University of Vienna, Vienna, Austria.
35. Ludwig Boltzmann Institute for Hematology and Oncology.
Abstract
In systemic mastocytosis (SM), the clinical features and survival vary greatly. Patient-related factors determining the outcome in SM are largely unknown.
Methods: We examined the impact of sex on the clinical features, progression-free survival (PFS), and overall survival (OS) in 3403 patients with mastocytosis collected in the registry of the European Competence Network on Mastocytosis (ECNM). The impact of cytogenetic and molecular genetic aberrations on sex differences was analyzed in a subset of patients.
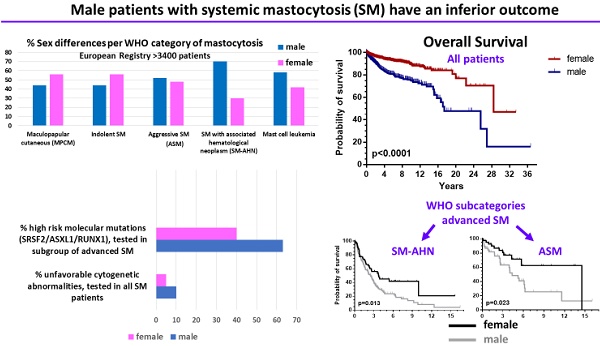
Results: Of all patients enrolled, 55.3% were females. However, a male predominance was found in a subset of advanced SM (AdvSM) patients, namely SM with an associated hematologic neoplasm (SM-AHN, 70%; p < 0.001). Correspondingly, organomegaly (male: 23% vs. female: 13%, p = 0.007) was more, whereas skin involvement (male: 71% vs. female: 86%, p = 0.001) was less frequent in males. In all patients together, OS (p < 0.0001) was significantly inferior in males, and also within the WHO sub-categories indolent SM, aggressive SM (ASM) and SM-AHN. PFS was significantly (p = 0.0002) worse in males when all patients were grouped together; due to low numbers of events, this significance persisted only in the subcategory smoldering SM. Finally, prognostically relevant cytogenetic abnormalities (10% vs. 5%, p = 0.006) or molecular aberrations (SRSF2/ASXL1/RUNX1 profile; 63% vs. 40%, p = 0.003) were more frequently present in males.
Conclusions: Male sex has a major impact on clinical features, disease progression, and survival in mastocytosis. Male patients have an inferior survival, which seems related to the fact that they more frequently develop a multi-mutated AdvSM associated with a high-risk molecular background.
Keywords: Mastocytosis, sex difference, cytogenetics, molecular mutations, survival
 Global reach, higher impact
Global reach, higher impact