13.3
Impact Factor
Theranostics 2022; 12(7):3178-3195. doi:10.7150/thno.68679 This issue Cite
Research Paper
Engineering of donor-acceptor-donor curcumin analogues as near-infrared fluorescent probes for in vivo imaging of amyloid-β species
1. Key Laboratory of Structure-Based Drug Design and Discovery, Ministry of Education, Shenyang Pharmaceutical University, Shenyang, 110016, China.
2. State Key Laboratory of Analytical Chemistry for Life Sciences, Chemistry and Biomedicine Innovation Center (ChemBIC), School of Chemistry and Chemical Engineering, Nanjing University, Nanjing, 210023, China.
3. State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, China.
#These authors have contributed equally to this work.
Abstract
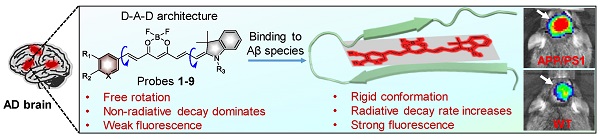
Near-infrared (NIR) fluorescent imaging of both soluble and insoluble Aβ species in the brain of Alzheimer's disease (AD) is crucial for the early diagnosis and intervention of AD. To date, a variety of NIR fluorescent probes have been reported for the detection of Aβ species. Among these probes, CRANAD-58 was reported to have the capability to detect both soluble and insoluble Aβ species, which is vital to monitor the changes of Aβ species during the pathological course of the disease. Though CRANAD-58 has shown promise to noninvasively detect Aβ species in transgenic AD mice, the emission wavelength (~670 nm) is still too short for further applications. Therefore, new probes with longer emission wavelength and improved physiological properties are in highly demand. Herein, we report the design and engineering of nine donor-acceptor-donor molecules as “off-on” near-infrared fluorescent probes for in vivo imaging of both soluble and insoluble Aβ species in living AD mice owing to its improved in vitro properties and in vivo performance.
Methods: We report a two-round strategy to develop nine “off-on” NIR fluorescence probes via structural modification of a curcumin analogue-based donor-acceptor-donor architecture. In round one, probes 1 and 2 were synthesized, and probe 2 was identified to be an optimum probe as it showed distinct “off-on” NIR fluorescence at > 690 nm upon binding to Aβ monomers, oligomers and aggregates. To further improve the in vivo performance, further structural modification of probe 2 into probes 3-9 was then conducted. The fluorescence response with Aβ species and histological staining in vitro and in vivo imaging of Aβ species in APP/PS1 transgenic AD mice and age-matched wild-type mice were performed.
Results: We demonstrate that, compared to probe 2, probe 9 with improved physiological properties hold the fastest kinetics (~10 min) to produce not only higher brain fluorescence intensity in 10-month-old APP/PS1 transgenic AD mice, but also afford a higher discrepancy in brain fluorescence to discriminate AD mice from wild-type (WT) mice. Probe 9 also hold the ability to detect soluble Aβ species in 6-month-old APP/PS1 transgenic mice. Probe 9 was further applied for dynamic visualization of Aβ plaques in a skull-thinning 14-month-old APP/PS1 mouse, which revealed its immediate penetration into brain parenchyma and selective labeling of both parenchymal and angiopathic Aβ plaques. In addition, probe 9 possessed significantly high attenuation effect on the aggregation of Aβ monomers.
Conclusion: Our results demonstrate the good potential of probe 9 for longitudinal NIR fluorescence imaging of soluble and insoluble Aβ species in APP/PS1 transgenic AD mice, which may act as a useful tool for early diagnosis and intervention of AD.
Keywords: Alzheimer's disease, Amyloid-β, NIR fluorescence probe, in vivo imaging, D-A-D molecules
 Global reach, higher impact
Global reach, higher impact