Impact Factor
Theranostics 2022; 12(7):3288-3315. doi:10.7150/thno.68667 This issue Cite
Research Paper
Surface-engineered extracellular vesicles for targeted delivery of therapeutic RNAs and peptides for cancer therapy
1. Department of Pharmacology and Institute for Digital Medicine, Yong Loo Lin School of Medicine, National University of Singapore, 16 Medical Drive, Singapore.
2. Department of Surgery, Immunology Programme, Cancer Programme and Nanomedicine Translational Programme, Yong Loo Lin School of Medicine, National University of Singapore, 1E Kent Ridge Road, Singapore.
3. Department of Biomedical Sciences, Jockey Club College of Veterinary Medicine and Life Sciences, City University of Hong Kong, Tat Chee Avenue, Kowloon, Hong Kong.
4. City University of Hong Kong Shenzhen Institute, Shenzhen, Guangdong, China.
5. Queen Mary Hospital and Department of Medicine, Li Ka Shing Faculty of Medicine, The University of Hong Kong, Pok Fu Lam, Hong Kong Island, Hong Kong.
6. Department of Clinical Oncology, Queen Elizabeth Hospital, 30 Gascoigne Rd, Kowloon, Hong Kong.
7. Department of Biochemistry and Synthetic Biology Program, Yong Loo Lin School of Medicine, National University of Singapore, 8 Medical Drive, Singapore.
*Equal contributions to this work.
#Present address: Jotbody (HK) Ltd, Science Park, New Territories, Hong Kong.
Abstract
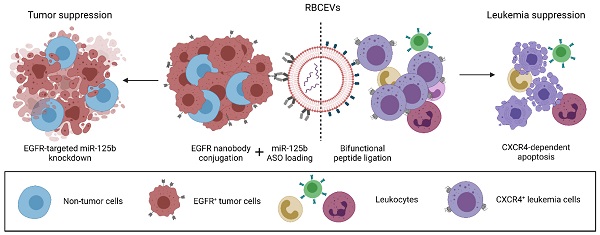
The advent of novel therapeutics in recent years has urged the need for a safe, non-immunogenic drug delivery vector capable of delivering therapeutic payloads specifically to diseased cells, thereby increasing therapeutic efficacy and reducing side effects. Extracellular vesicles (EVs) have garnered attention in recent years as a potentially ideal vector for drug delivery, taking into account their intrinsic ability to transfer bioactive cargo to recipient cells and their biocompatible nature. However, natural EVs are limited in their therapeutic potential and many challenges need to be overcome before engineered EVs satisfy the levels of efficiency, stability, safety and biocompatibility required for therapeutic use.
Here, we demonstrate that an enzyme-mediated surface functionalization method in combination with streptavidin-mediated conjugation results in efficient surface functionalization of EVs. Surface functionalization using the above methods permits the stable and biocompatible conjugation of peptides, single domain antibodies and monoclonal antibodies at high copy number on the EV surface. Functionalized EVs demonstrated increased accumulation in target cells expressing common cancer associated markers such as CXCR4, EGFR and EpCAM both in vitro and in vivo. The functionality of this approach was further highlighted by the ability of targeting EVs to specifically deliver therapeutic antisense oligonucleotides to a metastatic breast tumor model, resulting in increased knockdown of a targeted oncogenic microRNA and improved metastasis suppression. The method was also used to equip EVs with a bifunctional peptide that targets EVs to leukemia cells and induces apoptosis, leading to leukemia suppression. Moreover, we conducted extensive testing to verify the biocompatibility, and safety of engineered EVs for therapeutic use, suggesting that surface modified EVs can be used for repeated dose treatment with no detectable adverse effects. This modular, biocompatible method of EV engineering offers a promising avenue for the targeted delivery of a range of therapeutics while addressing some of the safety concerns associated with EV-based drug delivery.
 Global reach, higher impact
Global reach, higher impact