13.3
Impact Factor
Theranostics 2021; 11(12):5970-5985. doi:10.7150/thno.58364 This issue Cite
Research Paper
A pain killer without analgesic tolerance designed by co-targeting PSD-95-nNOS interaction and α2-containning GABAARs
1. Department of Pharmacology, School of Pharmacy, Nanjing Medical University, China.
2. Department of Pharmacy, Nanjing First Hospital, Nanjing Medical University, China.
3. Department of Neurology, Nanjing First Hospital, Nanjing Medical University, China.
4. Department of Medicinal Chemistry, School of Pharmacy, Nanjing Medical University, China.
5. Institution of Stem Cells and Neuroregeneration, Nanjing Medical University, China.
6. Center of Drug Metabolism & Pharmacokinetics, Yantai YenePharma Co., Ltd, China.
Abstract
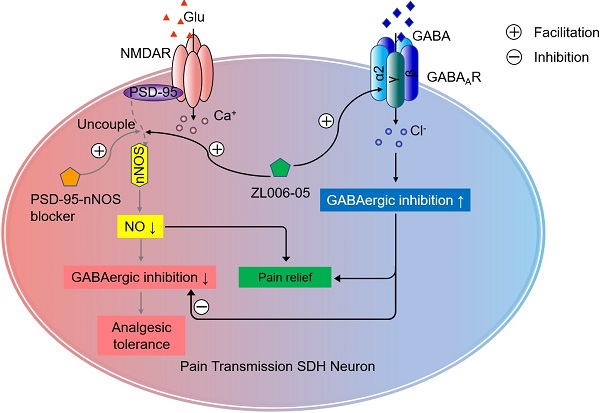
Overactivation of N-methyl-D-aspartate receptor (NMDAR) in the spinal cord dorsal horn (SDH) in the setting of injury represents a key mechanism of neuropathic pain. However, directly blocking NMDAR or its downstream signaling, interaction between postsynaptic density-95 (PSD-95) and neuronal nitric oxide synthase (nNOS), causes analgesic tolerance, mainly due to GABAergic disinhibition. The aim of this study is to explore the possibility of preventing analgesic tolerance through co-targeting NMDAR downstream signaling and γ-aminobutyric acid type A receptors (GABAARs).
Methods: Mechanical/thermal hyperalgesia were quantified to assess analgesic effects. Miniature postsynaptic currents were tested by patch-clamp recording to evaluate synaptic transmission in the SDH. GABA-evoked currents were tested on HEK293 cells expressing different subtypes of recombinant GABAARs to assess the selectivity of (+)-borneol and ZL006-05. The expression of α2 and α3 subunits of GABAARs and BDNF, and nNOS-PSD-95 complex levels were analyzed by western blotting and coimmunoprecipitation respectively. Open field test, rotarod test and Morris water maze task were conducted to evaluate the side-effect of ZL006-05.
Results: (+)-Borneol selectively potentiated α2- and α3-containing GABAARs and prevented the disinhibition of laminae I excitatory neurons in the SDH and analgesic tolerance caused by chronic use of ZL006, a nNOS-PSD-95 blocker. A dual-target compound ZL006-05 produced by linking ZL006 and (+)-borneol through an ester bond blocked nNOS-PSD-95 interaction and potentiated α2-containing GABAAR selectively. Chronic use of ZL006-05 did not produce analgesic tolerance and unwanted side effects.
Conclusion: By targeting nNOS-PSD-95 interaction and α2-containing GABAAR simultaneously, chronic use of ZL006-05 can avoid analgesic tolerance and unwanted side effects. Therefore, we offer a novel candidate drug without analgesic tolerance for treating neuropathic pain.
Keywords: neuropathic pain, central sensitization, excitatory/inhibitory synaptic transmission, analgesic tolerance, GABAA receptors
 Global reach, higher impact
Global reach, higher impact