13.3
Impact Factor
Theranostics 2021; 11(2):470-490. doi:10.7150/thno.51462 This issue Cite
Research Paper
Immunomodulatory role of reactive oxygen species and nitrogen species during T cell-driven neutrophil-enriched acute and chronic cutaneous delayed-type hypersensitivity reactions
1. Werner Siemens Imaging Center, Department of Preclinical Imaging and Radiopharmacy, Eberhard Karls University, Tübingen, Germany.
2. Department of Nuclear Medicine and Clinical Molecular Imaging, Eberhard Karls University, Tübingen, Germany.
3. Cluster of Excellence iFIT (EXC 2180) "Image-Guided and Functionally Instructed Tumor Therapies", Eberhard Karls University, 72076 Tübingen, Germany
4. Section of Immunology, Vetsuisse Faculty, University of Zürich, Zürich, Switzerland.
5. Interfaculty Institute for Biochemistry, Eberhard Karls University, Tübingen, Germany.
6. Department of Dermatology, Eberhard Karls University, Tübingen, Germany.
7. Department of Pathology, Eberhard Karls University, Tübingen, Germany
8. Institute for Cardiovascular Physiology, Goethe-University, Frankfurt, Germany.
9. Center for Thrombosis and Hemostasis, University Medical Center Mainz, Mainz, Germany.
10. Institute for Clinical Epidemiology and Applied Biometry, Eberhard Karls University, Tübingen, Germany
11. Department of Dermatology, Venereology and Allergology, Charité - Universitätsmedizin Berlin, Berlin, Germany.
Abstract
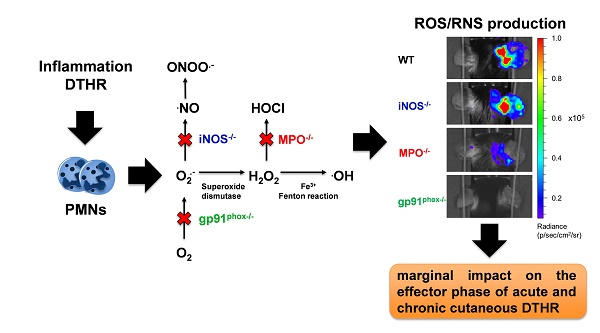
Rationale: Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are important regulators of inflammation. The exact impact of ROS/RNS on cutaneous delayed-type hypersensitivity reaction (DTHR) is controversial. The aim of our study was to identify the dominant sources of ROS/RNS during acute and chronic trinitrochlorobenzene (TNCB)-induced cutaneous DTHR in mice with differently impaired ROS/RNS production.
Methods: TNCB-sensitized wild-type, NADPH oxidase 2 (NOX2)- deficient (gp91phox-/-), myeloperoxidase-deficient (MPO-/-), and inducible nitric oxide synthase-deficient (iNOS-/-) mice were challenged with TNCB on the right ear once to elicit acute DTHR and repetitively up to five times to induce chronic DTHR. We measured ear swelling responses and noninvasively assessed ROS/RNS production in vivo by employing the chemiluminescence optical imaging (OI) probe L-012. Additionally, we conducted extensive ex vivo analyses of inflamed ears focusing on ROS/RNS production and the biochemical and morphological consequences.
Results: The in vivo L-012 OI of acute and chronic DTHR revealed completely abrogated ROS/RNS production in the ears of gp91phox-/- mice, up to 90 % decreased ROS/RNS production in the ears of MPO-/- mice and unaffected ROS/RNS production in the ears of iNOS-/- mice. The DHR flow cytometry analysis of leukocytes derived from the ears with acute DTHR confirmed our in vivo L-012 OI results. Nevertheless, we observed no significant differences in the ear swelling responses among all the experimental groups. The histopathological analysis of the ears of gp91phox-/- mice with acute DTHRs revealed slightly enhanced inflammation. In contrast, we observed a moderately reduced inflammatory immune response in the ears of gp91phox-/- mice with chronic DTHR, while the inflamed ears of MPO-/- mice exhibited the strongest inflammation. Analyses of lipid peroxidation, 8-hydroxy-2'deoxyguanosine levels, redox related metabolites and genomic expression of antioxidant proteins revealed similar oxidative stress in all experimental groups. Furthermore, inflamed ears of wild-type and gp91phox-/- mice displayed neutrophil extracellular trap (NET) formation exclusively in acute but not chronic DTHR.
Conclusions: MPO and NOX2 are the dominant sources of ROS/RNS in acute and chronic DTHR. Nevertheless, depletion of one primary source of ROS/RNS exhibited only marginal but conflicting impact on acute and chronic cutaneous DTHR. Thus, ROS/RNS are not a single entity, and each species has different properties at certain stages of the disease, resulting in different outcomes.
Keywords: DTHR, Neutrophils, ROS/RNS, NETs, acute chronic inflammation
 Global reach, higher impact
Global reach, higher impact