13.3
Impact Factor
Theranostics 2021; 11(20):10001-10011. doi:10.7150/thno.65399 This issue Cite
Research Paper
Rational design of non-toxic GOx-based biocatalytic nanoreactor for multimodal synergistic therapy and tumor metastasis suppression
1. The Department of Medical Imaging, Guangdong Second Provincial General Hospital, Guangzhou, 518037, P. R. China.
2. Key Laboratory of Materials Physics, Institute of Solid State Physics, HFIPS, Chinese Academy of Sciences, Hefei, 230031, P. R. China.
3. Division of Molecular Medicine, Hefei National Laboratory for Physical Sciences at Microscale, School of Life Sciences, University of Science and Technology of China, Hefei, 230001, P. R. China.
4. Interventional Radiology Department, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei, 230001, P. R. China.
#These authors contributed equally to this work.
Abstract
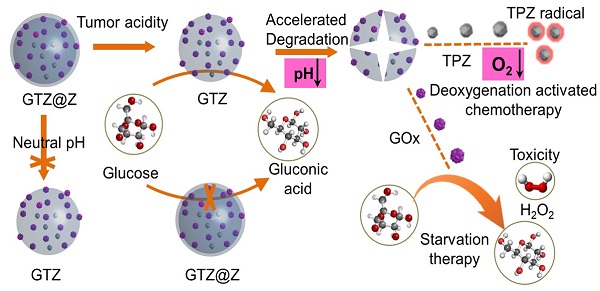
Rationale: Glucose oxidase (GOx)-based biocatalytic nanoreactors can cut off the energy supply of tumors for starvation therapy and deoxygenation-activated chemotherapy. However, these nanoreactors, including mesoporous silica, calcium phosphate, metal-organic framework, or polymer nanocarriers, cannot completely block the reaction of GOx with glucose in the blood, inducing systemic toxicity from hydrogen peroxide (H2O2) and anoxia. The low enzyme loading capacity can reduce systemic toxicity but limits its therapeutic effect. Here, we describe a real 'ON/OFF' intelligent nanoreactor with a core-shell structure (GOx + tirazapamine (TPZ))/ZIF-8@ZIF-8 modified with the red cell membrane (GTZ@Z-RBM) for cargo delivery.
Methods: GTZ@Z-RBM nanoparticles (NPs) were prepared by the co-precipitation and epitaxial growth process under mild conditions. The core-shell structure loaded with GOx and TPZ was characterized for hydrate particle size and surface charge. The GTZ@Z-RBM NPs morphology, drug, and GOx loading/releasing abilities, system toxicity, multimodal synergistic therapy, and tumor metastasis suppression were investigated. The in vitro and in vivo outcomes of GTZ@Z-RBM NPs were assessed in 4T1 breast cancer cells.
Results: GTZ@Z-RBM NPs could spatially isolate the enzyme from glucose in a physiological environment, reducing systemic toxicity. The fabricated nanoreactor with high enzyme loading capacity and good biocompatibility could deliver GOx and TPZ to the tumors, thereby exhausting glucose, generating H2O2, and aggravating hypoxic microenvironment for starvation therapy, DNA damage, and deoxygenation-activated chemotherapy. Significantly, the synergistic therapy effectively suppressed the breast cancer metastasis in mice and prolonged life without systemic toxicity. The in vitro and in vivo results provided evidence that our biomimetic nanoreactor had a powerful synergistic cascade effect in treating breast cancer.
Conclusion: GTZ@Z-RBM NPs can be used as an 'ON/OFF' intelligent nanoreactor to deliver GOx and TPZ for multimodal synergistic therapy and tumor metastasis suppression.
Keywords: Glucose oxidase, tirapazamine, metal-organic framework, biocatalytic nanoreactor, starvation therapy
 Global reach, higher impact
Global reach, higher impact