Impact Factor
Theranostics 2021; 11(20):10125-10147. doi:10.7150/thno.62525 This issue Cite
Research Paper
Peptide ligands targeting FGF receptors promote recovery from dorsal root crush injury via AKT/mTOR signaling
1. Institute of Biomedicine & Department of Cell Biology, College of Life Science and Technology, Jinan University; Guangdong Province Key Laboratory of Bioengineering Medicine; Guangdong Provincial biotechnology drug & Engineering Technology Research Center; National Engineering Research Center of Genetic Medicine, Guangzhou, Guangdong, 510632, P. R. China.
2. Guangdong-Hong Kong-Macau Institute of CNS Regeneration, Ministry of Education CNS Regeneration Collaborative Joint Laboratory, Jinan University, Guangzhou, Guangdong 510632, China.
3. School of Pharmaceutical Sciences, Health Science Centre, Shenzhen University, Shenzhen, 518060, China.
#These authors contributed to this work equally.
Abstract
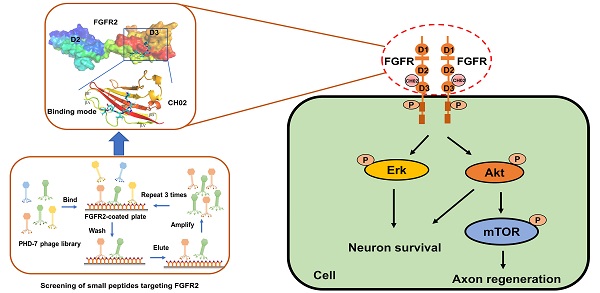
Background: Fibroblast growth factor receptors (FGFRs) are key targets for nerve regeneration and repair. The therapeutic effect of exogenous recombinant FGFs in vivo is limited due to their high molecular weight. Small peptides with low molecular weight, easy diffusion, low immunogenicity, and nontoxic metabolite formation are potential candidates. The present study aimed to develop a novel low-molecular-weight peptide agonist of FGFR to promote nerve injury repair.
Methods: Phage display technology was employed to screen peptide ligands targeting FGFR2. The peptide ligand affinity for FGFRs was detected by isothermal titration calorimetry. Structural biology-based computer virtual analysis was used to characterize the interaction between the peptide ligand and FGFR2. The peptide ligand effect on axon growth, regeneration, and behavioral recovery of sensory neurons was determined in the primary culture of sensory neurons and dorsal root ganglia (DRG) explants in vitro and a rat spinal dorsal root injury (DRI) model in vivo. The peptide ligand binding to other membrane receptors was characterized by surface plasmon resonance (SPR) and liquid chromatography-mass spectrometry (LC-MS)/MS. Intracellular signaling pathways primarily affected by the peptide ligand were characterized by phosphoproteomics, and related pathways were verified using specific inhibitors.
Results: We identified a novel FGFR-targeting small peptide, CH02, with seven amino acid residues. CH02 activated FGFR signaling through high-affinity binding with the extracellular segment of FGFRs and also had an affinity for several receptor tyrosine kinase (RTK) family members, including VEGFR2. In sensory neurons cultured in vitro, CH02 maintained the survival of neurons and promoted axon growth. Simultaneously, CH02 robustly enhanced nerve regeneration and sensory-motor behavioral recovery after DRI in rats. CH02-induced activation of FGFR signaling promoted nerve regeneration primarily via AKT and ERK signaling downstream of FGFRs. Activation of mTOR downstream of AKT signaling augmented axon growth potential in response to CH02.
Conclusion: Our study revealed the significant therapeutic effect of CH02 on strengthening nerve regeneration and suggested a strategy for treating peripheral and central nervous system injuries.
Keywords: Peptides, Fibroblast growth factor receptor, Dorsal root ganglia, Nerve regeneration, Dorsal root crush injury
 Global reach, higher impact
Global reach, higher impact