Impact Factor
Theranostics 2021; 11(17):8218-8233. doi:10.7150/thno.59280 This issue Cite
Research Paper
GATA3 functions downstream of BRCA1 to suppress EMT in breast cancer
1. Guangdong Provincial Key Laboratory of Regional Immunity and Diseases, International Cancer Center, Marshall Laboratory of Biomedical Engineering, Shenzhen University Health Science Center, Shenzhen 518060, China.
2. Department of Pathology, Shenzhen University Health Science Center, Shenzhen 518060, China.
3. Dewitt Daughtry Family Department of Surgery, University of Miami, Miami, FL 33136, USA.
4. School of Basic Medical Sciences, Lanzhou University, Lanzhou, Gansu, 730000, China.
5. The Affiliated Cancer Hospital of Zhengzhou University, Zhengzhou, Henan 450008, China.
6. The Second Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi, 710061, China.
7. Department of Mathematics and Statistics, University of North Carolina at Greensboro, Greensboro, NC 27402, USA
8. Department of Biochemistry and Molecular Biology, International Cancer Center, Shenzhen University Health Science Center, Shenzhen 518060, China.
9. Department of Anatomy and Histology, Shenzhen University Health Science Center, Shenzhen 518060, China.
Abstract
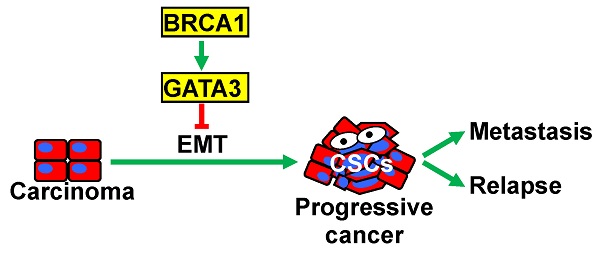
Purpose: Functional loss of BRCA1 is associated with poorly differentiated and metastatic breast cancers that are enriched with cancer stem cells (CSCs). CSCs can be generated from carcinoma cells through an epithelial-mesenchymal transition (EMT) program. We and others have previously demonstrated that BRCA1 suppresses EMT and regulates the expression of multiple EMT-related transcription factors. However, the downstream mediators of BRCA1 function in EMT suppression remain elusive.
Methods: Depletion of BRCA1 or GATA3 activates p18INK4C, a cell cycle inhibitor which inhibits mammary epithelial cell proliferation. We have therefore created genetically engineered mice with Brca1 or Gata3 loss in addition to deletion of p18INK4C, to rescue proliferative defects caused by deficiency of Brca1 or Gata3. By using these mutant mice along with human BRCA1 deficient as well as proficient breast cancer tissues and cells, we investigated and compared the role of Brca1 and Gata3 loss in the activation of EMT in breast cancers.
Results: We discovered that BRCA1 and GATA3 expressions were positively correlated in human breast cancer. Depletion of BRCA1 stimulated methylation of GATA3 promoter thereby repressing GATA3 transcription. We developed Brca1 and Gata3 deficient mouse system. We found that Gata3 deficiency in mice induced poorly-differentiated mammary tumors with the activation of EMT and promoted tumor initiating and metastatic potential. Gata3 deficient mammary tumors phenocopied Brca1 deficient tumors in the induction of EMT under the same genetic background. Reconstitution of Gata3 in Brca1-deficient tumor cells activated mesenchymal-epithelial transition, suppressing tumor initiation and metastasis.
Conclusions: Our finding, for the first time, demonstrates that GATA3 functions downstream of BRCA1 to suppress EMT in controlling mammary tumorigenesis and metastasis.
Keywords: BRCA1, GATA3, EMT, tumorigenesis, metastasis
 Global reach, higher impact
Global reach, higher impact