Impact Factor
Theranostics 2021; 11(8):3916-3931. doi:10.7150/thno.52496 This issue Cite
Research Paper
Engineering extracellular vesicles with platelet membranes fusion enhanced targeted therapeutic angiogenesis in a mouse model of myocardial ischemia reperfusion
1. Department of Cardiology, Zhongshan Hospital, Fudan University, Shanghai Institute of Cardiovascular Diseases, National Clinical Research Center for Interventional Medicine, 180 Feng Lin Road, Shanghai 200032, China.
2. School of Pharmacy, Fudan University, Key Laboratory of Smart Drug Delivery, Ministry of Education, 826 Zhangheng Road, Shanghai 201203, China.
3. Institute of Biomedical Science, Fudan University, 180 Feng Lin Road, Shanghai 200032, China.
4. Department of Surgical Oncology and General Surgery, First Affiliated Hospital of China Medical University, Shenyang, China.
5. Integrated Laser Microscopy System at National Facility for Protein Sciencein Shanghai, Zhangjiang Laboratory (NFPS, ZJLab), China.
* These authors contributed equally to this work.
Abstract
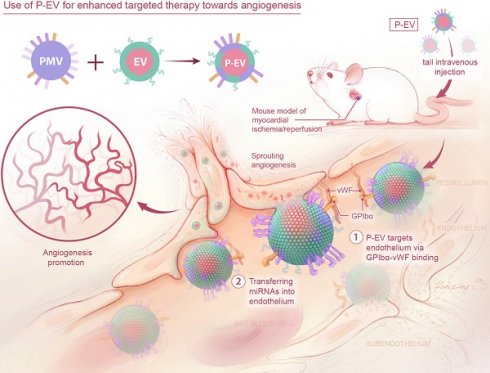
Therapeutic angiogenesis is one promising strategy for the treatment of ischemic heart disease, which is the leading cause of death globally. In recent years, extracellular vesicles (EVs) have quickly gained much attention as a cell-free approach to stimulate angiogenesis. However, clinical applications of EVs are limited by their insufficient targeting capability. Herein, we introduce a method to enhance therapeutic angiogenesis based on platelet membrane-engineered EVs.
Methods: Platelet-mimetic EVs (P-EVs) were fabricated by fusing the membranes of EVs with platelet membranes by extrusion. A mouse model of myocardial ischemia reperfusion (MI/R) was established and injected with PBS, EVs, and P-EVs to evaluate their targeting ability and therapeutic angiogenesis efficacy.
Results: P-EVs inherited the adhesive proteins and natural targeting ability to injured vasculature of platelets and retained the pro-angiogenic potential of EVs. In the MI/R model, P-EVs preferentially accumulated in the injured endothelium of the ischemic hearts and enhanced the angiogenesis potency of EVs.
Conclusions: This engineering strategy to modify pre-isolated EVs with platelet membranes by membrane fusion bestows EVs with the targeting ability of platelets and offers an exciting opportunity to design other targeted EVs fused with cell membranes from different sources for therapeutic angiogenesis.
Keywords: extracellular vesicles, platelet-mimetic, membrane fusion, targeted delivery, angiogenesis.
 Global reach, higher impact
Global reach, higher impact